Abstract
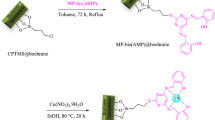
Here we report the synthesis of Ni(II) immobilized on modified boehmite nanostructures as an environmentally friendly heterogeneous catalyst for the one-pot domino synthesis of benzo[a]pyrano[2,3-c]phenazine and benzo[a]benzo[6,7]chromeno[2,3-c]phenazine in PEG as a green solvent. Elemental analysis, FT-IR, XRD, TGA, SEM, EDX, and ICP-OEC confirmed the structure of this nanocatalyst. Importantly, this nanocatalyst can be easily separated from the reaction media and reused for at least four consecutive cycles without a discernible reduction in its catalytic activity.












Similar content being viewed by others
References
D. Jariwala, V.K. Sangwan, L.J. Lauhon, T.J. Marks, M.C. Hersam, Chem. Soc. Rev. 42, 2824 (2013)
E.E. Perl, W.E. McMahon, J.E. Bowers, D.J. Friedman, Opt. Express 22, A1243 (2014)
C. Guo, J. Zhong, L. Gao, J. Gao, S. Huang, J. Saudi Chem. Soc. 16, 307 (2017)
L.S. Zhang, L.Y. Jiang, C.Q. Chen, W. Li, W.G. Song, Y.G. Guo, Chem. Mater. 22, 414 (2009)
M.A. Zolfigol, A.R. Moosavi-Zare, P. Moosavi, V. Khakyzadeh, A. Zare, C. R. Chim. 6, 962 (2013)
Y. Wang, F. Wang, H. Wang, M. Song, Sci. Rep. 7, 16510 (2017)
A. Ghorbani-Choghamarani, Z. Taherinia, NJC 41, 9414 (2017)
S. Narayanan, A. Sultana, K. Krishna, P. Mériaudeau, C. Naccache, Catal. Lett. 34, 129 (1995)
S. Li, M. Meng Lin, M.S. Toprak, D.K. Kim, M. Muhammed, Nano Rev. 1, 5214 (2010)
B. Tahmasbi, A. Ghorbani-Choghamarani, Catal. Lett. 147, 649 (2017)
V. Vatanpour, S.S. Madaeni, L. Rajabi, S. Zinadini, A.A. Derakhshan, J. Membr. Sci. 401, 132 (2017)
M. Abdollahifar, M. Hidaryan, P. Jafari, Bol. Soc. Esp. Ceram. V. 57, 66 (2018)
R.W.N. Denigres Filho, G.D.A. Rocha, C.R. Montes, A.C. Vieira-Coelho, Mater Res. 19, 659 (2016)
M.K. Naskar, M. Chatterjee, D. Ganguly, Bull. Mater. Sci. 25, 115 (2002)
S. Brühne, S. Gottlieb, W. Assmus, E. Alig, M.U. Schmidt, Cryst. Growth Des. 8, 489 (2008)
B.Y. Yeom, E. Shim, B. Pourdeyhimi, Macromol. Res. 18, 884 (2010)
Y. Mathieu, B. Lebeau, V. Valtchev, Langmuir 23, 9435 (2007)
H.W. Hou, Y. Xie, Q.N. Yang, Q.X. Guo, C.R. Tan, Nanotechnology 16, 741 (2005)
B. Tang, J.C. Ge, L.H. Zhuo, G.L. Wang, J.Y. Niu, Z.Q. Shi, Y.B. Dong, Eur. J. Inorg. Chem. 21, 4366 (2005)
Z.Q. Yu, C.X. Wang, T. Gu, C. Li, J. Lumin. 106, 153 (2002)
X.Y. Chen, S.W. Lee, Chem. Phys. Lett. 438, 279 (2007)
Y. Li, J. Liu, Z. Jia, Mater. Lett. 60, 3586 (2006)
W. Cai, J. Yu, S. Gu, M. Jaroniec, Cryst. Growth Des. 10, 3977 (2010)
M. Abdollahifar, R.M. Zamani, E. Beiygie, H. Nekouei, J. Serb. Chem. Soc. 79, 1007 (2014)
Y. Feng, W. Lu, L. Zhang, X. Bao, B. Yue, Y. Lv, X. Shang, Cryst. Growth Des. 8, 1426 (2008)
L. Zhang, Y.J. Zhu, J. Phys. Chem. C. 112, 16764 (2008)
Y. Zhu, H. Hou, G. Tang, Q. Hu, Eur. J. Inorg. Chem. 6, 872 (2010)
A. Váradi, T.C. Palmer, R. Notis Dardashti, S. Majumdar, Molecules 21, 19 (2015)
R.C. Cioc, E. Ruijter, R.V. Orru, Green Chem. 16, 2958 (2014)
B.B. Toure, D.G. Hall, Chem. Rev. 109, 4439 (2009)
C. Hulme, V. Gore, Curr. Med. Chem. 10, 51 (2003)
M. Haji, Beilstein J. Org. Chem. 12, 1269 (2016)
P.W. Crawford, R.G. Scamehorn, U. Hollstein, M.D. Ryan, P. Kovacic, Chem. Biol. Interact. 60, 67 (1986)
C. Neves-Pinto, V.R. Malta, M.D.C.F. Pinto, R.H. Santos, S.L. de Castro, A.V. Pinto, J. Med. Chem. 45, 2112 (2002)
S. Sun, L. Zhou, K. Jin, H. Jiang, Y.W. He, Sci. Rep. 6, 30352 (2016)
S.K. Ku, H. Yoo, W. Zhou, M. Na, J.S. Bae, Anim. Cells Syst. 18, 204 (2014)
A. Ghorbani-Choghamarani, G. Azadi, RSC Adv. 5, 9752 (2015)
T. Tamoradi, M. Ghadermazi, A. Ghorbani-Choghamarani, Catal. Lett. 148, 857 (2018)
A. Ghorbani-Choghamarani, P. Moradi, B. Tahmasbi, RSC Adv. 6, 56638 (2016)
J. Geng, D.A. Jefferson, B.F. Johnson, ChemComm 9, 969 (2007)
S.L. Wang, F.Y. Wu, C. Cheng, G. Zhang, Y.P. Liu, B. Jiang, S.J. Tu, ACS. Comb. Sci. 13, 135 (2011)
S. Abbasi Pour, A. Yazdani-Elah-Abadi, M. Afradi, Appl. Organomet. Chem. 31, e3791 (2017)
H.R. Shaterian, M. Mohammadnia, J. Mol. Liq. 177, 162 (2013)
J. Gao, M. Chen, X. Tong, H. Zhu, H. Yan, D. Liu, Y. Lu, Comb. Chem. High 18, 960 (2015)
S.A. Mirmiran-Yazdi, A.Y.E. Abadi, N. Shams, R. Mohebat, Turk. J. Chem. 41, 567 (2017)
A. Hasaninejad, S. Firoozi, Mol. Divers 17, 499 (2013)
A. Yazdani-Elah-Abadi, R. Mohebat, M. Kangani, J. Chem. Res. 40, 722 (2016)
Acknowledgements
The authors thank the research facilities of Ilam University, Ilam, Iran, for financial support of this research project.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ghorbani-Choghamarani, A., Sahraei, R. & Taherinia, Z. Ni(II) immobilized on modified boehmite nanostructures: a novel, inexpensive, and highly efficient heterogeneous nanocatalyst for multicomponent domino reactions. Res Chem Intermed 45, 3199–3214 (2019). https://doi.org/10.1007/s11164-019-03787-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-019-03787-3