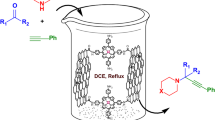
Abstract
A covalently cross-linked graphene oxide (GO) catalyst was prepared by a cross-linking process using nucleophilic reaction of copper(II)‐coordinated 5,10,15,20-tetrakis(aminophenyl)porphyrin (CuPPh) with carboxyl groups at the edges of GO (GO–CuPPh). The structure of the catalyst was characterized using different techniques such as Fourier-transform infrared (FT-IR) spectroscopy, scanning electron microscopy (SEM), transmission electron microscopy (TEM), energy-dispersive x-ray spectroscopy (EDS), thermogravimetric analysis (TGA), and inductively coupled plasma (ICP) spectroscopy. All analyses confirmed successful covalent immobilization of CuPPh on GO. The activity of the catalyst was then studied for synthesis of 1,4-disubstituted 1,2,3-triazoles derivatives by reaction of various aryl azides and different terminal alkynes in mild reaction condition with good to excellent yield.
Graphical abstract










Similar content being viewed by others
References
S. Muratsugu, H. Baba, T. Tanimoto, K. Sawaguchi, S. Ikemoto, M. Tasaki, Y. Terao, M. Tada, Chem. Commun. 54, 40 (2018)
M.-G. Fan, Z.-C. Luo, G. Huang, F. Xiang, Y.-A. Guo, H. Zhou, J. Exp. Nanosci. 8, 4 (2013)
Y. Zhu, S. Murali, W. Cai, X. Li, J.W. Suk, J.R. Potts, R.S. Ruoff, Adv. Mater. 22, 35 (2010)
S. Eigler, S. Grimm, F. Hof, A. Hirsch, J. Mater. Chem. 1, 38 (2013)
J. Pyun, Angew. Chem. Int. 1, 50 (2010)
R. Huisgen, Angew. Chem. Int. 2, 10 (1963)
C.W. Tornøe, C. Christensen, M. Meldal, J. Org. Chem. 67, 9 (2002)
V.V. Rostovtsev, L.G. Green, V.V. Fokin, K.B. Sharpless, Angew. Chem. Int. 114, 14 (2002)
M. Meldal, C.W. Tornøe, Chem. Rev. 108, 8 (2008)
W. Zhang, X. He, B. Ren, Y. Jiang, Z. Hu, Tetrahedron Lett. 56, 19 (2015)
A.A. Ali, M. Chetia, D. Sarma, Tetrahedron Lett. 57, 15 (2016)
Y.-J. Song, C.-Y. Yoo, J.-T. Hong, S.-J. Kim, S.-U. Son, H.-Y. Jang, Bull. Korean Chem. Soc. 29, 8 (2008)
R.P. Jumde, C. Evangelisti, A. Mandoli, N. Scotti, R. Psaro, J. Catal. 324, 25 (2015)
L. Mohammadi, M.A. Zolfigol, A. Khazaei, M. Yarie, S. Ansari, S. Azizian, M. Khosravi, Appl. Organomet. Chem. 32, 1 (2018)
M.K. Barman, A.K. Sinha, S. Nembenna, Green Chem. 18, 8 (2016)
H. Sharghi, R. Khalifeh, M.M. Doroodmand, Adv. Synth. Catal. 351, 1 (2009)
M. Keyhaniyan, A. Shiri, H. Eshghi, A. Khojastehnezhad, New J. Chem. 42, 24 (2018)
M.B.M. Krishna, N. Venkatramaiah, R. Venkatesan, D.N. Rao, J. Mater. Chem. 22, 7 (2012)
B.C. Bookser, T.C. Bruice, J. Am. Chem. Soc. 113, 11 (1991)
R. Fareghi-Alamdari, M. Golestanzadeh, O. Bagheri, RSC Adv. 6, 110 (2016)
L. Jiang, L. Cui, X. He, J. Solid State Electrochem. 19, 2 (2015)
R. Zeng, G. Chen, C. Xiong, G. Li, Y. Zheng, J. Chen, Y. Long, S. Chen, Appl. Surf. Sci. 434, 756 (2018)
M. Chen, Z. Zhang, L. Li, Y. Liu, W. Wang, J. Gao, RSC Adv. 4, 58 (2014)
H.B. El Ayouchia, L. Bahsis, H. Anane, L.R. Domingo, S.-E. Stiriba, RSC Adv. 8, 14 (2018)
L. Liang, D. Astruc, Coord. Chem. Rev. 255, 23–24 (2011)
B.E. Velasco, G. López-Téllez, N. González-Rivas, I. García-Orozco, E. Cuevas-Yañez, Can. J. Chem. 91, 4 (2012)
M. Chetia, P.S. Gehlot, A. Kumar, D. Sarma, Tetrahedron Lett. 59, 4 (2018)
M. Chetia, A.A. Ali, D. Bhuyan, L. Saikia, D. Sarma, New J. Chem. 39, 8 (2015)
N. Boechat, V.F. Ferreira, S.B. Ferreira, M.L.G. Ferreira, F.C. da Silva, M.M. Bastos, M.S. Costa, M.C.S. Lourenço, A.C. Pinto, A.U. Krettli, J. Med. Chem. 54, 17 (2011)
A.A. Ali, M. Chetia, P.J. Saikia, D. Sarma, RSC Adv. 4, 110 (2014)
N. Almirante, S. Cicardi, C. Napoletano, M. Serravalle, Tetrahedron 43, 3 (1987)
K. Namitharan, M. Kumarraja, K. Pitchumani, Chem. Eur. J. 15, 12 (2009)
A.A. Ali, M. Chetia, B. Saikia, P.J. Saikia, D. Sarma, Tetrahedron Lett. 56, 43 (2015)
D. Wang, N. Li, M. Zhao, W. Shi, C. Ma, B. Chen, Green Chem. 12, 12 (2010)
P. Veerakumar, M. Velayudham, K.-L. Lu, S. Rajagopal, Catal. Sci. Technol. 1, 8 (2011)
Acknowledgements
The authors are grateful to the Iran National Science Foundation and Ferdowsi University of Mashhad for financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Khojastehnezhad, A., Bakavoli, M., Javid, A. et al. Synthesis, characterization, and investigation of catalytic activity of copper(II) porphyrin graphene oxide for azide–alkyne cycloaddition. Res Chem Intermed 45, 4473–4485 (2019). https://doi.org/10.1007/s11164-019-03843-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-019-03843-y