Abstract
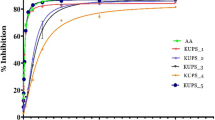
Firstly, thiosemicarbazides were prepared by the reaction of hydrazine monohydrate with isothiocyanates in cold dry ethanol at 0 °C for 1 h. After that, new isatin-β-thiosemicarbazones were synthesized by treatment of 5-methoxyisatin with thiosemicarbazides in aqueous ethanol containing one drop of hydrochloric acid at reflux for 3 h. The chemical structures of products were confirmed by means of IR, 1H NMR, and 13C NMR data and by elemental analysis. All the synthesized compounds were evaluated for their antioxidant activity by 1,1-diphenyl-2-picryl hydrazyl (DPPH) radical scavenging method. The synthesized molecules showed lower antioxidant activity than the standard Trolox (8.757 μM). IC50 values of the newly synthesized isatin-β-thiosemicarbazone derivatives ranged from 12.455 to 73.471 μM.
Graphic abstract








Similar content being viewed by others
References
M.C. Liu, T.S. Lin, J.G. Cory, A.H. Cory, A.C. Sartorelli, J. Med. Chem. 39, 2586 (1996)
P. Tarasconi, S. Capacchi, G. Pelosi, M. Cornia, R. Albertini, A. Bonati, P.P. Dall’Aglio, P. Lunghi, S. Pinelli, Bioorg. Med. Chem. 8, 157 (2000)
G.K. Kumar, G.E. Chavarria, A.K. Charlton-Sevcik, W.M. Arispe, M.T. MacDonough, T.E. Strecker, S.E. Chen, B.G. Siim, D.J. Chaplin, M.L. Trawick, Bioorg. Med. Chem. Lett. 20, 1415 (2010)
N. Fujii, J.P. Mallari, E.J. Hansell, Z. Mackey, P. Doyle, Y. Zhou, J. Gut, P.J. Rosenthal, J.H. McKerrow, R.K. Guy, Bioorg. Med. Chem. Lett. 15, 121 (2005)
W.X. Hu, W. Zhou, C.N. Xia, X. Wen, Bioorg. Med. Chem. Lett. 16, 2213 (2006)
M.S. Almutairi, A.S. Zakaria, P.P. Ignasius, R.I. Al-Wabli, I.H. Joe, M.I. Attia, J. Mol. Struct. 1153, 333 (2018)
Z. Bakherad, M. Safavi, A. Fassihi, H. Sadeghi-Aliabadi, M. Bakherad, H. Rastegar, J.B. Ghasemi, S. Sepehri, L. Saghaie, M. Mahdavi, Res. Chem. Intermed. 45, 2827 (2019)
T.R. Bal, B. Anand, P. Yogeeswari, D. Sriram, Bioorg. Med. Chem. Lett. 15, 4451 (2005)
D. Banerjee, P. Yogeeswari, P. Bhat, A. Thomas, M. Srividya, D. Sriram, Eur. J. Med. Chem. 46, 106 (2011)
J. Dimmock, J. McColl, S. Wonko, R. Thayer, D. Hancock, Eur. J. Med. Chem. 26, 529 (1991)
A. Kshirsagar, M.P. Toraskar, V.M. Kulkarni, S. Dhanashire, V. Kadam, Int. J. Chem. Tech. Res 1, 696 (2009)
D.L. Klayman, J.P. Scovill, J.F. Bartosevich, C.J. Mason, J. Med. Chem. 22, 1367 (1979)
R.B. de Oliveira, E.M. de Souza-Fagundes, R.P. Soares, A.A. Andrade, A.U. Krettli, C.L. Zani, Eur. J. Med. Chem. 43, 1983 (2008)
I.H. Hall, S. Chen, K. Rajendran, D. West, Appl. Organomet. Chem. 10, 485 (1996)
H. Pervez, N. Manzoor, M. Yaqub, A. Khan, K.M. Khan, F.U.H. Nasim, M.I. Choudhary, Lett. Drug Des. Discov. 7, 102 (2010)
C. Shipman Jr., S.H. Smith, J.C. Drach, D.L. Klayman, Antimicrob. Agents Chemother. 19, 682 (1981)
M. Bingul, E. Şenkuytu, M.F. Saglam, M. Boga, H. Kandemir, I.F. Sengul, Res. Chem. Intermed. 45, 4487 (2019)
