Abstract
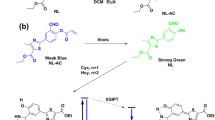
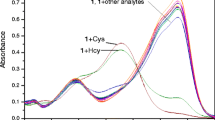
Biothiols of cysteine (Cys), homocysteine (Hcy), glutathione (GSH) and hydrogen polysulfides (H2Sn, n > 1) play vital roles in physiological and pathological processes. In this manuscript, a fluorescent probe (compound 1) for simultaneous detection and discrimination of Cys/Hcy, GSH and H2Sn was developed, employing fluorophore of 1-(benzo[d]thiazol-2-yl)naphthalen-2-oxy linked with 7-nitrobenzo-2-oxa-1,3-diazole (NBD) as a response unit and a quencher. Compound 1 exhibited different ratiometric colorimetric responses toward GSH at 430 nm and 375 nm from Cys/Hcy at 480 nm and 375 nm, while it displayed a different colorimetric response at 561 nm toward H2Sn. Meanwhile it could act as a naked-eye probe toward GSH, Cys/Hcy and H2Sn with corresponding solution color of light yellow, orange and purple, respectively. Upon excitation at 370 nm, compound 1 responded to all these biothiols with 'turn-on' blue fluorescence, whereas upon excitation at 470 nm, it could respond to Cys/Hcy with 'turn-on' green fluorescence. Data of mass spectra, lifetimes and 1H NMR spectra interpreted the aromatic nucleophilic substitutions of compound 1 by GSH, Cys/Hcy and H2Sn to produce fluorophore1-(benzo[d]thiazol-2-yl)naphthalen-2-ol (compound 2) and corresponding NBD derivatives which could be discriminated through their absorption and fluorescence signals as well as solution colors. Furthermore, compound 1 was applied to image Cys/Hcy and H2Sn/GSH in 786-O cells.










Similar content being viewed by others
References
T.V. Mishanina, M. Libiad, R. Banerjee, Nat. Chem. Biol. 11, 457 (2015)
C.E. Paulsen, K.S. Carroll, Chem. Rev. 113, 4633 (2013)
Y. Fang, W. Chen, W. Shi, H.Y. Li, M. Xian, H.M. Ma, Chem. Commun. 53, 8759 (2017)
R. Wang, Physiol. Rev. 92, 791 (2012)
Q.Q. Wan, Y.C. Song, Z. Li, X.H. Gao, H.M. Ma, Chem. Commun. 49, 502 (2013)
L.A. Montoya, M.D. Pluth, Anal. Chem. 88, 5769 (2016)
W. Chen, C.R. Liu, B. Peng, Y. Zhao, A. Pacheco, M. Xian, Chem. Sci. 4, 2892 (2013)
J.I. Toohey, A.J.L. Cooper, Molecules 19, 12789 (2014)
K. Ono, T. Akaike, T. Sawa, Y. Kumagai, D.A. Wink, D.J. Tantillo, A.J. Hobbs, P. Nagy, M. Xian, J. Lin, J.M. Fukuto, Free Radic. Biol. Med. 77, 82 (2014)
H. Kimura, Antioxid. Redox Signal 22, 362 (2015)
B.L. Predmore, D.J. Lefer, G. Gojon, Antioxid. Redox Signal 17, 119 (2012)
L. Zeng, S. Chen, T. Xia, W. Hu, C. Li, Z. Liu, Anal. Chem. 87, 3004 (2015)
H. Shang, H. Chen, Y. Tang, R. Guo, W. Lin, Sens. Actuators B Chem. 230, 773 (2016)
M. Gao, F. Yu, H. Chen, L. Chen, Anal. Chem. 87, 3631 (2015)
L. Fu, C. Chen, W. Chen, J. Huang, J. Xiao, L. Yang, J. Sheng, X. Song, Sens. Actuators B Chem. 304, 127382 (2020)
C.R. Liu, W. Chen, W. Shi, B. Peng, Y. Zhao, H.M. Ma, M. Xian, J. Am. Chem. Soc. 136, 7257 (2014)
Q. Fang, X. Yue, S. Han, B. Wang, X. Song, Spectrochim. Acta Part A 224, 117410 (2020)
L. Liang, W. Li, J. Zheng, R. Li, H. Chen, Z. Yuan, Biomater. Sci. 8, 224 (2020)
J. Zhang, X. Zhu, X. Hu, H. Liu, J. Li, L. Feng, X. Yin, X. Zhang, W. Tan, Anal. Chem. 88, 11892 (2016)
K. Li, F. Chen, Q. Yin, S. Zhang, W. Shi, D. Han, Sens. Actuators B 254, 222 (2018)
N. Gupta, S.I. Reja, V. Bhalla, M. Kumar, Org. Biomol. Chem. 15, 6692 (2017)
