Abstract
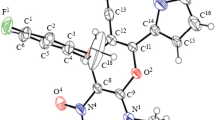
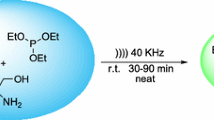
We demonstrate here a rapid and comfortable synthetic sequence to benzothiadiazinone dioxide (six-membered cyclic sulfamide) derivatives, analogues of Monastrol, which is known to be a specific mitotic kinesin Eg5 inhibitor. Fourteen novel compounds (1d–14d) were synthesized through three component Biginelli reaction, by condensation of aromatic aldehyde, sulfamide, and cyclohexane-1,3-dione. A wide range of substrates is compatible in this reaction, producing excellent yields (82–92%) in short time (5–10 min). The reaction is performed under solvent-free, catalysed by H2SO4/CH3COOH (1/9) using microwave irradiation. The structure of the obtained substances was confirmed by 1H, 13C, HMBC, HSQC NMR spectroscopy, IR spectroscopy, and mass spectrometry, as well as elemental analysis. A single crystal of the (4-phenyl-4,6,7,8-tetrahydro-1H benzo[c][1,2,6]thiadiazin-5(3H)-one 2,2-dioxide has been obtained after recrystallization in ethanol/acetone (9/1) and selected for X-ray study.
Graphic abstract









Similar content being viewed by others
References
R.D. Vale, Cell 112, 467 (2003)
A.J. Kim, S.A. Endow, J. Cell Sci. 113, 3681 (2000)
G.D. Marconi, S. Carradori, A. Ricci, P. Guglielmi, A. Cataldi, S. Zara, Molecules 24, 3948 (2019)
B. Nyamaa, H.K. Kim, Y.J. Jeong, I.S. Song, J. Han, J Lipid Atheroscler. 3, 63 (2014)
C. Perez-Melero, Curr. Top. Med. Chem. 14, 2286 (2014)
T.U. Mayer, T.M. Kapoor, S.J. Haggarty, R.W. King, S.L. Schreiber, T.J. Mitchison, Science 286, 971 (1999)
P.J. Coleman, M.E. Fraley, Expert Opin. Ther. Patents 14, 1659 (2004)
M. Gartner, N. Sunder-Plassmann, J. Seiler, M. Utz, I. Vernos, T. Surrey, A. Giannis, ChemBioChem 6, 1173 (2005)
H.O. Tawfik, M.H. El-Hamamsy, N.A. Sharafeldin, T.F. El-Moselhy, Bioorg. Med. Chem. 27, 115126 (2019)
V. Sarli, A. Giannis, ChemMedChem 1, 293 (2006)
L. Terrab, C.J. Rosenker, L. Johnstone, L.K. Ngo, L. Zhang, N.F. Ware, B. Miller, A.Z. Topacio, S. Sannino, J.L. Brodsky, P. Wipf, A.C.S. Med, Chem. Lett. 11, 984 (2020)
J. Hulten, H.O. Andersson, W. Schaal, H.U. Danielson, B. Classon, I. Kvarnstro, A. Karle, T. Unge, B. Samuelson, A. Hallberg, J. Med. Chem. 42, 4054 (1999)
P.K. Jadhav, F.J. Woerner, Tetrahedron. Lett. 36, 6383 (1995)
A. Bendjeddou, H. Djebbar, M. Berredjem, Z. Hattab, Z. Regainia, N. Aouf, Phosphorus Sulfur Silicon Relat. Elem. 181, 1381 (2006)
R. Kuang, J.B. Epp, S. Ruan, L.S. Chong, J.T. Venkataraman, S. He, T.M. Truong, W.C. Groutas, Bioorg. Med. Chem. 8, 1005 (2000)
W. Schaal, A. Karlsson, G. Ahlse, J. Lindberg, H.O. Andersson, U.H. Danieson, B. Classon, T. Unge, B. Samuelsson, J. Hulten, A. Hallberg, A. Karle, J. Med. Chem. 44, 155 (2001)
K. Bäckbro, S. Löwgren, K. Osterlund, J. Atepo, T. Unge, J. Hulte, N.M. Bonham, W. Schaal, A. Karle, A. Hallberg, J. Med. Chem. 40, 898 (1997)
W.C. Groutas, S. He, R. Kuang, S. Ruan, J. Tu, H.T. Chan, Bioorg. Med. Chem. 9, 1543 (2001)
S. He, R. Kuang, R. Venkataraman, J. Tu, T.M. Truong, H.K. Chan, W.C. Groutas, Bioorg. Med. Chem. 8, 1713 (2000)
L. Ducry, S. Reinelt, P. Seiler, F. Diederich, D.R. Bolin, R.M. Campbell, G.M. Olson, Helv. Chim. Acta 82, 2432 (1999)
P. Biginelli, P. Gazz, Chim. Ita. 23, 360 (1893)
