Abstract
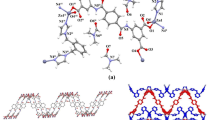
A dibasic tetradentate N3O3 Schiff base ligand {(Z)-2-hydroxy-N′-((8-hydroxyquinolin-2-yl)methylene)benzohydrazide}(H3L) and its binuclear phenoxido-bridged gadolinium complex, [Gd2(HL)2(NO3)2(DMF)4], were synthesised. The ligand was characterised by 1H-NMR, ESI-Mass, FT-IR, and UV–visible spectroscopic methods. The gadolinium complex was characterised by FT-IR, UV–visible spectroscopy, and single-crystal X-ray diffraction. The central gadolinium(III) ion is nine-coordinated, with a distorted tri-capped-trigonal-prismatic geometry. The equatorial coordination sites are satisfied by donor atoms such as phenolate oxygen in μ2 bridging modes, quinoline nitrogen, azomethine nitrogen from the 8-hydroxyquinoline-2-carbaldehyde and one carbonyl oxygen of salicyl moiety. In addition, two DMF molecules via their carbonyl oxygens and one nitrate in a bidentate fashion via O1 and O2 in the axial region complete the coordination. The gadolinium ions are separated by a distance of 4.04 Å in the complex. The Photoluminescence properties of the ligand and the gadolinium complex were studied in the solid-state at room temperature.
Graphic abstract







Similar content being viewed by others
References
A. Boussadia, A. Beghidja, L. Gali, C. Beghidja, M. Elhabiri, P. Rabu, G. Rogez, Inorg. Chim. Acta 508, 119656 (2020)
M. Albrecht, O. Osetska, R. Fröhlich, Dalton Trans. 23, 3757 (2005)
F. Rizzo, F. Meinardi, R. Tubino, R. Pagliarin, G. Dellepiane, A. Papagni, Synth. Met. 159, 356–360 (2009)
I.G. Santos, J. Sanmartín, A.M.G. Deibe, M. Fondo, E. Gómez, Inorg. Chim. Acta 363, 193 (2010)
B.S.K. Chong, E.G. Moore, Inorg. Chem. 57, 14062 (2018)
H. Zhang, L.F. Han, K.A. Zachariasse, Y.B. Jiang, Org. Lett. 7, 4217 (2005)
M. Kumar, S. Roy, M.S.H. Faizi, S. Kumar, M.K. Singh, S. Kishor, S.C. Peter, R.P. John, J. Mol. Struct. 1128, 195 (2017)
Q.H. Shi, C.L. Xue, C.J. Fan, L.L. Yan, N. Qiao, M. Fang, S.F. Wang, Polyhedron 195, 114938 (2021)
D. Dermitzaki, M. Pissas, V. Psycharis, Y. Sanakis, C.P. Raptopoulou, Polyhedron 195, 114960 (2021)
W.M. Wang, S.Y. Wang, H.X. Zhang, B. Zhao, J.Y. Zou, H.L. Gao, J.Z. Cui, Inorg. Chim. Acta 439, 106 (2016)
Y. Pan, H. Zhang, Y. Qin, Y. Ge, Y. Cui, Y. Li, W. Liu, Y. Dong, New J. Chem. 42, 5153 (2018)
Y. Lu, S. Li, S. Xu, Y. Chen, J. Liang, B. Liao, J. Solid State Chem. 289, 121475 (2020)
R.N. Soek, C.M. Ferreira, F.S. Santana, D.L. Hughes, G. Poneti, R.R. Ribeiro, F.S. Nunes, J. Mol. Struct. 1184, 254 (2019)
W.M. Wang, W.Z. Qiao, H.X. Zhang, S.Y. Wang, Y.Y. Nie, H.M. Chen, Z. Liu, H.L. Gao, J.Z. Cui, B. Zhao, Dalton Trans. 45, 8182 (2016)
