Abstract
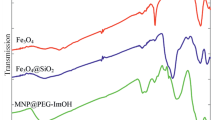
An effective synthesis of anion-exchanged supported ionic liquid using magnetically separable nanoparticles and its catalytic effect on N-alkylation reactions is described. Anionic polyoxometalate derivative was used in the anion-exchange step in catalyst design. The catalytic system can be easily separated from the reaction mixture with external magnetic field and recycled in subsequent reactions. In order to evaluate catalyst repeatability, N-alkylation of some more amines such as Aniline, 4-aminobenzenesulfonamide, 4-methoxyaniline, 2-aminopyrimidin and 4,5,6,7-tetrahydrobenzo[d]thiazole-2,6-diamine in the presence of recoverable catalyst was successfully examined in this article. In addition, pramipexole dihydrochloride as an active pharmaceutical ingredient was successfully synthesized using the catalytic system. The structure of catalyst was determined by infrared spectroscopy, X-ray powder diffraction, and scanning electron microscope techniques. The structure of organic products was determined by 1H NMR, 13C NMR, infrared and Mass spectroscopy.
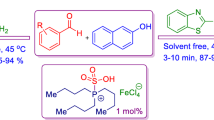
Graphical Abstract






Similar content being viewed by others
References
M. Wang, P. Wang, Q. Tian, J. Liu, J. Deng, N. Li, J. Zhou, Res. Chem. Intermed. 41, 8891 (2015)
S. Hegde, S.S. Joshi, T. Mukherjee, S. Kapoor, Res. Chem. Intermed. 40, 1125 (2014)
G. Hu, H. Miao, H. Mei, S. Zhou, Y. Xu, Dalton Trans. 45, 7947 (2016)
W. Zhao, C. Yang, New J. Chem. 37, 1867 (2013)
Y. Ding, W. Zhao, W. Song, Z. Zhang, B. Ma, Green Chem. 13, 1486 (2011)
D.L. Long, R. Tsunashima, L. Cronin, Angew. Chem. Int. Ed. 49, 1736 (2010)
Q. Huang, Q. Song, J. Cai, X. Zhang, S. Lin, Adv. Synth. Catal. 355, 1512 (2013)
I.V. Kozhevnikov, Chem. Rev. 98, 171 (1998)
S. Damyanova, J.L.G. Fierro, I. Sobrados, J. Sanz, Langmuir 15, 469 (1999)
H.R. Shaterian, F. Moradi, Res. Chem. Intermed. 41, 223 (2015)
S. Roy, K.K. Senapati, P. Phukan, Res. Chem. Intermed. 41, 5753 (2015)
Z. Li, K. Gao, G. Han, R. Wang, H. Li, X.S. Zhao, P. Guo, New J. Chem. 39, 361 (2015)
C.W. Lim, I.S. Lee, Nano Today 5, 412 (2010)
M.H. Ghasemi, E. Kowsari, S.K. Hosseini, Tetrahedron Lett. 57, 387 (2016)
S. Asghari, M. Mohammadnia, Res. Chem. Intermed. 42, 1899 (2016)
A. Kong, P. Wang, H. Zhang, F. Yang, S.P. Huang, Y. Shan, Appl. Catal. A 417, 183 (2012)
P.B. Bhat, B.R. Bhat, New J. Chem. 39, 273 (2015)
H. Alinezhad, M. Tajbakhsh, N. Ghobadi, Res. Chem. Intermed. 41, 9113 (2015)
G.M. Ziarani, L. Seyedakbari, S. Asadi, A. Badiei, M. Yadavi, Res. Chem. Intermed. 42, 499 (2016)
P. Rai, M. Srivastava, J. Singh, J. Singh, New J. Chem. 38, 3181 (2014)
X. Zheng, S. Luo, L. Zhang, J.P. Cheng, Green Chem. 11, 455 (2009)
W. Zhu, P. Wu, L. Yang, Y. Chang, Y. Chao, H. Li, Y. Jiang, W. Jiang, S. Xun, Chem. Eng. J. 229, 250 (2013)
W. Ding, W. Zhu, J. Xiong, L. Yang, A. Wei, M. Zhang, H. Li, Chem. Eng. J. 266, 213 (2015)
W. Jiang, W. Zhu, H. Li, X. Wang, S. Yin, Y. Chang, H. Li, Fuel 140, 590 (2015)
M.H. Ghasemi, E. Kowsari, A. Shafiee, Tetrahedron Lett. 57, 1150 (2016)
H. Alinezhad, M. Tajbakhsh, N. Ghobadi, Res. Chem. Intermed. 41, 9979 (2015)
R. Kawahara, K. Niinomi, J.N. Kondo, M. Hibino, N. Mizuno, S. Uchida, Dalton Trans. 45, 2805 (2016)
Y. Qiao, Z. Hou, H. Li, Y. Hu, B. Feng, X. Wang, L. Hua, Q. Huang, Green Chem. 11, 1955 (2009)
H.G.O. Alvim, G.A. Bataglion, L.M. Ramos, A.L. de Oliveira, H.C.B. de Oliveira, M.N. Eberlin, J.L. de Macedo, W.A. da Silva, B.A.D. Neto, Tetrahedron 70, 3306 (2014)
Y. Ju, R.S. Varma, Green Chem. 6, 219 (2004)
R. Cano, D.J. Ramon, M. Yus, J. Org. Chem. 76, 5547 (2011)
M.B. Gawande, P.S. Branco, R.S. Varma, Chem. Soc. Rev. 42, 3371 (2013)
J.W. Kim, K. Yamaguchi, N. Mizuno, J. Catal. 263, 205 (2009)
J. He, K. Yamaguchi, N. Mizuno, Chem. Lett. 39, 1182 (2010)
C.H. Tang, L. He, Y.M. Liu, Y. Cao, H.Y. He, K.N. Fan, Chem. Eur. J. 17, 7172 (2011)
M. Zivec, B. Anzic, S. Gobec, Org. Process Res. Dev. 14, 1125 (2010)
D. Chen, W. Li, Y. Wu, Q. Zhu, Z. Lu, G. Du, Chem. Eng. J. 221, 8 (2013)
Y.H. Deng, C.C. Wang, J.H. Hu, W.L. Yang, S.K. Fu, Colloids Surf. A 262, 87 (2005)
A. Chrobok, S. Baj, W. Pudło, A. Jarzebski, Appl. Catal. A Gen. 366, 22 (2009)
L.T.A. Sofia, A. Krishnan, M. Sankar, N.K. Kala Raj, P. Manikandan, P.R. Rajamohanan, T.G. Ajithkumar, J. Phys. Chem. C 113, 21114 (2009)
I.V. Kozhevnikov, Catal. Rev. Sci. Eng. 37, 311 (1995)
Acknowledgments
The authors wish to gratefully thank the Research Affairs Division at Amir Kabir University of Technology (AUT), Tehran, for financial support.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ghasemi, M.H., Kowsari, E. Convenient N-Alkylation of amines using an effective magnetically separable supported ionic liquid containing an anionic polyoxometalate. Res Chem Intermed 43, 1957–1968 (2017). https://doi.org/10.1007/s11164-016-2741-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-016-2741-2