C.O. Kappe, Tetrahedron 49, 69 (1993)
C.O. Kappe, S.F. Falsone, Synlett. 718 (1998)
S. Xue, Y.C. Shen, Y.L. Li, X.M. Shen, Q.X. Guo, Chin. J. Chem. 20, 385 (2002)
J. Lu, H. Ma, Synlett. 63 (2000)
E.H. Hu, D.R. Sidler, U.H. Dolling, J. Org. Chem. 63, 3454 (1998)
J. Lu, Y. Bai, Z. Wang, B. Yang, H. Ma, Tetrahedron Lett. 41, 9075 (2000)
D. Dallinger, C.O. Kappe, Nat. Protoc. 2, 321 (2007)
Y. Ma, C. Qian, L. Wang, M. Yang, J. Org. Chem. 65, 3864 (2000)
B.C. Ranu, A. Hazra, U. Jana, J. Org. Chem. 65, 6270 (2000)
D.S, Bose, R.K, Kumar, L. Fatima, Synlett. 279 (2004)
D.S. Bose, M. Sudharshan, S.W. Chavhan, Arkivoc iii, 228 (2005)
S.K. De, R.A. Gibbs, Synth. 1748 (2005)
E. Rafiee, H. Jafari, Bioorg. Med. Chem. Lett. 16, 2463 (2006)
N.Y. Fu, Y.F. Yauan, Z. Cao, S.W. Wang, J.T. Wang, C. Pepple, Tetrahedron 58, 4801 (2002)
O.M. Portilla-Zuñiga, Á.G. Sathicq, J.J. Martínez, S.A. Fernandes, T.R.M. Rezende, G.P. Romanelli, Sustain Chem. Pharm. 10, 50 (2018)
V.R. Choudhary, V.H. Tillu, V.S. Narkhede, H.B. Borate, R.D. Wakharkar, CatalCommun. 4, 449 (2003)
A.C. Boukis, B. Monney, M.A.R. Meier, Beilstein J. Org. Chem. 13, 54 (2017)
B. Belhani, M. Berredjem, M. Le Borgne, Z. Bouaziz, J. Lebreton, N.E. Aouf, RSC Adv. 49, 39324 (2015)
A. Bouzina, N.E. Aouf, M. Berredjem, Res. Chem. Intermed. 42, 5993 (2016)
G.A. Patani, E.J. LaVoie, Chem. Rev. 96, 3147 (1996)
H. Kagechika, E. Kawachi, Y. Hashimoto, K. Shudo, Chem. Pharm. Bull. 34, 2275 (1986)
H. Kagechika, T. Himi, E. Kawachi, Y. Hashimoto, K. Shudo, J. Med. Chem. 32, 2182 (1989)
M.L. Vazquez, M.L. Bryant, M. Clare, G.A. DeCrescenzo, E.L. Doherty, J.N. Freskos, D.P. Getman, K.A. Houseman, J.A. Julien, G.P. Kocan, R.A. Mueller, H.S. Shieh, W.C. Stallings, R.A. Stegman, J.J. Talley, J. Med. Chem. 38, 581 (1995)
J.N. Freskos, D.E. Bertenshaw, D.P. Getman, R.M. Heintz, B.V. Mischke, L.W. Blystone, M.L. Bryant, C. Funckes-Shippy, K.A. Houseman, N.N. Kishore, G.P. Kocan, P.P. Mehta, Bioorg. Med. Chem. Lett. 6, 445 (1996)
T.C. McMorris, R. Chimmani, K. Alisala, M.D. Staake, G. Banda, M.J. Kelner, J. Med. Chem. 53, 1109 (2010)
M.C. Burla, R. Caliandro, M. Camalli, B. Carrozzini, G.L. Cascarano, L. de Caro, C. Giacovazzo, G. Polidori, R. Spagna, J. Appl. Crystallogr. 38, 381 (2005)
G.M. Sheldrick, ActaCryst. 64, 112 (2008)
L.J. Farrugia, J. Appl. Cryst. 45, 849 (2012)
A. Bouzina, B. Belhani, N.E. Aouf, M. Berredjem, RSC Adv. 57, 46272 (2015)
A. Bouzina, M. Berredjem, S. Bouacida, H. Merazig, N.E. Aouf, RSC Adv. 5, 99775 (2015)
S. Lakrout, H. K’Tir, A. Amira, M. Berredjem, N.E. Aouf, RSC Adv. 4, 16027 (2014)
Acknowledgements
This work was supported financially by The General Directorate for Scientific Research and Technological Development (DG-RSDT), Algerian Ministry of Scientific Research, Applied Organic Chemistry Laboratory (FNR 2000).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
About this article
Cite this article
Bouzina, A., Berredjem, M., Belhani, B. et al. Microwave-accelerated multicomponent synthesis and X-ray characterization of novel benzothiadiazinone dioxide derivatives, analogues of Monastrol. Res Chem Intermed 47, 1359–1376 (2021). https://doi.org/10.1007/s11164-020-04378-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-020-04378-3