Abstract
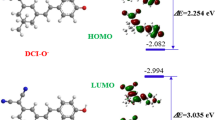
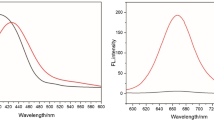
A new near-infrared fluorescent “turn-on” chemodosimeter (probe 1) based on dicyanomethylene-4H-chromene fluorophore for sulfide was developed. Probe 1 showed good fluorescence selectivity to sulfide over other anions and thiol-containing compounds. The discrimination of sulfide from disulfide could be achieved by comparison of the spectral difference of probe 1 after interaction with them. The absorption of probe 1 at 437 nm decreased, accompanied by red-shifting to 470 nm as a shoulder, and a strong peak at 610 nm appeared in the presence of sulfide. The absorption of 1 was slightly red-shifted to 452 nm with increased absorbance in the presence of disulfide. Probe 1 showed more than 1000-fold fluorescence enhancement at 712 nm with a Stokes shift of 102 nm in the presence of sulfide. The probe exhibited a good sensitivity in detection of sulfide with a wide pH span (4.0–11.0). The absorption change and fluorescence enhancement could be attributed to thiolysis of the dinitrophenyl ether group in probe 1 by the sulfide. The proposed mechanism was proved by 1H NMR spectra and EI-MS explorations. The probe was successfully applied for the detection of sulfide anions in real water samples.









Similar content being viewed by others
References
C. Kar, M.D. Adhikari, A. Ramesh, G. Das, Inorg. Chem. 52, 743 (2013)
C. Gao, X. Liu, X. Jin, J. Wu, Y. Xie, W. Liu, X. Yao, Y. Tang, Sens. Actuators B Chem. 185, 125 (2013)
X.W. Cao, W.Y. Lin, L.W. He, Org. Lett. 13, 4716 (2011)
F. Hou, J. Cheng, P. Xi, F. Chen, L. Huang, G. Xie, Y. Shi, H. Liu, D. Bai, Z. Zeng, Inorg. Chem. 51, 2454 (2012)
M.W. Warenycia, L.R. Goodwin, C.G. Benishin, R.J. Reiffenstein, D.M. Francom, J.D. Taylor, F.R. Dieken, Biochem. Pharmacol. 38, 973 (1989)
T.V. Mishanina, M. Libiad, R. Banerjee, Nat. Chem. Biol. 11, 457 (2015)
A. Papapetropoulos, A. Pyriochou, Z. Altaany, G. Yang, A. Marazioti, Z. Zhou, M.G. Jeschke, L.K. Branski, D.N. Herndon, R. Wang, C. Szabò, Proc. Natl. Acad. Sci. USA 106, 21972 (2009)
G. Yang, L. Wu, W. Yang, J. Qi, B. Jiang, K. Cao, Q. Meng, A.K. Mustafa, W. Mu, S.H. Snyder, R. Wang, Science 322, 587 (2008)
G. Yang, W. Yang, L. Wu, R. Wang, J. Biol. Chem. 282, 16567 (2007)
P. Kamoun, M.C. Belardinelli, A. Chabli, K. Lallouchi, B. Chadefaux-Vekemans, Am. J. Med. Genet. A 116, 310 (2003)
B. Vallejo, P. Richter, I. Toral, C. Tapia, M.D. Luque de Castro, Anal. Chim. Acta 436, 301 (2001)
M.G. Choi, S. Cha, H. Lee, H.L. Jeon, S.K. Chang, Chem. Commun. 45, 7390 (2009)
J.C. Savage, D.H. Gould, J. Chromatogr. 526, 540 (1990)
J. Radford-Knoery, G.A. Cutter, Anal. Chem. 65, 976 (1993)
R. Kaushik, A. Ghosh, D.A. Jose, Coord. Chem. Rev. 347, 141 (2017)
J. Zhang, Y. Gao, X. Kang, Z. Zhu, Z. Wang, Z. Xi, L. Yi, Org. Biomol. Chem. 15, 4212 (2017)
L. Yi, Z. Xi, Org. Biomol. Chem. 15, 3828 (2017)
K. Zhang, J. Zhang, Z. Xi, L.-Y. Li, X. Gu, Q.-Z. Zhang, L. Yi, Chem. Sci. 8, 2776 (2017)
L. Chen, D. Wu, C.S. Lim, D. Kim, S.-J. Nam, W. Lee, G. Kim, H.M. Kim, J. Yoon, Chem. Commun. 53, 4791 (2017)
A. Bamesberger, G. Kim, J. Woo, H. Cao, J. Fluoresc. 25, 25 (2015)
C. Liu, B. Peng, S. Li, C.-M. Park, A.R. Whorton, M. Xian, Org. Lett. 14, 2184 (2012)
J. Zhang, J. Zhou, X. Dong, X. Zheng, W. Zhao, RSC Adv. 6, 51304 (2016)
C. Liu, J. Pan, S. Li, Y. Zhao, L.Y. Wu, C.E. Berkman, A.R. Whorton, M. Xian, Angew. Chem. Int. Ed. 50, 10327 (2011)
