Abstract
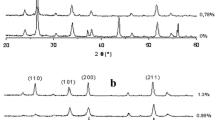
The effect of Er3+ doping (1%) on the structural, optical and photocatalytic properties of In2O3 thin films deposited on quartz substrates by spin coating was investigated. The In2O3:1% Er3+ films, annealed in the temperature range 800–1000 °C, were characterized by X-ray diffraction, scanning electron microscopy (SEM), atomic force microscopy, UV–Vis spectroscopy, ellipsometry and photoluminescence (PL). The films are polycrystalline with a cubic structure and the lattice parameter increases with the incorporation of Er3+ owing to its larger radius. The SEM images of the film show a granular morphology with large grains (~ 200 nm). The doped In2O3 film exhibits less transparency than In2O3 in the UV–visible region with band gaps of 3.42 and 3.60 eV, respectively. PL shows strong lines at 548 and 567 nm, assigned to Er3+ under direct excitation at 532 nm. The energy diagram of the junction In2O3:1% Er3+/Na2SO4 (0.1 M) solution plotted from physical and photoelectrochemical characterizations shows the feasibility of the films for Rhodamine B (RhB) degradation under solar light. The conduction band at 2.22 V deriving from the In3+:5s orbital is suitably positioned with respect to the O2/O ·2 level (~ 1.40 VSCE), leading to oxidation of 32% of 10 ppm RhB within 40 min of solar irradiation.









Similar content being viewed by others
References
L.N. Lau, N.B. Ibrahim, H. Baqiah, Appl. Surf. Sci. 345, 355–362 (2015)
P. King, T.D. Veal, F. Fuchs, C.Y. Wang, D. Payne, A. Bourlange, H. Zhang, G.R. Bell, V. Cimalla, O. Ambacher, Phys. Rev. B 79, 205–211 (2009)
M.A. Majeed Khan, W. Khan, M. Ahamed, M. Alhoshan, Mater. Lett. 79, 119–121 (2012)
Y.H. Shin, S.B. Kang, S. Lee, J.J. Kim, H.K. Kim, Org. Electron. 14, 926–935 (2013)
C. Cantalini, W. Wlodarski, H.T. Sun, M.Z. Atashbar, M. Passacantando, A.R. Phani, S. Santucci, Thin Solid Films 350, 276–282 (1999)
D.W. Sheel, J.M. Gaskell, Thin Solid Films 520, 1242–1245 (2011)
A. Patra, C.S. Friend, R. Kapoor, P.N. Prasad, J. Phys. Chem. B 106, 1909–1912 (2002)
P.T. Neuvonen, K. Sigvardt, S.R. Johannsen, J. Chevallier, B. Julsgaard, S.K. Ram, A.N. Larsen, Appl. Phys. Lett. 104, 102–106 (2014)
S. Yerci, R. Li, S.O. Kucheyev, T.V. Buuren, S.N. Basu, L. Dal Negro, J. Appl. Phys. Lett. 95, 031107 (2009)
T.O.L. Sunde, Ph.D. Thesis (SINTEF) Trondheim, Norway (2013)
N.V. Gaponenko, A.V. Mudryi, O.V. Sergeev, V.E. Borisenko, E.A. Stepanova, A.S. Baran, A.I. Ratko, J.C. Pivin, J.F. McGilp, Phys Stat. Sol. A. 165, 131 (1998)
A. Podhorodecki, R. Kudrawiec, J. Misiewicz, N.V. Gaponenko, D.A.B. Tsyrkunov, Opt. Mater. 28, 685–687 (2006)
H.K. Kim, C.C. Li, G. Nykolak, P.C. Becker, J. Appl. Phys. 76(12), 8209 (1994)
S. Boumaza, F. Kaouah, T. Berrama, M. Trari, Z. Bendjama, J. Clean. Prod. 54, 296–306 (2013)
R. Outemzabet, M. Doulache, M. Trari, Appl. Phys. A 119, 589–596 (2015)
Y.G. Choi, S.M. Yu, W.J. Chung, Chem. Phys. Lett. 461, 290–293 (2008)
A. Ghosh, A. Mondal, A. Das, S. Chattopadhyay, K.K. Chattopadhyay, J. Alloys Compd. 695, 1260–1265 (2017)
M.J. Buerger, X-ray Crystallography (Wiley, New York, 1960), p. 23
Q. Xiao, Y. Liu, L. Liu, R. Li, W. Luo, X. Chen, J. Phys. Chem. 114, 9314–9321 (2010)
S. Bucak, D. Rende, Colloid and Surface Chemistry: A Laboratory Guide for Exploration of the Nanoworld (CRC Press, New York, 2014)
Z. Yuan, X. Zhu, X. Wang, Thin Solid Films 519, 3254–3258 (2011)
TSh Atabaev, M. Kurisu, K. Konishi, N.H. Hong, Am. J. Nanosci. Nanotechnol. 2(1), 13–16 (2014)
Z. Pan, A. Ueda, S.K. Hark, S. Mu, J. Nanophotonics 6, 063508 (2012)
B. Pipeleers, S.M. Hogg, A. Vantomme, Nucl. Instrum. Methods Phys. Res. B 206, 95–98 (2003)
L.N. Lau, Appl. Surf. Sci. 345, 355–359 (2015)
W. Chen, J.O. Bovin, A.G. Joly, S.P. Wang, F.H. Su, G.H. Li, J. Phys. Chem. B 108, 11927–11934 (2004)
S.Y. Bae, C.W. Na, J.H. Kang, J. Park, J. Phys. Chem. B 109, 2526 (2005)
S.J. Wen, G. Couturier, G. Campet, J. Portier, J. Claverie, Phys. Stat. Sol. 130, 407 (1992)
G. Rekhila, Y. Bessekhouad, M. Trari, Int. J. Hydrog. Energy. 38, 6335–6343 (2013)
V.M. Aroutiounian, V.M. Arakelyan, G.E. Shahnazaryan, M.G. Stepanyan, E.A. Khachaturyan, J.A. Turner, C. R. Chim. 9, 325–333 (2006)
V.M. Aroutiounian, V.M. Arakelyan, G.E. Shakh Nazaryan, G.M. Stephanyan, J.A. Turner, O. Khaselev, Russ. J. Electrochem. Soc. 38, 378–383 (2002)
K. Dib, R. Brahimi, Y. Bessekhouad, N. Tayebi, M. Trari, Mater. Sci. Semicond. Process. 48, 52–59 (2016)
H. Moualkia, G. Rekhila, M. Izerrouken, A. Mahdjoub, M. Trari, Mater. Sci. Semicond. Process. 21, 186–193 (2014)
Acknowledgements
This work was financially supported by the Faculties of Physics and Chemistry (Algiers). Special thanks to Prof. Raşit Turan and Dr Selcuk Yerci in The Center for Solar Energy Research and Applications for their helpful discussions and technical assistance in this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Keriti, Y., Keffous, A., Dib, K. et al. Photoluminescence and photocatalytic properties of Er3+-doped In2O3 thin films prepared by sol–gel: application to Rhodamine B degradation under solar light. Res Chem Intermed 44, 1537–1550 (2018). https://doi.org/10.1007/s11164-017-3183-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-017-3183-1