Abstract
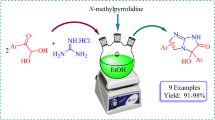
4-Substituted 3-hydroxy-2(1H)-pyridinones 2 were prepared by Mannich reaction of 3-hydroxy-2(1H)-pyridinones with secondary amines and formaldehyde. The isolated products were then reacted with aromatic amines in the presence of NaIO3 as oxidant via oxidation–Michael addition to yield 4,5,6-trisubstituted-2,3-pyridinediones 3 in good yields (80.2–88.9%). The structures of the products were characterized by 1H NMR, ESI-MS, and IR, and elemental analysis.


Similar content being viewed by others
References
J.W. Daly, J. Nat. Prod. 61, 162 (1998)
P.A.O. Lunkett, Nat. Prod. Rep. 11, 581 (1994)
M. Balasubramanian, J. G. Keay, in Comprehensive Heterocyclic Chemistry II, vol. 5 eds. by A.R. Katritzky, C.W. Rees, E.F.V. Scriven (Pergamon Press, Oxford, 1996) pp. 245–252
M. Abaszadeh, H. Sheibani, K. Saidi, J. Heterocyclic. Chem 46, 96 (2009)
M. Streater, P.D. Taylor, R.C. Hider, J. Porter, J. Med. Chem. 33, 1749 (1990)
P.S. Dobbin, R.C. Hider, A.D. Hall, P. Taylor, P. Sarpong, J.B. Porter, G. Xiao, J. Med. Chem. 36, 2448 (1993)
O. Shalev, T. Repka, A. Goldfarb, L. Grinberg, A. Abrahamov, N.F. Olivieri, E.A. Rachmilewitz, R.P. Hebbel, Blood 86, 2008 (1995)
J.J. Molenda, M.M. Jones, D.S. Johnston, E.M. Walker, D. Cannon, J. Med. Chem. 37, 4363 (1994)
A.G. Osborne, L. Jackson, P.D. Taylor, Spectrochim. Acta. Port A 49, 1703 (1993)
A. Nakamura, S. Kamiya, Chem. Pharm. Bull. 16, 1466 (1968)
Acknowledgments
We are grateful for financial support from the Key Laboratory of Theoretical Chemistry and Molecular Simulation of Ministry of Education (no. LKF0905) and the science and technology project of Hunan Province (no. 2011SK3122).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xie, W.L., Xiao, C.P., Xie, L. et al. Synthesis of 4,5,6-trisubstituted 2,3-pyridinediones via Mannich reaction and oxidation–Michael addition of 3-hydroxy-2(1H)-pyridinones. Res Chem Intermed 38, 1699–1705 (2012). https://doi.org/10.1007/s11164-012-0495-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-012-0495-z