H. Pervez, M.S. Iqbal, M.Y. Tahir, F.U.H. Nasim, M.I. Choudhary, K.M. Khan, J. Enzyme Inhib. Med. Chem. 23, 848 (2008)
H. Govender, C. Mocktar, H.M. Kumalo, N.A. Koorbanally, Phosphorus Sulfur Silicon Relat. Elem. 194, 1074 (2019)
Z.H. Chohan, H. Pervez, A. Rauf, K.M. Khan, C.T. Supuran, J. Enzyme Inhib. Med. Chem. 19, 417 (2004)
S.Y. Abbas, A.A. Farag, Y.A. Ammar, A.A. Atrees, A.F. Mohamed, A.A. El-Henawy, Monatsh. Chem. 144, 1725 (2013)
S. Dilber, M. Saban, A. Gelineo, L. Arsenijević, M. Bogavac, S. Pavlov, Pharmazie 45, 800 (1990)
A. Patel, S. Bari, G. Talele, J. Patel, M. Sarangapani, Iran. J. Pharm. Sci. 5, 249 (2010)
A. Andreani, S. Burnelli, M. Granaiola, A. Leoni, A. Locatelli, R. Morigi, M. Rambaldi, L. Varoli, M.A. Cremonini, G. Placucci, Eur. J. Med. Chem. 45, 1374 (2010)
J. Haribabu, G. Subhashree, S. Saranya, K. Gomathi, R. Karvembu, D. Gayathri, J. Mol. Struct. 1110, 185 (2016)
T. Aboul-Fadl, F.A. Bin-Jubair, Int. J. Res. Pharm. Sci. 1, 113 (2010)
D. Sriram, P. Yogeeswari, K. Meena, Pharmazie 61, 274 (2006)
S.N. Pandeya, A.S. Raja, J.P. Stables, J. Pharm. Pharm. Sci. 5, 266 (2002)
K. Tadele, J. Pharm. Med. Res. 3, 73 (2017)
W. Brand-Williams, M.-E. Cuvelier, C. Berset, Lwt-Food Sci. Technol. 28, 25 (1995)
N. Naik, H. Vijay Kumar, P.B. Vidyashree, J. Pharm. Res. 4, 2686 (2011)
H.P. Ebrahimi, J.S. Hadi, T.A. Alsalim, T.S. Ghali, Z. Bolandnazar, Spectrochim Acta A Mol. Biomol. Spectrosc. 137, 1067 (2015)
D. Williams, I. Fleming, Spectroscopic Methods in Organic Chemistry (McGraw-Hill, New York, 1996)
F. Shahidi, Handbook of Antioxidants for Food Preservation (Elsevier, Amsterdam, 2015), pp. 1–14
G. Kiran, T. Maneshwar, Y. Rajeshwar, M. Sarangapani, J. Chem. (2013). https://doi.org/10.1155/2013/192039
A. Božić, N. Filipović, I. Novakovic, S. Bjelogrlić, J. Nikolić, S. Drmanić, A. Marinković, J. Serb. Chem. Soc. 82, 5 (2017)
G. Kiran, M. Sarangapani, T. Gouthami, A.R. Narsimha Reddy, Toxicol. Environ. Chem. 95, 367 (2013)
P. Pakravan, S. Kashanian, M.M. Khodaei, F.J. Harding, Pharmacol. Rep. 65, 313 (2013)
H. Sudhamani, S.K. Thaslim Basha, S. Adam, B. Vijay Bhaskar, C. Naga Raju, Monatsh. Chem. 148, 1525 (2017)
M. AlGhorbania, N.D. Rekhab, V. Lakshmi Ranganathaa, T. Prashantha, T. Veerabasappagowdab, S.A. Khanuma, Russ. J. Bioorg. Chem. 41, 554 (2015)
K.M. Khan, M. Khan, N. Ambreen, F. Rahim, B. Muhammad, S. Ali, S.M. Haider, S. Perveen, M. Choudhary, J. Pharm. Res 4, 10 (2011)
Acknowledgements
I would like to thank the Scientific Research Center for Industrial and Technological Applications and Research Centre (BETUM) and Dr. Hatice Karadeniz for taking the NMR spectra.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Muğlu, H. Synthesis, characterization, and antioxidant activity of some new N4-arylsubstituted-5-methoxyisatin-β-thiosemicarbazone derivatives. Res Chem Intermed 46, 2083–2098 (2020). https://doi.org/10.1007/s11164-020-04079-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-020-04079-x