Abstract
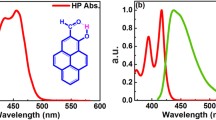
(E)-1-[2-Hydroxy-4-(phenylethynyl)phenyl]-3-[4-(phenylethynyl)phenyl]prop-2-en-1-one (1), (E)-1-[2-hydroxy-4-(phenylethynyl)phenyl]-3-phenylprop-2-en-1-one (2), and (E)-1-(2-hydroxyphenyl)-3-[4-(phenylethynyl)phenyl]prop-2-en-1-one (3), which belong to a new class of 2′-hydroxychalcones with phenylethynyl group(s) at the para position of the phenyl ring, were synthesized, and their photochemical properties were investigated. The lowest energy absorption band of 1 peaks at a longer wavelength (383 nm) with a much larger molar extinction coefficient (5.0 × 104 M −1 cm−1) than that of the parent 2′-hydroxychalcone (2′HC) (2.0 × 104 M −1 cm−1 at 318 nm). Upon photoexcitation, all three compounds underwent excited-state intramolecular hydrogen atom transfer (ESIHT) to produce an excited tautomer that emitted fluorescence with a large Stokes shift in the longer wavelength region at 600–700 nm. The quantum yield of the tautomer fluorescence of 1 was not high at 298 K (Φ f = 9.1 × 10−5), but was highest among 2′HC and its analogues. The Φ f values of 1–3 increased 10–30 fold upon reducing the temperature from 298 to 77 K.






Similar content being viewed by others
References
R.S. Becker, W.F. Richey, J. Am. Chem. Soc. 89, 1298 (1967)
M. Kasha, J. Chem. Soc., Faraday Trans. 2 82, 2379 (1986)
P.F. Barbara, P.K. Walsh, L.E. Brus, J. Phys. Chem. 93, 29 (1989)
L.G. Arnaut, S.J. Formosinho, J. Photochem. Photobiol. A 75, 1 (1993)
S.J. Formosinho, L.G. Arnaut, J. Photochem. Photobiol. A 75, 21 (1993)
M. Kasha, J. Heldt, D. Gormin, J. Phys. Chem. 99, 7281 (1995)
J. Catalán, J. Palomar, J.L.G. de Paz, J. Phys. Chem. A 101, 7914 (1997)
W.-S. Yu, C.-C. Cheng, Y.-M. Cheng, P.-C. Wu, Y.-H. Song, Y. Chi, P.-T. Chou, J. Am. Chem. Soc. 125, 10800 (2003)
A. Roque, C. Lodeiro, F. Pina, M. Maestri, R. Ballardini, V. Balzani, Eur. J. Org. Chem. 16, 2699 (2002)
A. Ohshima, A. Momotake, T. Arai, J. Photochem. Photobiol. A 162, 473 (2004)
A. Ohshima, A. Momotake, T. Arai, Chem. Lett. 34, 1288 (2005)
A. Ohshima, A. Momotake, T. Arai, Bull. Chem. Soc. Jpn. 79, 305 (2006)
A. Ohshima, A. Momotake, T. Arai, Tetrahedron Lett. 45, 9377 (2004)
A. Ohshima, A. Momotake, R. Nagahata, T. Arai, J. Phys. Chem. A 109, 9731 (2005)
A. Ohshima, M. Ikegami, Y. Shinohara, A. Momotake, T. Arai, Bull. Chem. Soc. Jpn. 80, 561 (2007)
T. Kanda, A. Momotake, Y. Shinohara, T. Sato, Y. Nishimura, T. Arai, Bull. Chem. Soc. Jpn. 82, 118 (2009)
T. Iijima, A. Momotake, Y. Shinohara, T. Sato, Y. Nishimura, T. Arai, J. Phys. Chem. A 114, 1603 (2010)
T. Arai, M. Ikegami, Chem. Lett. 28, 965 (1999)
M. Obi, H. Sakuragi, T. Arai, Chem. Lett. 27, 169 (1998)
T. Arai, M. Moriyama, K. Tokumaru, J. Am. Chem. Soc. 116, 3171 (1994)
F.D. Lewis, B.A. Yoon, T. Arai, T. Iwasaki, K. Tokumaru, J. Am. Chem. Soc. 117, 3029 (1995)
T. Arai, M. Obi, T. Iwasaki, K. Tokumaru, F.D. Lewis, J. Photochem. Photobiol. A 96, 65 (1996)
J.B. Harborne, Comparative Biochemistry of the Flavonoids (Academic Press, New York, 1967)
Z. Nowakowska, Eur. J. Med. Chem. 42, 125 (2007)
P.-T. Chou, M.L. Martinez, W.C. Copper, J. Am. Chem. Soc. 114, 4943 (1992)
T. Arai, Y. Norikane, Chem. Lett. 26, 339 (1997)
Y. Norikane, H. Itoh, T. Arai, J. Phys. Chem. A 106, 2766 (2002)
K. Tokumura, K. Nagaosa, Y. Ohta, R. Matsushima, Chem. Phys. Lett. 295, 516 (1998)
K. Kaneda, T. Arai, Photochem. Photobiol. Sci. 2, 402 (2003)
K. Kaneda, T. Arai, Org. Biomol. Chem. 1, 2041 (2003)
K. Kaneda, S. Sato, H. Hamaguchi, T. Arai, Bull. Chem. Soc. Jpn. 77, 1529 (2004)
T. Teshima, M. Takeishi, T. Arai, New J. Chem. 33, 1393 (2009)
Y. Sugiyama, Y. Shinohara, A. Momotake, K. Takahashi, Y. Kanna, Y. Nishimura, T. Arai, J. Phys. Chem. A 114, 10929 (2010)
W.H. Melhuish, J. Phys. Chem. 65, 229 (1961)
K. Sonogashira, Y. Tohda, N. Hagiwara, Tetrahedron Lett. 50, 4467 (1975)
Y. Amatatsu, M. Hosokawa, J. Phys. Chem. A 108, 10238 (2004)
R. Matsushima, H. Kageyama, J. Chem. Soc. Perkin Trans. 2 6, 743 (1985)
R. Matsushima, I. Hirao, Bull. Chem. Soc. Jpn. 53, 518 (1980)
Acknowledgments
This work was supported by a Grant-in-Aid for Scientific Research in a Priority Area “New Frontiers in Photochromism” (no. 471) from the Ministry of Education, Culture, Sports, Science, and Technology (MEXT) and a Grant-in-Aid for Scientific Research (B) (no. 23350075) from the Japan Society for the Promotion of Science (JSPS).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tasaki, S., Momotake, A., Kanna, Y. et al. Excited state intramolecular hydrogen atom transfer of phenylethynyl-substituted 2′-hydroxychalcones. Res Chem Intermed 39, 61–72 (2013). https://doi.org/10.1007/s11164-012-0632-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-012-0632-8