N. Turan, K. Buldurun, Y. Alan, A. Savci, N. Çolak, A. Mantarcı, Res. Chem. Intermed. 45, 3525 (2019)
G.S. Kurdekar, S.M. Puttanagouda, N.V. Kulkarni, S. Budagumpi, V.K. Revankar, Med. Chem. Res. 20, 421 (2011)
E.R. Milaeva, Curr. Top. Med. Chem 11, 2703 (2011)
Z. Li, H. Yan, G. Chang, M. Hong, J. Dou, M. Niu, J. Photochem. Photobiol. B Biol. 163, 403 (2016)
W. Lu, J. Chen, J. Shi, L. Xu, S. Yang, B. Gao, J. Biol. Inorg. Chem. 26, 57 (2021)
S.A. Beyramabadi, M.S. Far, A.F. Shovey, M.J. Khoshkholgh, A. Morsali, J. Mol. Struct. 1208, 127898 (2020)
M. Umadevi, V. Muthuraj, R. Vanajothi, J. Mol. Struct. 1221, 128778 (2020)
D.J. Fanna, Y. Zhang, A. Salih, J.K. Reynolds, F. Li, J. Coord. Chem. 69, 11 (2016)
P. Ghorai, P. Brandão, S. Benmansour, C.J.G. García, A. Saha, Polyhedron 188, 114708 (2020)
T. Hata, T. Uno, I. Bull, Chem. Soc. Jpn. 45, 477 (1972)
R. Musiol, J. Jampilek, K. Kralova, D.R. Richardson, D. Kalinowski, B. Podeszwa, J. Finster, H. Niedbala, A. Palka, J. Polanski, Bioinorg. Med. Chem. 15, 1280 (2007)
C. Pan, B. Yang, F. Wang, J. Xu, G. Chen, W. Ji, Y. Huo, J. Mol. Struct. 1212, 127770 (2020)
C.H. Chen, J.M. Shi, Coord. Chem. Rev. 171, 161 (1998)
W. Paulus, Microbicides for the Protection of Materials Handbook, vol. 307 (Chapman and Hall, London, 1993)
R. Mladenova, M. Ignatova, N. Manolova, T. Petrova, I. Rashkov, Eur. Polym. J. 38, 989 (2002)
P. Baret, C.G. Beguin, H. Boukhalfa, C. Caris, J.P. Laulhere, J.L. Pierre, G. Serratrice, J. Am. Chem. Soc. 117, 9760 (1995)
B. Tummler, G. Maas, E. Weber, W. Wehner, F. Vogtle, J. Am. Chem. Soc. 99, 4683 (1977)
N.S. Ahmed, K.O. Badahdah, H.M. Qassar, Med. Chem. Res. 26, 1201 (2017)
L. Yurttaş, G.A. Çiftçi, Anticancer Agents Med. Chem. 18, 1122 (2018)
D.F. Wu, Z. Liu, P. Ren, X.H. Liu, N. Wang, J.Z. Cui, H.L. Gao, Dalton Trans. 48, 1392 (2019)
H.Y. Shen, W.M. Wang, Y.X. Bi, H.L. Gao, S. Liu, J.Z. Cui, Dalton Trans. 44, 18893 (2015)
C. Saturnino, M. Napoli, G. Paolucci, M. Bortoluzzi, A. Popolo, A. Pinto, P. Longo, Eur. J. Med. Chem. 45, 4169 (2010)
M. Haghighat, A. Naroie, A. Rezvani, M. Hakimi, H. Saravani, M. Darroudi, A. Amini, M. Sabaghan, M. Khatami, Bio Nano Sci. 11, 696–702 (2021)
Q.Y. Yang, Q.Q. Cao, Y.L. Zhang, X.F. Xu, C.X. Deng, R. Kumar, X.M. Zhu, X.J. Wang, H. Liang, Z.F. Chen, J. Inorg. Biochem 211, 111175 (2020)
H.F.E. Shafiy, M. Shebl, J. Mol. Struct. 1156, 403 (2018)
Y.C. Liu, Z.Y. Yang, J. Organomet. Chem. 664, 309 (2009)
X. Liu, L. Guo, B. Song, Z. Tang, J. Yuan, Methods Appl. Fluoresc. 5, 14009 (2017)
R.J. Palmer, J.L. Butenhoff, J.B. Stevens, Environ. Res. 43, 142 (1987)
Y.B. Zeng, N. Yang, W.S. Liu, N. Tang, J. Inorg. Biochem. 97, 258 (2003)
