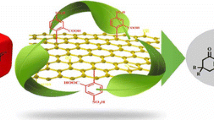
Abstract
A practical and efficient method for the synthesis of thiazolidine derivatives via Nef-isocyanide three-component reaction using supported phosphoric acid on graphene oxide (GO-H2PO4) is described. In this study, a relatively simple method starting with cyclic and acyclic thiourea was employed. The catalyst was characterized using FTIR, scanning electron microscopy, energy-dispersive X-ray spectroscopy, and X-ray diffraction techniques. Using a combination of acetonitrile as solvent and GO-H2PO4 as a catalytic system, the best results were obtained. All products were characterized using melting point, FTIR, MASS, 1H NMR, and 13C NMR techniques. The use of commercially available chemicals, reusability of the catalyst, high yields, decreased environmental hazards, with no need for the separation of stereoisomers, and, consequently, a reduced number of overall steps are the advantages of this approach that make it an appropriate choice at an increased scale.










Similar content being viewed by others
References
A. Domling, Chem. Rev. 106, 17 (2006)
S. Sadjadi, M.M. Heravi, N. Nazari, RSC Adv. 6, 53203 (2016)
S. Long, Chem. Soc. Rev. 42, 4867 (2013)
J.U. Nef, Justus Liebigs Ann. Chem. 270, 267 (1892)
F.L. Spisa, G.C. Tron, L.E. Kaim, Synthesis 46, 829 (2014)
S. Ghorbani, E. Soleimani, L.E. Kaim, Tetrahedron Lett. 58, 4595 (2017)
A.S. Ripka, D.D. Diaz, K.B. Sharpless, M.G. Finn, Org. Lett. 5, 1531 (2003)
I. Yavari, R. Pashazadeh, R. Hosseinpour, Helv. Chim. Acta 95, 169 (2012)
I. Yavari, R. Hosseinpour, R. Pashazadeh, Synlett 23, 1662 (2012)
R. Lesyk, B. Zimenkovsky, Curr. Org. Chem. 8, 1547 (2004)
R. Sharma, P. Samadhiya, S. Srivastava, S. Srivastava, Latv. J. Chem. 50, 296 (2012)
D. Prasad, A. Kumar, S.P. Kumar, M. Nath, Org. Med. Chem. Lett. 14, 2 (2011)
K.M. Amin, D.E. Rahman, Y.A. Al-Eryani, Bioorg. Med. Chem. 16, 5377 (2008)
Y. Liu, F. Jing, Y. Xu, Y. Xie, F. Shi, H. Fangm, M. Lim, W. Xu, W. Bioorg. Med. Chem. 19, 2342 (2011)
A.M. Isloor, D. Sunil, P. Shetty, S. Malladi, K.S.R. Pai, N. Maliyakki, Med. Chem. Res. 22, 758 (2013)
J. Balzarini, B. Orzeszko, J.K. Maurin, A. Orzeszko, Eur. J. Med. Chem. 42, 993 (2007)
A.K.M. Iqbal, A.Y. Khan, M.B. Kalashetti, N.S. Belavagi, Y.D. Gong, I.A. Khazi, Eur. J. Med. Chem. 53, 308 (2012)
G. Derosa, P. Maffioli, Diabetes Technol. Ther. 12, 491 (2010)
H.G. Alvim, J.R. Correa, J.A. Assumpção, W.A. da Silva, M.O. Rodrigues, J.L. de Macedo, M. Fioramonte, F.C. Gozzo, C.C. Gatto, B.A. Neto, J. Org. Chem. 83, 4044 (2018)
L. Zeng, B. Huang, Y. Shen, S. Cui, Org. Lett. 20, 3460 (2018)
M.M. Khan, S. Khan, S. Iqbal, RSC adv. 6, 42045 (2016)
M.M. Khan, R. Yousuf, S. Khan, RSC Adv. 5, 57883 (2015)
I. Yavari, R. Pashazadeh, R. Hosseinpour, E. Ghanbari, Helv. Chim. Acta 54, 2785 (2013)
I. Yavari, R. Pashazadeh, R. Hosseinpour, E. Ghanbari, Helv. Chim. Acta 96, 2191 (2013)
L. Salehitabar, I. Yavari, Phosphorus Sulfur Silicon 92, 1195 (2017)
R. Akbarzadeh, T. Amanpour, H.R. Khavasi, A. Bazgir, Tetrahedron 70, 169 (2014)
D. He, C. Zhang, G. Zeng, Y. Yang, D. Huang, L. Wang, H. Wang, Appl. Catal. B Environ. 258, 117957 (2019)
Y. Yang, C. Zhang, D. Huang, G. Zeng, J. Huang, C. Lai, C. Zhou, W. Wang, H. Guo, W. Xue, R. Deng, Appl. Catal. B Environ. 245, 87 (2019)
M.H. Ghasemi, E. Kowsari, Res. Chem. Intermed. 42, 7963 (2016)
M.H. Ghasemi, E. Kowsari, A. Shafiee, Tetrahedron Lett. 57, 1150 (2016)
M.H. Ghasemi, E. Kowsari, Res. Chem. Intermed. 43, 3691 (2017)
H.D. Hanoon, E. Kowsari, M. Abdouss, H. Zandi, M.H. Ghasemi, Res. Chem. Intermed. 43, 1751 (2017)
H.D. Hanoon, E. Kowsari, M. Abdouss, M.H. Ghasemi, H. Zandi, Res. Chem. Intermed. 43, 4023 (2017)
W.S. Hummers, R.E. Offeman, J. Am. Chem. Soc. 80, 1339 (1958)
E. Kowsari, A. Zare, V. Ansari, Int. J. Hydrog. Energy 40, 13964 (2015)
Acknowledgements
The authors wish to gratefully thank the Research Affairs Division at Amir Kabir University of Technology (AUT), Tehran, for financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Daghlavi, A., Kowsari, E., Abdouss, M. et al. Catalytic synthesis of thiazolidines by the reaction of Nef-isocyanide reaction. Res Chem Intermed 46, 3593–3605 (2020). https://doi.org/10.1007/s11164-020-04163-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-020-04163-2