Z. Xu, L. Xu, J. Zhou, Y. Xu, W. Zhu, X. Qian, Chem. Commun. 48, 10871 (2012)
F. Huo, Y. Zhang, P. Ning, X. Meng, C. Yin, J. Mater. Chem. B 5, 2798 (2017)
T. Liu, Z. Xu, D.R. Spring, J. Cui, Org. Lett. 15, 2310 (2013)
Y. Liu, G. Feng, Org. Biomol. Chem. 12, 438 (2014)
L. Zhou, D. Lu, Q. Wang, S. Liu, Q. Lin, H. Sun, Biosens. Bioelectron. 91, 699 (2017)
D. Maity, A. Raj, P.K. Samanta, D. Karthigeyan, T.K. Kundu, S.K. Pati, T. Govindaraju, RSC Adv. 4, 11147 (2014)
S. Chen, C. Ma, M.-S. Yuan, W. Wang, D.-E. Wang, S.-W. Chen, J. Wang, RSC Adv. 6, 85529 (2016)
H. Chen, W. Lin, H. Cui, W. Jiang, Chem. Eur. J. 21, 733 (2015)
S. El Sayed, C. de la Torre, L.E. Santos-Figueroa, R. Martínez-Máñez, F. Sancenón, M. Orzáez, A.M. Costero, M. Parra, S. Gil, Supramol. Chem. 27, 244 (2015)
J. Lv, F. Wang, J. Qiang, X. Ren, Y. Chen, Z. Zhang, Y. Wang, W. Zhang, X. Chen, Biosens. Bioelectron. 87, 96 (2017)
K.M. Vengaian, C.D. Britto, K. Sekar, G. Sivaraman, S. Singaravadivel, RSC Adv. 6, 7668 (2016)
Y. Wang, D. Qiu, M. Li, Y. Liu, H. Chen, H. Li, Spectrochimica Acta Part A 185, 256 (2017)
K.M. Vengaian, C.D. Britto, K. Sekar, G. Sivaraman, S. Singaravadivel, Sens. Actuators B Chem. 235, 232 (2016)
T.S. Bailey, L.N. Zakharov, M.D. Pluth, J. Am. Chem. Soc. 136, 10573 (2014)
Y. Kimura, Y. Mikami, K. Osumi, M. Tsugane, J. Oka, H. Kimura, FASEB J. 27, 2451 (2013)
Q. Han, Z. Mou, H. Wang, X. Tang, Z. Dong, L. Wang, X. Dong, W. Liu, Anal. Chem. 88, 7206 (2016)
C. Liu, W. Chen, W. Shi, B. Peng, Y. Zhao, H. Ma, M. Xian, J. Am. Chem. Soc. 136, 7257 (2014)
K. Li, F.-Z. Chen, Q.-H. Yin, S. Zhang, W. Shi, D.-M. Han, Sens. Actuators B: Chem. 254, 222 (2018)
H. Shang, H. Chen, Y. Tang, R. Guo, W. Lin, Sens. Actuators B: Chem. 230, 773 (2016)
L. Zeng, S. Chen, T. Xia, W. Hu, C. Li, Z. Liu, Anal. Chem. 87, 3004 (2015)
W. Chen, E.W. Rosser, D. Zhang, W. Shi, Y. Li, W.-J. Dong, H. Ma, D. Hu, M. Ming, Org. Lett. 17, 2776 (2015)
W. Chen, E.W. Rosser, T. Matsunaga, A. Pacheco, T. Akaike, M. Xian, Angew. Chem. Int. Ed. 54, 13961 (2015)
X. Gong, X.-F. Yang, Y. Zhong, H. Chen, Z. Li, RSC Adv. 6, 88519 (2016)
Z. Liu, X. Wang, Z. Yang, W. He, J. Org. Chem. 76, 10286 (2011)
Z. Guo, W. Zhu, H. Tian, Chem. Commun. 48, 6073 (2012)
W. Zhu, X. Huang, Z. Guo, X. Wu, H. Yu, H. Tian, Chem. Commun. 48, 1784 (2012)
J. Cao, C.C. Zhao, W.H. Zhu, Tetrahedron Lett. 53, 2107 (2012)
Y. Zheng, M. Zhao, Q. Qiao, H. Liu, H. Lang, Z. Xu, Dyes Pigments 98, 367 (2013)
M. Li, X. Wu, Y. Wang, Y. Li, W. Zhu, T.D. James, Chem. Commun. 50, 1751 (2014)
P. Xie, F. Guo, G. Gao, W. Fan, G. Yang, L. Xie, J. Fluoresc. 26, 1737 (2016)
W. Sun, J. Fan, C. Hu, J. Cao, H. Zhang, X. Xiong, J. Wang, S. Cui, S. Sun, X. Peng, Chem. Commun. 49, 3890 (2013)
H. Filik, M. Hayvali, E. Kilic, Anal. Chim. Acta 535, 177 (2005)
Z.F. Li, Y. Xiang, A.J. Tong, Anal. Chim. Acta 619, 75 (2008)
S.Y. Lee, C. Kim, RSC Adv. 6, 85091 (2016)
L. Tang, Z. Zheng, K. Zhong, Y. Bian, Tetrahedron Lett. 57, 1361 (2016)
Acknowledgements
The authors thank the National Natural Science Foundation of China (No. 21102037) and Henan Agricultural University (Nos. KF1609, KF1720 and 201710466) for the financial support.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Xie, P., Zhu, Y., Huang, X. et al. A new near-infrared fluorescent chemodosimeter for discrimination of sulfide from disulfide. Res Chem Intermed 44, 2823–2837 (2018). https://doi.org/10.1007/s11164-018-3263-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-018-3263-x