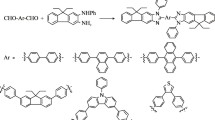
Abstract
The reaction of the free amine group in polyaryl-substituted imidazole structures with phenyl isocyanate or dimethyl acetylenedicarboxylate gave two new series of polyaryl-substituted imidazoles: biaryl ureas or vinyl esters, respectively. Besides their spectroscopic analysis, we explored the optical and electrochemical properties of these highly conjugated scaffolds. Comparing these properties in two categories of products yielded interesting results.
Graphic abstract





Similar content being viewed by others
References
T.M. Figueira-Duarte, K. Mullen, Chem. Rev. 11, 7260 (2011)
E. Holder, B.M.W. Langeveld, U.S. Schubert, Adv. Mater. 17, 1109 (2005)
J.H. Burroughes, D.D. Bradley, A.R. Brown, R.N. Marks, K. Mackay, R.H. Friend, P.L. Burns, A.B. Holmes, Nature 347(6293), 539 (1990)
M.D. McGehee, A.J. Heeger, Adv. Mater. 12, 1655 (2000)
A.R. Murphy, J.M.J. Frechet, Chem. Rev. 107, 1066 (2007)
B. Crone, A. Dodabalapur, A. Gelperin, L. Torsi, H.E. Katz, A.J. Lovinger, Z. Bao, Appl. Phys. Lett. 78, 2229 (2001)
T. Someya, H.E. Katz, A. Gelperin, A.J. Lovinger, A. Dodabalapur, Appl. Phys. Lett. 81, 3079 (2002)
T. Kowada, J. Kituta, A. Kubo, M. Ishii, H. Maeda, S. Mizukami, K. Kikuchi, J. Am. Chem. Soc. 133, 17772 (2011)
S. Yao, H.Y. Ahn, X.H. Wang, J. Fu, E.W.V. Stryland, D.J. Hagan, K.D. Belfield, J. Org. Chem. 75, 3965 (2010)
N. Fu, Y.J. Xiong, T.C. Squier, J. Am. Chem. Soc. 134, 18530 (2012)
Y.L. Wang, T.L. Liu, L.Y. Bu, J.F. Li, C. Yang, X.J. Li, Y. Tao, W.J. Yang, J. Phys. Chem. 116, 15576 (2012)
Y.J. Cheng, J.D. Luo, S. Hau, D.H. Bale, T.D. Kim, Z.W. Shi, D.B. Lao, N.M. Tucker, Y.Q. Tian, L.R. Dalton, P.J. Reid, A.K.Y. Jen, Chem. Mater. 19, 1154 (2007)
D.J. Armitt, G.T. Crisp, J. Org. Chem. 71, 3417 (2006)
H. Jin, X. Li, T. Tan, S. Wang, Y. Xiao, J. Tian, Dyes Pigments 106, 154 (2014)
R.E. Martin, F. Diederich, Angew. Chem. Int. Ed. 38, 1350 (1999)
J.L. Segura, N. Martín, J. Mater. Chem. 10, 2403 (2000)
J. Kulhánek, F. Bureš, Beilstein J. Org. Chem. 8, 25 (2012)
Z.-P. Chen, H.-B. Wang, Y.-Q. Wang, Q.-H. Zhu, Y. Xie, S.-W. Liu, Tetrahedron 29, 4379 (2014)
G. Choudhary, R.K. Peddinti, Green Chem. 13, 3290 (2011)
M. Lashkari, M.T. Maghsoodlou, M. Karima, M. Kangani, J. Chil. Chem. Soc. 63, 3799 (2018)
J. Liu, W. Wei, T. Zhao, X. Liu, J. Wu, W. Yu, J. Chang, J. Org. Chem. 81, 9326 (2016)
W. Liu, L. Tan, P. Zhou, C. Chen, Q. Zhang, Synlett 24, 991 (2013)
J. Pan, X. Li, F. Lin, J. Liu, N. Jiao, Chem 6, 1427 (2018)
Y. Wang, C.-M. Jiang, H.-L. Li, F.-S. He, X. Luo, W.-P. Deng, J. Org. Chem. 81, 8653 (2016)
F. Huang, P. Wu, L. Wang, J. Chen, C. Sun, Z. Yu, J. Org. Chem. 79, 10553 (2014)
R. Sharma, R.A. Vishwakarma, S.B. Bharate, Adv. Synth. Catal. 358, 3027 (2016)
C. Zheng, Y. Wang, R. Fan, Org. Lett. 17, 916 (2015)
