Abstract
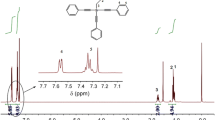
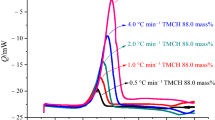
Methyl-tri(phenylethynyl)silane ((ph-C≡C)3-Si-CH3) (MTPES) was synthesized with methyltrichlorosilane and phenylethylene by Grignard reaction. Its molecular structure was characterized by Fourier transform infrared spectroscopy, nuclear magnetic resonance (1H-NMR, 13C-NMR, and 29Si-NMR). Its curing behavior was analyzed by non-isothermal differential scanning calorimetry and rheometry, and the corresponding kinetic parameters and kinetic model were also discussed by Kissinger, Ozawa, Flynn–Wall–Ozawa and Friedman methods. The results showed that the melting point of MTPES was 130 °C and the processing window was 200 °C. The activation energy E a, pre-exponential factor lnA and the reaction order n, m were 112.58 kJ/mol, 21.22 (s−1), 1.20 and 0.56, respectively. The curing behavior of MTPES followed the autocatalytic kinetic model.










Similar content being viewed by others
References
S.B. Sastri, J.P. Armistead, T.M. Keller, Cure kinetics of a multisubstituted acetylenic monomer. Polymer 36(7), 1449–1454 (1995)
M. Itoh, K. Inoue, K. Iwata et al., New highly heat-resistant polymers containing silicon: poly(silyleneethynylenephenyleneethynylene)s. Macromolecules 30(4), 694–701 (1997)
Z. Jiang, Y. Zhou, L. Du, Characterization of a modified silicon-containing arylacetylene resin with POSS functionality. Chin. J. Polym. Sci. 29(6), 726–731 (2011)
Y. Wu, R. Yu, L. Hu et al., Thermal stability of cocured blends of vinyl trimethoxysilane and aryl acetylene resins with different posttreatments. J. Appl. Polym. Sci. 131(8), 40158–40163 (2014)
S. Tannenbaum, S. Kaye, G.F. Lewenz, Synthesis and properties of some alkylsilanes. J. Am. Chem. Soc. 75(15), 3753–3757 (1953)
A.P. Melissaris, M.H. Litt, New high-Tg, heat-resistant, cross-linked polymers. 1. Synthesis and characterization of di-p-ethynyl-substituted benzyl phenyl ether monomers. Macromolecules 27(4), 883–887 (1994)
Z.L. Dai, Q. Chen, L.Z. Ni et al., Curing kinetics and structural changes of a of di[(N-m-acetenylphenyl) phthalimide] ether/[(methyl) diphenylacetylene]silane copolymer. J. Appl. Polym. Sci. 100, 2126–2130 (2006)
B. Wrackmeyer, E. Khan, S. Bayer et al., Alkynylsilanes and alkynyl (vinyl) silanes. Synthesis, molecular structures and multinuclear magnetic resonance study. Zeitschrift für Naturforschung B 65(6), 725–744 (2010)
I. Kownacki, B. Orwat, B. Marciniec et al., A new and efficient route for the synthesis of alkynyl functionalized silicon derivatives. Tetrahedron Lett. 55(2), 548–550 (2014)
C. Hamdouchi, H.M. Walborsky, Mechanism of Grignard Reagent Formation (Marcel Dekker, New York, 1996), pp. 145–218
Q. Chen, Y. Li, Z.L. Dai et al., Synthesis and characterization of methyl-di (phenylethynyl) silane and its network polymer. Acta. Chim. Sin. 63(3), 254–258 (2005)
A. Boudin, G. Cerveau, C. Chuit et al., Reactivity of anionic pentacoordinated silicon complexes towards nucleophiles. Angew. Chem. Int. Ed. 25(5), 473–474 (1986)
R. J. Corriu, G. E. Cerveau, C. G. Chuit, et al. Crosslinking agents; drugs. U. S. Pat. 4617413, 1986
C. Kim, K. Jeong, I. Jung, Progress toward limiting generation of dendritic ethynylsilanes (PhC≡C)4−nMenSi(n=0–2). J. Polym. Sci. Part A Polym. Chem. 38(15), 2749–2759 (2000)
Q. Zhou, X. Feng, L. Ni et al., Novel heat resistant methyl-tri(phenylethynyl) silane resin: synthesis, characterization and thermal properties. J. Appl. Polym. Sci. 102(3), 2488–2492 (2006)
Q. Zhou, X. Feng, H.Q. Zhao et al., Curing behavior and thermal property of methyl-tri(phenylethynyl)silaneresin. J. Funct. Polym. 20(1), 97–103 (2007)
Z. Luo, L. Wei, F. Liu et al., Study on thermal cure and heat-resistant properties of N-(3-acetylenephenyl) maleimide monomer. Eur. Polym. J. 43(8), 3461–3470 (2007)
Q. Zhou, L. Ni, Thermal cure behavior and pyrolysis of methyl-tri(phenylethynyl) silane resin. J. Appl. Polym. Sci. 113(1), 10–16 (2009)
D. Tan, T. Shi, Z. Li, Synthesis, characterization, and non-isothermal curing kinetics of two silicon-containing arylacetylenic monomers. Res. Chem. Intermed. 37(8), 831–845 (2011)
H.E. Kissinger, Reaction kinetics in differential thermal analysis. Anal. Chem. 29(11), 1702–1706 (1957)
T. Ozawa, Non-isothermal kinetics and generalized time. Thermochim. Acta 100(1), 109–118 (1986)
Y. Lu, M. Li, Y. Zhang et al., Synthesis and curing kinetics of benzoxazine containing fluorene and furan groups. Thermochim. Acta 515(1), 32–37 (2011)
N. Sbirrazzuoli, Y. Girault, L. Elégant, Simulations for evaluation of kinetic methods in differential scanning calorimetry. Part 3: peak maximum evolution methods and isoconversional methods. Thermochim. Acta 293(1), 25–37 (1997)
S.B. Sastri, T.M. Keller, M. Kenneth, Studies on cure chemistry of new acetylenic resins. Macromolecules 26(23), 6171–6174 (1993)
C. Jubsilp, K. Punson, T. Takeichi et al., Curing kinetics of benzoxazine-epoxy copolymer investigated by non-isothermal differential scanning calorimetry. Polym. Degrad. Stab. 95(6), 918–924 (2010)
H.L. Friedman, Kinetics of thermal degradation of char-forming plastics from thermogravimetry. J. Polym. Sci. Part C Polym. Symp. 6(1), 183–195 (1964)
L. Ke, D. Hu, Y. Lu et al., Copolymerization of maleimide-based benzoxazine with styrene and the curing kinetics of the resultant copolymer. Polym. Degrad. Stab. 97(2), 132–138 (2012)
A.J. Bissette, S.P. Fletcher, Mechanisms of autocatalysis. Angew. Chem. Int. Ed. 52(49), 12800–12826 (2013)
Acknowledgments
We gratefully acknowledged the financial support of the National Nature Science Foundation of China (Nos. 51477002, 51303005), the Educational Commission of Anhui Province of China (Nos. KJ2013A087 and KJ2013A095) and the Doctor Foundation of the Anhui University of Science and Technology.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tan, D., Wu, X., Wang, Y. et al. Synthesis, characterization and curing behavior of methyl-tri(phenylethynyl)silane. Res Chem Intermed 42, 4669–4681 (2016). https://doi.org/10.1007/s11164-015-2307-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-015-2307-8