Abstract
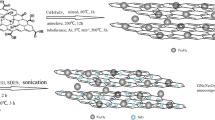
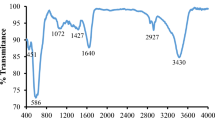
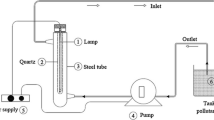
Magnetic ZnFe2O4@MnO–graphene oxide and ZnFe2O4@MnO–reduced graphene oxide nanocomposites were prepared via a facile co-precipitation and hydrothermal methods and characterized by X-ray powder diffraction, diffuse reflectance UV–Vis spectroscopy, photoluminescence (PL) spectra, Transmission electron microscopy, field emission scanning electron microscopy, Fourier transform infrared spectroscopy, vibrating sample magnetometry techniques and Bruner–Emmett–Teller (BET). The ZnFe2O4@MnO, ZnFe2O4@MnO–GO and ZnFe2O4@MnO–rGO nanoparticles were found to have a size of 20–40 nm and were spread out on the graphene oxide nanosheets and reduced graphene oxide nanosheets. Magnetic studies demonstrated that the ZnFe2O4@MnO–graphene oxide and ZnFe2O4@MnO–reduced graphene oxide nanocomposites can be easily separated from the solution by an external magnetic field. The photocatalytic degradation of Congo red dye (CR) was evaluated based on the removal of CR in aqueous solution in 35 min of visible light irradiation. The photocatalytic activity was affected by the structural and optical properties as well as the surface area of the samples. Compared with pure ZnFe2O4@MnO and ZnFe2O4@MnO–reduced graphene oxide nanocomposite, the ZnFe2O4@MnO–graphene oxide nanocomposite displayed a high photocatalytic activity on the photodegradation of Congo red. The prepared ZnFe2O4@MnO–graphene oxide nanocomposite can be potentially applied as a visible light responsive catalyst and magnetically separable photocatalyst and thus as a powerful separation tool for solving water pollution problems.





























Similar content being viewed by others
References
M. Batool, M.Z. Qureshi, F. Hashmi, N. Mehboob, A.S. Shah, Indones. J. Chem. 19(3), (2019)
R.S. Das, S.K. Warkhade, A. Kumar, A.V. Wankhade, Res. Chem. Intermed. 45, 1 (2019)
G.M. Walker, L.R. Weatherley, Water Res. 31(8), 2093 (1997)
B. Acemioğlu, J. Colloid Interface Sci. 274(2), 371 (2004)
E.S. Agorku, A.T. Kuvarega, B.B. Mamba, A.C. Pandey, A.K. Mishra, J. Rare Earths 33(5), 498 (2015)
S.O. Oppong, W.W. Anku, S.K. Shukla, P.P. Govender, Res. Chem. Intermed. 43(1), 481 (2017)
P. Bakhtkhosh, A. Mehrizad, J. Mol. Liq. 240, 65 (2017)
F. Tran, P. Blaha, Phys. Rev. Lett. 102(22), 226401 (2009)
X. Liu, C. Chen, Y. Zhao, B. Jia, J. Nanomater. 2013, 1 (2013)
M.M.A. Sinthiya, K. Ramamurthi, S. Mathuri, T. Manimozhi, N. Kumaresan, M.M. Margoni, P.C. Karthika, Int. J. Chem. Tech. Res. 7, 2144 (2015)
M. Gurumoorthy, K. Parasuraman, M. Anbarasu, K. Balamurugan, Nano Vis. 5, 63 (2015)
H. Zhang, X. Lv, Y. Li, Y. Wang, J. Li, ACS Nano 4(1), 380 (2009)
L. Sun, J. Li, C. Wang, S. Li, Y. Lai, H. Chen, C. Lin, J. Hazard. Mater. 171(1–3), 1045 (2009)
H. Huang, D. Li, Q. Lin, W. Zhang, Y. Shao, Y. Chen, X. Fu, Environ. Sci. Technol. 43(11), 4164 (2009)