J.K. Barton, J.M. Goldberg, C.V. Kumar, N.J. Turro, J. Am. Chem. Soc. 108, 2081 (1986)
P. Caravan, J.J. Ellison, T.J. McMurry, R.B. Lauffer, Chem. Rev. 99, 2293 (1999)
A.E. Merbach, E. Toth, The Chemistry of Contrast Agents in Medical Magnetic Resonance Imaging (Wiley, Chichester, 2001)
N.P. Kuz’mina, S.V. Eliseeva, Russ. J. Inorg. Chem. 51, 73 (2006)
H.J. Kim, J.E. Lee, Y.S. Kim, N.G. Park, Opt. Mater. 21, 181 (2002)
G. Vicentini, L.B. Zinner, J.Z. Schpector, K. Zinner, Coord. Chem. Rev. 196, 353 (2000)
G.J.C. Bünzli, C. Piguet, Chem. Rev. 102, 1897 (2002)
K. Binnemans, Chem. Rev. 109, 4283 (2009)
Y. Hasegawa, Y. Wada, S. Yanagida, J. Photochem. Photobiol. C Photochem. Rev. 5, 183 (2004)
S. Faulkner, J.L. Matthews, Comprehensive Coordination Chemistry II, Elsevier, Oxford, UK 9, 913 (2004)
H. Tsukube, S. Shinoda, H. Tamiaki, Chem. Rev. 102, 227 (2002)
E.G. Moore, A.P.S. Samuel, K.N. Raymond, Acc. Chem. Res. 42, 542 (2009)
M.A. Katkov, M.N. Bochkarev, Dalton Trans. 39, 6599 (2010)
C. Benelli, D. Gatteschi, Chem. Rev. 102, 2369 (2002)
P. Zhou, Y.G. Zhao, Y. Bai, K.L. Pang, C. He, Inorg. Chim. Acta. 360, 3965 (2007)
B. Samanta, J. Chakraborty, S. Shit, S.R. Batten, P. Jensen, J.D. Masuda, S. Mitra, Inorg. Chim. Acta. 360, 2471 (2007)
P. Barbazan, R. Carballo, E.M.V. Lopez, Cryst. Eng. Comm. 9, 668 (2007)
M.G. Matos, P.S. Calefi, K.J. Ciuffi, E.J. Nassar, Inorg. Chim. Acta 375, 63 (2011)
S.M. Ying, X.R. Zeng, X.N. Fang, X.F. Li, D.S. Liu, Inorg. Chim. Acta 359, 1589 (2006)
Y.C. Liu, Z.Y. Yang, J. Organomet. Chem. 694, 3091 (2009)
L. Chen, C. Yan, B.B. Du, K. Wu, L.Y. Zhang, S.Y. Yin, M. Pan, Inorg. Chem. Commun. 47, 13 (2014)
L.V. de Freitas, C.C.P. da Silva, J. Ellena, L.A.S. Costa, N.A. Rey, Spectrochim. Acta A Mol. Biomol. Spectrosc. 116, 41 (2013)
SAINT, Bruker AXS, (Madison, WI, 1999)
G.M. Sheldrick, (University of Gottingen, Gottingen, Germany, 1997)
G.M. Sheldrick, SHELXTL Version 5.10 (University of Gottingen, Gottingen, 1997)
G.M. Sheldrick, Acta Crystallogar. A 64, 112 (2008)
G.M. Sheldrick, T.R. Schneider, Method Enzymol. 277, 319 (1997)
L.J. Farrugia, J. Appl. Crystallogr. 32, 837 (1999)
M. Kumar, A. Kumar, M.K. Singh, S.K. Sahu, R.P. John, Sens. Actuators B 241, 1218 (2017)
M. Kumar, S. Kishor, A. Kumar, A.K. Bhagi, R.P. John, J. Mol. Struct. 1199, 126996 (2020)
M.T. Kaczmarek, R. Jastrza, M. Kubicki, M. Gierszewski, M. Sikorski, Inorg. Chim. Acta 430, 108 (2015)
R. Golbedaghi, S. Moradi, S. Salehzadeh, A.G. Blackman, J. Mol. Struct. 1108, 727 (2016)
S.T. Hatscher, W. Urland, Angew. Chem. Int. Ed. 42, 2862 (2003)
D. John, W. Urland, Eur. J. Inorg. Chem. 2005, 4486 (2005)