X. Duan, X. Kong, X. Zhao, K. Yang, H. Zhou, D. Zhou, Y. Zhang, J. Liu, J. Ma, N. Liu, Z. Wang, Tetrahedron Lett. 57, 1446 (2016)
F.D. Lewis, T.L. Kurth, W. Liu, Photochem. Photobiol. Sci. 1, 30 (2002)
J.K. Dannecker, H. Rust, Cryst. Struct. Commun. 8, 429 (1979)
M.C. Etter, T.W. Panunto, J. Am. Chem. Soc. 110, 5896 (1988)
P. Ganis, G. Avitabile, E. Benedetti, C. Pedone, M. Goodman, Proc. Natl. Acad. Sci. U. S. A. 67, 426 (1970)
H. Kagechika, I. Azumaya, K. Yamaguchi, K. Shudo, Chem. Pharm. Bull. 44, 460 (1996)
K. Mori, A. Kanie, Y. Horiguchi, K. Isobe, Heterocycles 51, 2377 (1999)
R.R. Schmidt, J. Kast, H. Speer, Synthesis 9, 725 (1983)
A.Z. Halimehjani, M.R. Saidi, Tetrahedron Lett. 49, 1244 (2008)
N.D. Heindel, I.S. Bechara, P.D. Kennewell, J. Molnar et al., J. Org. Chem. 11, 1218 (1968)
D. Zewge, C. Chen, C. Deer, P.G. Dormer, D.L. Hughes, J. Org. Chem. 72, 4276 (2007)
R. Hosseinzadeh, Y. Sarrafi, M. Mohadjerani, F. Mohammadpourmir, Tetrahedron Lett. 49, 840 (2008)
R. Hosseinzadeh, M. Tajbakhsh, M. Mohadjerani, H. Mehdinejad, Synlett 9, 1517 (2004)
R. Hosseinzadeh, M. Tajbakhsh, M. Mohadjerani, M. Alikarami, Synlett 7, 1101 (2005)
R. Hosseinzadeh, M. Tajbakhsh, M. Alikarami, Tetrahedron Lett. 47, 5203 (2006)
A.G. Sergeev, G.A. Artamkina, I.P. Beletskaya, Tetrahedron Lett. 44, 4719 (2003)
S.N. Gavade, R.S. Balaskar, M.S. Mane, P.N. Pabrekar, M.S. Shingare, D.V. Mane, Chin. Chem. Lett. 22, 292 (2011)
R. Hosseinzadeh, Y. Sarrafi, N. Aghili, Chin. Chem. Lett. 21, 1171 (2010)
A.A. Fakhree, Z. Ghasemi, A. Shahrisa, H. Mostafavi, ChemistrySelect 4, 2959 (2019)
Z. Ghasemi, A. Mirzaie, R. Arabzadeh, Z. Fathi, A. Abolghassemi Fakhree, J. Chem. Res. 43, 262 (2019)
Z. Ghasemi, Z. Fathi, Res. Chem. Intermediates. 43, 3131 (2017)
Z. Ghasemi, Z. Zakeri, M. Allahvirdinesbat, Turk. J. Chem. 40, 729 (2016)
H. Kessler, H. Oschkinat, C. Griesinger, W.J.J. Bermel, Magn. Reson. 70, 106 (1986)
J. Stonehouse, P. Adell, J. Keeler, A.J. Shaka, J. Am. Chem. Soc. 116, 6037 (1994)
K. Stott, J. Stonehouse, J. Keeler, T.L. Hwang, A.J. Shaka, J. Am. Chem. Soc. 117, 4199 (1995)
R. Hülnhagen, H. Baumgärtel, J. Electroanal. Chem. 98, 119 (1979)
K. Pekmez, H. Özyöruk, A. Yildiz, Ber. Bunsenges. Phys. Chem. 96, 1805 (1992)
T. Sakamoto, T. Nagano, Y. Kondo, H. Yamanaka, Synthesis 3, 215 (1990)
Y. Iwanami, Bull. Chem. Soc. Jpn. 44, 1311 (1971)
S.K. Khetan, J.G. Hiriyakkanavar, M.V. George, Tetrahedron 24, 1567 (1968)
Acknowledgements
Authors thank the research affairs of the University of Tabriz for their financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Fakhree, A.A., Mohammadzadeh, R., Alipour, E. et al. Synthesis of new polyaryl-substituted imidazoles bridged on enamine or urea moieties and evaluation of their optical and electrochemical properties. Res Chem Intermed 46, 2069–2082 (2020). https://doi.org/10.1007/s11164-020-04078-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-020-04078-y