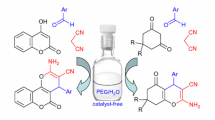
Abstract
Using K2CO3 as a base and CH3CN as solvent, different kinds of N-[5-alkoxy-2(5H)-furanonyl] amino acids were reacted with propargyl bromide via substitution reaction at 40 °C to give 16 N-[5-alkoxy-2(5H)-furanonyl] amino acid propargyl esters with the yields of 44–85% (mostly over 74%). The structures of all newly synthesized compounds were elucidated and confirmed by FTIR, UV, 1H NMR, 13C NMR, MS, and elemental analysis. The rapid, efficient, and brief synthesis of the series propargyl esters with multiple bioactive units, will afford not only a basis for the activity test of potential drug molecules, but also an important synthetic strategy for 2(5H)-furanone derivatives with polyfunctional groups.

Similar content being viewed by others
References
E. Lattmann, S. Dunn, S. Niamsanit, N. Sattayasai, Bioorg. Med. Chem. Lett. 15, 919 (2005)
E. Lattmann, N. Sattayasai, C.S. Schwalbe, S. Niamsanit, D.C. Billington, P. Lattmann, C.A. Langley, H. Singh, S. Dunn, Curr. Drug Discov. Technol. 3, 125 (2006)
M.D. Guerrero, M. Aquino, I. Bruno, M.C. Terencio, M. Paya, R. Riccio, L. Gomez-Paloma, J. Med. Chem. 50, 2176 (2007)
H.V.E. Juan, J.R. Saad, O.S. Giordano, C. Garcia, T. Martin, V.S. Martin, M.E. Sosa, C.E. Tonn, J. Nat. Prod. 71, 190 (2008)
M.-X. Wei, L. Feng, X.-Q. Li, X.-Z. Zhou, Z.-H. Shao, Eur. J. Med. Chem. 44, 3340 (2009)
S.M. Pimentel-Elardo, S. Kozytska, T.S. Bugni, C.M. Ireland, H. Moll, U. Hentschel, Mar. Drugs 8, 373 (2010)
K.R. Prasad, V.R. Gandi, Tetrahedron Asym. 21, 2848 (2010)
E. Gondela, K.Z. Walczak, Eur. J. Med. Chem. 45, 3993 (2010)
R. Surmont, G. Verniest, N. De Kimpe, J. Org. Chem. 75, 5750 (2010)
R.C.V. Sindhu, P.K. Sreekumar, Int. J. Pharm. Pharm. Sci. 3, 225 (2011)
Y.-Q. Mo, Z.-Y. Wang, W.-J. Mei, J.-H. Fu, Y.-H. Tan, S.-H. Luo, Monatsh. Chem. (2011, in press), http://www.springerlink.com/content/nw41x166414832r2/. doi:10.1007/s00706-011-0594-3
B. Sauer, J. Gilbert, J.A. Sakoff, A. McCluskey, Lett. Drug Des. Discov. 6, 1 (2009)
S. Mecklenburg, S. Shaaban, L.A. Ba, T. Burkholz, T. Schneider, B. Diesel, A.K. Kiemer, A. Roeseler, K. Becker, J. Reichrath, A. Stark, W. Tilgen, M. Abbas, L.A. Wessjohann, F. Sasse, C. Jacob, Org. Biomol. Chem. 7, 4753 (2009)
C.L. Martin, L.E. Overman, J.M. Rohde, J. Am. Chem. Soc. 132, 4894 (2010)
R. Gutierrez-Abad, O. Illa, R.M. Ortuno, Org. Lett. 12, 3148 (2010)
C.-C. Ma, Z.-P. Liu, H.-L. Song, R.-T. Jiang, F.-W. He, S.-T. Ma, J. Antibiot. 63, 3 (2010)