L. Huang, F. Peng, H. Wang, H. Yu, Z. Li, Catal. Commun. 10(14), 1839 (2009)
R.S. Das, S.K. Warkhade, A. Kumar, A.V. Res, Chem. Intermed. 45, 1 (2019)
V.K. Gupta, T. Eren, N. Atar, M. Lütfi Yola, C. Parlak, H. Karimi-Maleh, J. Mol. Liq. 208, 122 (2015)
E.S. Agorku, A.C. Pandey, B.B. Mamba, A.K. Mishra, Mater. Today Proc. 2(7), 3909 (2015)
M. Mzoughi, W.W. Anku, S.O. Oppong, S.K. Shukla, E.S. Agorku, P.P. Govender, Adv. Mater. Lett. 7(12), 946 (2016)
S.B. Narde, R.B. Lanjewar, S.M. Gadegone, M.R. Lanjewar, Der Pharma Chem. 9(7), 115 (2017)
F. Ciesielczyk, W. Szczekocka, K. Siwińska-stefańska, A. Piasecki, Open Chem. 15, 7 (2017)
A. Thakur, S. Kumar, V.S. Rangra, Synthesis of reduced graphene oxide (rGO) via chemical reduction. in AIP Conference Proceedings, vol. 1661, no. 1, p. 080032 (2015)
C. Suwanchawalit, V. Somjit, Dig. J. Nanomater. Biostruct. 10, 769 (2015)
B. Aslibeiki, P. Kameli, H. Salamati, J. Nanoparticle Res. 1–12, 15 (2013)
F.Y. Ban, S.R. Majid, N.M. Huang, H.N. Lim, Int. J. Electrochem. Sci. 7(5), 4345 (2012)
V. Loryuenyong, K. Totepvimarn, P. Eimburanapravat, W. Boonchompoo, A. Buasri, Adv. Mater. Sci. Eng. 2013, 1 (2013)
K. Krishnamoorthy, G.-S. Kim, S.J. Kim, Ultrason. Sonochem. 20, 644 (2013)
M.G. Naseri, E.B. Saion, Crystalization in spinel ferrite nanoparticles. In Advances in Crystallization Processes. IntechOpen (2012)
P. Sathishkumar, R.V. Mangalaraja, S. Anandan, M. Ashokkumar, Chem. Eng. J. 220, 302 (2013)
H. Guo, J. Chen, W. Weng, Q. Wang, S. Li, Chem. Eng. J. 239, 192 (2014)
K. Woan, G. Pyrgiotakis, W. Sigmund, Adv. Mater. 21(21), 2233 (2009)
R. Leary, A. Westwood, Carbon 49(3), 741 (2011)
P.V. Kamat, M. Gevaert, K. Vinodgopal, J. Phys. Chem. B 101(22), 4422 (1997)
W. Wang, P. Serp, P. Kalck, J.L. Faria, J. Mol. Catal. A: Chem. 235(1–2), 194 (2005)
Y. Zhang, N. Zhang, Z.R. Tang, Y.J. Xu, ACS Nano 6(11), 9777 (2012)
S.O.B. Oppong, W.W. Anku, S.K. Shukla, P.P. Govender, Res. Chem. Intermed. 43, 481 (2017)
R. Karthik, J.V. Kumar, S.-M. Chen, P.S. Kumar, V. Selvam, V. Muthuraj, Sci. Rep. 7, 7254 (2017)
R. Arunadevi, B. Kavitha, M. Rajarajan, A. Suganthi, A. Jeyamurugan, Surfaces Interfaces 10, 32 (2018)
D. Wang, X. Li, J. Chen, X. Tao, Chem. Eng. J. 198, 547 (2012)
K. Ullah, L. Zhu, Z.D. Meng, S. Ye, Q. Sun, W.C. Oh, Chem. Eng. J. 231, 76 (2013)
Acknowledgements
We thank science and research Branch, Islamic Azad University Tehran for supporting this study and Iran Nanotechnology Initiative.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zamani, A., Sadjadi, M.S., Mahjoub, A. et al. Synthesis, characterization and investigation of photocatalytic activity of ZnFe2O4@MnO–GO and ZnFe2O4@MnO–rGO nanocomposites for degradation of dye Congo red from wastewater under visible light irradiation. Res Chem Intermed 46, 33–61 (2020). https://doi.org/10.1007/s11164-019-03934-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-019-03934-w