Abstract
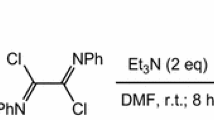
An efficient synthesis of N-mono-substituted β-amino tertiary thiols by reductive ring-opening of 3-thiazolines, obtained by use of the Asinger reaction, is described. This synthesis be regarded as a complementary approach for aminolytic ring-opening of thiiranes.



Similar content being viewed by others
Notes
Data for 3-thiazolines related to 3: 1H NMR (CDCl3): 1.56 (s, 6H, C (CH3)2), 1.67 (s, 6H, C(CH3)2), 6.90 (s, H, =CH); related to 5: 1H NMR (CDCl3): 1.57 (s, 6H, C(CH3)2), 5.50 (s, 1H, CH), 7.26 (s, 1H, =CH), 7.46–7.52 (m, 5H, Ph–H)
References
Q. Dong, X. Fang, J.D. Schroeder, D.S. Garvey, Practical, efficient synthesis of β-amino tertiary thiols via aminolysis ring-opening of thiiranes. Synthesis 3, 1106 (1999)
R. Luhowy, F. Menghini, An improved synthesis of aminoethanethiols. J. Org. Chem. 38, 2405 (1973)
I. Schlemminger, H.-H. Janknecht, W. Maison, W. Saak, Synthesis of the first enantiomerically pure 3-thiazolines via Asinger reaction. Tetrahedron Lett. 41, 7289–7292 (2000)
J. Martens, H. Offermanns, P. Scherberich, Facile synthesis of racemic cysteine. Angew. Chem. Int. Ed. Engl. 20, 668 (1981)
M. Hatam, D. Tehranfar, J. Martens, A novel and convenient route to phosphono-oligopeptides derived from 1,3-oxazolines, 1,3-oxazines and 1,3-thiazolines. Synth. Commun. 25, 1677–1688 (1995)
M. Ingolf, Pleuromutilins. US5164526. 11, 17 (1992)
Roy Beatrice, duMoulinet Amaury, Hardemare, Fontecave Marc, New Thionitrites: synthesis, Stability, and Nitric Oxide Generation. J. Org. Chem. 59, 7019–7026 (1994)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xin, S., Dengfeng, Z. & Jiangtao, S. Practical, efficient synthesis of N-mono-substituted β-amino tertiary thiols via reductive ring-opening of 3-thiazolines. Res Chem Intermed 39, 767–770 (2013). https://doi.org/10.1007/s11164-012-0705-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-012-0705-8