Abstract
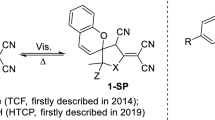
An approach to the synthesis of novel group of negative photochromes has been developed based on the interaction of the tricycanofuran TCF or hydroxytricyanopyrrole HTCP acceptors with methoxy-substituted 2-hydroxybenzaldehydes in ethanol in the presence of ammonium acetate. An essential part of the obtained photochromes is a combination of a phenol fragment and a butadienetricarbonitrile BDTC acceptor connected via a vinylene bridge. The synthesized compounds are sensitive to visible light T-type photochromes. A transformation degree of the initial form, as well as the rate of the reverse dark reaction, can be varied by changing both the position of a methoxy group at aromatic ring and the type of the heterocycle with the BDTC fragment.









Similar content being viewed by others
References
W. Szymanski, J.M. Beierle, H.A.V. Kistemaker, W.A. Velema, B.L. Feringa, Chem. Rev. 113, 6114 (2013)
W.A. Velema, W. Szymanski, B.L. Feringa, J. Am. Chem. Soc. 136, 2178 (2014)
J. Broichhagen, J.A. Frank, D. Trauner, Acc. Chem. Res. 48, 1947 (2015)
M. Dong, A. Babalhavaeji, S. Samanta, A.A. Beharry, G.A. Woolley, Acc. Chem. Res. 48, 2662 (2015)
M.M. Lerch, M.J. Hansen, G.M. van Dam, W. Szymanski, B.L. Feringa, Angew. Chem. Int. Ed. 55, 10978 (2016)
M.M. Russew, S. Hecht, Adv. Mater. 22, 3348 (2010)
H. Tian, J. Zhang, Photochromic Materials: Preparation, Properties and Applications (Wiley-VCH, Weinheim, 2016)
R. Klajn, Chem. Soc. Rev. 43, 148 (2014)
L. Wang, Q. Li, Chem. Soc. Rev. 47, 1044 (2018)
M. Natali, S. Giordani, Chem. Soc. Rev. 41, 4010 (2012)
S. Erbas-Cakmak, D.A. Leigh, C.T. McTernan, A.L. Nussbaumer, Chem. Rev. 115, 10081 (2015)
T. Yamaguchi, A. Maity, V. Polshettiwar, M. Ogawa, Inorg. Chem. 57, 3671 (2018)
M.J. Feeney, S.W. Thomas, Macromolecules 51, 8027 (2018)
W. Tian, J. Tian, Dyes Pigm. 105, 66 (2014)
G. Copley, J.G. Gillmore, J. Crisman, G. Kodis, C.L. Gray, B.R. Cherry, B.D. Sherman, P.A. Liddell, M.M. Raquette, L. Kelbauskas, N.L. Frank, A.L. Moore, T.A. Moore, D. Gust, J. Am. Chem. Soc. 136, 11994 (2014)
S. Hatano, T. Horino, A. Tokita, T. Oshima, J. Abe, J. Am. Chem. Soc. 135, 3164 (2013)
T. Yamaguchi, Y. Kobayashi, J. Abe, J. Am. Chem. Soc. 138, 906 (2016)
I. Yonekawa, K. Mutoha, J. Abe, Chem. Commun. 55, 1221 (2019)
C. Bohne, R.H. Mitchell, J. Photochem. Photobiol. C Photochem. Rev. 12, 126 (2011)
K. Ayub, R. Li, C. Bohne, R.V. Williams, R.H. Mitchell, J. Am. Chem. Soc. 133, 4040 (2011)
K. Ayub, R.H. Mitchell, J. Org. Chem. 79, 664 (2014)
S. Helmy, F.A. Leibfarth, S. Oh, J.E. Poelma, C.J. Hawker, J. Read de Alaniz, J. Am. Chem. Soc. 136, 8169 (2014)
M.M. Lerch, M.D. Donato, A.D. Laurent, M. Medved, A. Iagatti, L. Bussotti, A. Lapini, W.J. Buma, P. Foggi, W. Szymański, B.L. Feringa, Angew. Chem. Int. Ed. 57, 1863 (2018)
M.D. Donato, M.M. Lerch, A. Lapini, A.D. Laurent, A. Iagatti, L. Bussotti, S.P. Ihrig, M. Medved, D. Jacquemin, W. Szymański, W.J. Buma, P. Foggi, B.L. Feringa, J. Am. Chem. Soc. 139, 15596 (2017)
M.M. Lerch, W. Szymański, B.L. Feringa, Chem. Soc. Rev. 47, 1910 (2018)
S. Aiken, R.J.L. Edgar, C.D. Gabbutt, B.M. Heron, P.A. Hobson, Dyes Pigm. 149, 92 (2018)
V.A. Barachevsky, Rev. J. Chem. 7, 334 (2017)
C. Yang, T. Khalil, Y. Liao, RSC Adv. 6, 85420 (2016)
V.K. Johns, P. Peng, J. DeJesus, Z. Wang, Y. Liao, Chem. Eur. J. 20, 689 (2014)
M.Yu. Belikov, S.V. Fedoseev, M.Yu. Ievlev, O.V. Ershov, Synth. Commun. 48, 2850 (2018)
S.V. Fedoseev, M.Yu. Belikov, M.Yu. Ievlev, O.V. Ershov, V.A. Tafeenko, Res. Chem. Intermed. 44, 3565 (2018)
W.E. Moerner, R.J. Twieg, D.W. Kline, M. He, Patent US 2007:134737
Acknowledgements
The research was performed under the financial support of the Russian Science Foundation (Grant No. 18-73-10065).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Belikov, M.Y., Ievlev, M.Y., Fedoseev, S.V. et al. Novel group of negative photochromes containing a nitrile-rich acceptor: synthesis and photochromic properties. Res Chem Intermed 45, 4625–4636 (2019). https://doi.org/10.1007/s11164-019-03853-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-019-03853-w