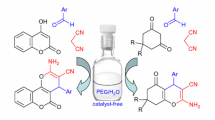
Abstract
This study presents a three-component reaction between malononitrile dimer, α-diketone and aliphatic amine that leads to the formation of previously undescribed alkylammonium 4-cyano-5-(dicyanomethylene)-2-hydroxy-2,3-dimethyl-2,5-dihydropyrrol-1-ides. This reaction is a novel approach for modification of donor–acceptor chromophores containing the buta-1,3-diene-1,1,3-tricarbonitrile moiety.










Similar content being viewed by others
References
M. Li, Y. Li, H. Zhang, S. Wang, Y. Ao, Z. Cui, J. Mater. Chem. C 5, 4111 (2017)
F. Bureš, RSC Adv. 4, 58826 (2014)
V. Malytskyi, J.-J. Simon, L. Patrone, J.-M. Raimundo, RSC Adv. 5, 354 (2015)
F. Terenziani, C. Katan, E. Badaeva, S. Tretiak, M. Blanchard-Desce, Adv. Mat. 20, 4641 (2008)
H. Li, P. Zhao, N. Zou, H. Wang, K. Sun, Tetrahedron Lett. 58, 30 (2017)
T. Chen, Y. Zheng, Z. Xu, M. Zhao, Y. Xu, J. Cui, Tetrahedron Lett. 54, 2980 (2013)
S.-H. Kim, S.-Y. Lee, S.-Y. Gwon, J.-S. Bae, Y.-A. Son, J. Photochem. Photobiol. A: Chem. 217, 224 (2011)
W. Lin, Y. Cui, J. Yu, Y. Yang, G. Qian, Dyes Pigm. 136, 791 (2017)
Y. Yang, J. Liu, H. Xiao, Z. Zhen, S. Bo, Dyes Pigm. 139, 239 (2017)
M.J. Cho, J. Seo, H.S. Oh, H. Jee, W.J. Kim, K.H. Kim, M.H. Hoang, D.H. Choi, P.N. Prasad, Sol. Energy Mater. Sol. Cells 98, 71 (2012)
S.M. Sharipova, A.A. Kalinin, Chem. Heterocycl. Compd. 53, 36 (2017)
Y. Hao, C.J. Wood, C.A. Clark, J.A. Calladine, R. Horvath, M.W.D. Hanson-Heine, X.-Z. Sun, I.P. Clark, M. Towrie, M.W. George, X. Yang, L. Sunaf, E.A. Gibson, Dalton Trans. 45, 7708 (2016)
J. Liua, W. Gaob, I.V. Kitykc, X. Liua, Z. Zhena, Dyes Pigm. 122, 74 (2015)
H. Huang, J. Liu, Z. Zhen, L. Qiu, X. Liu, G. Lakshminarayana, S. Tkaczyk, I.V. Kityk, Mater. Lett. 75, 233 (2012)
M. Ipuy, Y.-Y. Liao, E. Jeanneau, P.L. Baldeck, Y. Bretonnière, C. Andraud, J. Mater. Chem. C. 4, 766 (2016)
G. Deng, H. Huang, P. Si, H. Xu, J. Liu, S. Bo, X. Liu, Z. Zhen, L. Qiu, Polymer 54, 6349 (2013)
V. Parthasarathy, F. Castet, R. Pandey, O. Mongin, P.K. Das, M. Blanchard-Desce, Dyes Pigm. 130, 70 (2016)
Yi. Liao, B.E. Eichinger, K.A. Firestone, M. Haller, J. Luo, W. Kaminsky, J.B. Benedict, P.J. Reid, A. K.-Y. Jen, L.R. Dalton, B.H. Robinson, J. Am. Chem. Soc., 127, 2758 (2015)
V. Parthasarathy, R. Pandey, M. Stolte, S. Ghosh, F. Castet, F. Wìrthner, P.K. Das, M. Blanchard-Desce, Chem. Eur. J. 21, 14211 (2015)
J. Liu, S. Bo, X. Liu, Z. Zhen, J. Incl. Phenom. Macrocycl. Chem. 68, 253 (2010)
MYu. Belikov, S.V. Fedoseev, O.V. Ershov, MYu. Ievlev, V.A. Tafeenko, Tetrahedron Lett. 57, 4101 (2016)
S.V. Fedoseev, MYu. Belikov, O.V. Ershov, I.N. Bardasov, V.A. Tafeenko, Russ. J. Org. Chem. 52, 1440 (2016)
J.W. Ducker, M.J. Gunter, Aust. J. Chem. 26, 1551 (1973)
G. E. J. Howard, US 3178448, (1961); C.A., 63, 713b (1965)
J.P. Maciejewski, H. Gao, J.M. Shreeve, Chem. Eur. J. 19, 2947 (2013)
M. Warzecha, M. Oszajca, K. Pilarczyk, K. Szaciłowski, Chem. Commun. 51, 3559 (2015)
E. Shao, D. Li, J. Li, G. Zhang, W. Zhang, Gao, Z. Z. Anorg. Allg. Chem. 642, 871 (2016)
G. Cui, F. Zhang, X. Zhou, H. Li, J. Wang, C. Wang, Chem. Eur. J. 21, 5632 (2015)
N. Pelletier, Patent US 20130305685A1 (2013)
V.A. Tafeenko, R. Peschar, O.V. Kaukova, H. Schenk, L.A. Aslanov, Acta Cryst C 60, o62 (2004)
V.A. Tafeenko, O.V. Kaukova, R. Peschar, A.V. Petrov, L.A. Aslanov, Acta Cryst C 59, m421 (2003)
V.A. Tafeenko, S.I. Gurskiy, M.F. Fazylbekov, A.N. Baranov, L.A. Aslanov, Acta Cryst C 66, m32 (2010)
O.V. Ershov, V.P. Sheverdov, O.E. Nasakin, V.A. Tafeenko, Russ. J. Org. Chem. 37, 1509 (2001)
V.P. Sheverdov, O.V. Ershov, R.N. Efimov, O.E. Nasakin, S.I. Firgang, V.A. Tafeenko, Rus. J. Gen. Chem. 74, 744 (2004)
Acknowledgements
This work was supported by Russian Foundation for Basic Research, Grant No. 16-33-60156 mol_a_dc. The XRD study was carried out using the equipment purchased from the funds of the Program of development of Moscow University and within the framework of the Agreement on collaboration between the Chemical Department of the Lomonsov Moscow State University and the Chemical-Pharmaceutical Department of the I.N. Ulyanov Chuvash State University.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Fedoseev, S.V., Belikov, M.Y., Ievlev, M.Y. et al. Three-component synthesis of alkylammonium 4-cyano-5-(dicyanomethylene)-2-hydroxy-2,5-dihydropyrrol-1-ides. Res Chem Intermed 44, 3565–3579 (2018). https://doi.org/10.1007/s11164-018-3325-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-018-3325-0