Y. Makita, M. Inoue, N. Katayama, H.-H. Lee, T. Abe, C. Inui-Yamamoto, Y. Mori, Y. Yoshioka, Y. Honda, S. Gamoh, K. Shimizutani, S.-I. Fujiwara, A. Ogawa, Polyhedron 107, 148 (2016)
M. Albrecht, O. Osetska, R. Fröhlich, Dalton Trans. 7, 3757 (2005)
P. Alemany, E. Bernuz, A. Carreras, M. Llunell, Cosymlib: a Python library for continuous symmetry measures (v0.9.5). Zenodo. (2021)
D. Casanova, P. Alemany, J.M. Bofill, S. Alvarez, Chem. Eur. J. 9, 6 (2003)
A.R. Martınez, D. Casanova, S. Alvarez, Dalton Trans. 45, 2583 (2008)
Y.C. Liu, Z.Y. Yang, Eur. J. Med. Chem. 44, 5080 (2009)
Y.C. Liu, Y.Y. Li, H.L. Qi, K.J. Zhang, R.X. Lei, J.N. Liu, J. Coord. Chem 67, 3689 (2014)
Y.C. Liu, Z.Y. Yang, Biometals 22, 733 (2009)
X.F. Guan, J.X. Shen, X.Y. Hu, Y. Yang, X. Han, J.Q. Zhao, J. Wang, Y. Shi, W.M. Wang, Polyhedron 166, 17 (2019)
M.M. Moawad, W.G. Hanna, J. Coord. Chem. 55, 439 (2002)
T.M.A. Ismail, J. Coord. Chem. 58, 141 (2005)
G. Huang, P. Yang, N. Wang, J.Z. Wu, Y. Yu, Inorg. Chim. Acta 384, 333 (2012)
T.V. Balashova, A.P. Pushkarev, V.A. Ilichev, M.A. Lopatin, M.A. Katkova, E.V. Baranov, G.K. Fukin, M.N. Bochkarev, Polyhedron 50, 112 (2013)
A.W. Woodward, A. Frazer, A.R. Morales, J. Yu, A.F. Moore, A.D. Campiglia, K.D. Belfield, Dalton Trans. 43, 16626 (2014)
J. Vančo, Z. Trávníček, O. Kozák, R. Boča, Int. J. Mol. Sci. 16, 9520 (2015)
H.A.R. Pramanik, C. Chanda, P.C. Paul, C.R. Bhattacharjee, S.K. Prasad, D.S.S. Rao, J. Mol. Struct. 1180, 472 (2019)
L. Zhang, Y. Ji, X. Xu, Z. Liu, J. Tang, J. Lumin. 132, 1906 (2012)
W. Wen, X. Yawen, J. Yin, Z. Wang, Asian J. Org. Chem. 25, 8307 (2013)
A. Pui, T. Malutan, L. Tataru, C. Malutan, D. Humelnicu, G. Carja, Polyhedron 30, 2127 (2011)
C.R. Bhattacharjee, G. Das, P. Goswami, P. Mondal, S.K. Prasad, D.S.S. Rao, Polyhedron 30, 1040 (2011)
Acknowledgements
Author Mukesh Kumar thank the Indian Institute of Technology (ISM) Dhanbad for providing financial assistance as a Research Fellowship. The authors are thankful to Dr S. K Sharma and Navneet Kumar, Department of Physics, Indian Institute of Technology (ISM) Dhanbad, for providing the photoluminescence analysis facility. The authors are also grateful to the Sophisticated Analytical Instrument Facility, Punjab University Chandigarh, for providing the NMR and mass spectral facilities.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kumar, M., Kumar, A., Manav, N. et al. A binuclear gadolinium complex of 8-hydroxyquinoline-2-carbaldehyde salicylhydrazone: structural characterisation and photoluminescence properties. Res Chem Intermed 47, 5119–5133 (2021). https://doi.org/10.1007/s11164-021-04589-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-021-04589-2