P. Xie, Y. Zhu, X. Huang, G. Gao, F. Guo, G. Yang, Res. Chem. Intermed. 44, 2823 (2018)
S. Singh, D. Padovani, R.A. Leslie, T. Chiku, R. Banerjee, J. Biol. Chem. 284, 22457 (2009)
L.-Y. Niu, Y.-Z. Chen, H.-R. Zheng, L.-Z. Wu, C.-H. Tung, Q.-Z. Yang, Chem. Soc. Rev. 44, 6143 (2015)
S.Y. Zhang, C.-N. Ong, H.-M. Shen, Cancer Lett. 208, 143 (2004)
D.M. Townsend, K.D. Tew, H. Tapiero 57, 145 (2003)
M. Kemp, Y.M. Go, D.P. Jones, Free Radical Biol. Med. 44, 921 (2008)
F. Wang, L. Zhou, C. Zhao, R. Wang, Q. Fei, S. Luo, Z. Guo, H. Tian, W.-H. Zhu, Chem. Sci. 6, 2584 (2015)
J. Liu, Y.-Q. Sun, H. Zhang, Y. Huo, Y. Shi, W. Guo, Chem. Sci. 5, 3183 (2014)
W. Chen, X. Yue, H. Zhang, W. Li, L. Zhang, Q. Xiao, C. Huang, J. Sheng, X. Song, Anal. Chem. 89, 12984 (2017)
H.-J. Xiang, H.P. Tham, M.D. Nguyen, S.Z.F. Phua, W.Q. Lim, J.-G. Liu, Y. Zhao, Chem. Commun. 53, 5220 (2017)
X. Gao, X. Li, L. Li, J. Zhou, H. Ma, Chem. Commun. 51, 9388 (2015)
W. Chen, H. Luo, X. Liu, J.W. Foley, X. Song, Anal. Chem. 88, 3638 (2016)
M.D. Hammers, M.D. Pluth, Anal. Chem. 86, 7135 (2014)
L. Yang, Y. Su, Y. G., Y. Zhang, X. Ren, L. He, X. Song, ACS Sens. 3, 1863 (2018)
H. Zhang, X. Xia, H. Zhao, G.-N. Zhang, D.-Y. Jiang, X.-Y. Xue, J. Zhang, Dyes Pigments 163, 183 (2019)
L. Zhai, Z. Shi, Y. Tu, S. Pu, Dyes Pigments 165, 164 (2019)
L. Zhai, Y. Tu, Z. Shi, S. Pu, Spectrochim. Acta Part A 218, 171 (2019)
H. Zhu, C. Liu, R. Yuan, R. Wang, H. Zhang, Z. Li, P. Jia, B. Zhu, W. Sheng, Analyst 144, 4258 (2019)
M. Yang, J. Fan, W. Sun, J. Du, S. Long, X. Peng, Dyes Pigments 168, 189 (2019)
F. Qi, Y. Zhang, B. Wang, W. Chen, L. Yang, Z. Yang, X. Song, Sens. Actuators B 296, 126533 (2019)
Z. Lu, Y. Lu, C. Fan, X. Sun, M. Zhang, Y. Lu, J. Mater. Chem. B 6, 8221 (2018)
Y.-X. Liao, M.-D. Wang, K. Li, Z.-X. Yang, J.-T. Hou, M.-Y. Wu, Y.-H. Liu, X.-Q. Yu, RSC Adv. 5, 18275 (2015)
J. Ma, J. Fan, H. Li, Q. Yao, F. Xu, J. Wang, X. Peng, J. Mater. Chem. B 5, 2574 (2017)
L. He, X. Yang, K. Xu, X. Kong, W. Lin, Chem. Sci. 8, 6257 (2017)
R. Kawagoe, I. Takashima, S. Uchinomiya, A. Ojida, Chem. Sci. 8, 1134 (2017)
S. Uchiyama, T. Santa, T. Fukushima, H. Homma, K. Imai, J. Chem. Soc. Perkin Trans. 2(2), 2165 (1998)
Y.H. Chen, J.C. Tsai, T.H. Cheng, S.S. Yuan, Y.M. Wang, Biosens. Bioelectron. 56, 117 (2014)
L.Y. Niu, H.R. Zheng, Y.Z. Chen, L.Z. Wu, C.H. Tung, Q.Z. Yang, Analyst 139, 1389 (2014) 48. D. Lee, G. Kim, J. Yin, J. Yoon, Chem. Commun. 51, 6518 (2015)
Z. Ye, C. Duan, Q. Hu, Y. Zhang, C. Qin, L. Zeng, J. Mater. Chem. B 5, 3600 (2017)
D. Kand, T. Saha, P. Talukdar, Sens. Actuators B 196, 440 (2014)
Acknowledgements
The authors thank the National Natural Science Foundation of China (21702047, 81700578), Scientific and Technological Project of the Henan Province of China (192102110053), and Henan International Joint Laboratory of Laser Technology in Agricultural Sciences for Financial Support.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Xie, P., Zhu, Y., Wang, Y. et al. A fluorescent probe for discrimination of cysteine/homocysteine, glutathione and hydrogen polysulfides. Res Chem Intermed 47, 1123–1138 (2021). https://doi.org/10.1007/s11164-020-04320-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-020-04320-7