X.-M. Li, M. Zhao, Y.-R. Tang, C. Wang, Z.-D. Zhang, S.-Q. Peng, Eur. J. Med. Chem. 43, 8 (2008)
Q.-D. Wang, S.-J. Xue, J.-F. Shen, Z.-J. Cai, Chin. J. Org. Chem. 28, 521 (2008)
X. Tian, A.G. Switzer, S.A. Derose, R.K. Mishra, M.G. Solinsky, R.N. Mumin, F.H. Ebetino, L.R. Jayasinghe, M.E. Webster, A.O. Colson, D. Crossdoersen, B.B. Pinney, J.A. Farmer, M.E. Dowty, C.M. Obringer, C.A. Cruze, M.L. Burklow, P.M. Suchanek, L. Dong, M.K. Dirr, R.J. Sheldon, J.A. Wos, J. Med. Chem. 51, 6055 (2008)
A.R. Katritzky, Q.Y. Chen, S.R. Tala, Chem. Biol. Drug Des. 73, 611 (2009)
A. Karakurt, M. Oezalp, S. Isik, J.P. Stables, S. Dalkara, Bioorg. Med. Chem. 18, 2902 (2010)
C. Sundararajan, T.R. Besanger, R. Labiris, K.J. Guenther, T. Strack, R. Garafalo, T.T. Kawabata, D. Finco-Kent, J. Zubieta, J.W. Babich, J.F. Valliant, J. Med. Chem. 53, 2612 (2010)
Y. Luo, B. Knuckley, M. Bhatia, P.J. Pellechia, P.R. Thompson, J. Am. Chem. Soc. 128, 14468 (2006)
T. Lee, M. Cho, S.Y. Ko, H.J. Youn, D.J. Baek, W.J. Cho, C.Y. Kang, S. Kim, J. Med. Chem. 50, 585 (2007)
I. Carvalho, P. Andrade, V.L. Campo, P.M.M. Guedes, R. Sesti-Costa, J.S. Silva, S. Schenkman, S. Dedola, L. Hill, M. Rejzek, S.A. Nepogodiev, R.A. Field, Bioorg. Med. Chem. 18, 2412 (2010)
C.O. Kappe, E.V. Eycken, Chem. Soc. Rev. 39, 1280 (2010)
J.-Q. Zhang, J. Kemmink, D.T.S. Rijkers, R.M.J. Liskamp, Org. Lett. 13, 3438 (2011)
C.-H. Liu, X.-F. Gu, Y.-Z. Zhu, Bioorg. Med. Chem. Lett. 20, 6942 (2010)
G.M. Raner, S. Cornelious, K. Moulick, Y.-Q. Wang, A. Mortenson, N.B. Cech, Food Chem. Toxicol. 45, 2359 (2007)
S. Roy, S.B. Singh, J. Chromatogr. A 1065, 199 (2005)
Y.-H. Tan, Z.-Y. Wang, Z.-F. Hao, J.-X. Li, Chin. J. Org. Chem. 31, 1222 (2011)
L.J. Loeffler, Z. Sajadi, I.H. Hall, J. Med. Chem. 20, 1578 (1977)
S. Werner, P.S. Iyer, M.D. Fodor, C.M. Coleman, L.A. Twining, B. Mitasev, K.M. Brummond, J. Comb. Chem. 8, 368 (2006)
K.M. Brummond, B. Mitasev, Org. Lett. 6, 2245 (2004)
S. Loethen, T. Ooya, C.H. Soo, N. Yui, D.H. Thompson, Biomacromolecules 7, 2501 (2006)
V. Haridas, K. Lal, Y.K. Sharma, S. Upreti, Org. Lett. 10, 1645 (2008)
S.P. Bew, G.D. Hiatt-Gipson, J. Org. Chem. 75, 3897 (2010)
Y. Liao, R. Fathi, Z. Yang, Org. Lett. 5, 909 (2003)
G. Hilt, C. Hengst, M. Arndt, Synthesis 395 (2009)
X. Meng, C.-B. Li, B.-C. Han, T.-S. Wang, B.-H. Chen, Tetrahedron 66, 4029 (2010)
J.-X. Li, H.-R. Liang, Z.-Y. Wang, J.-H. Fu, Monatsh. Chem. 142, 507 (2011)
J.-X. Li, F.-L. Xue, Y.-H. Tan, S.-H. Luo, Z.-Y. Wang, Acta. Chim. Sinica 69, 1688 (2011)
R.J. Cox, D.J. Ritson, T.A. Dane, J. Berge, J.P.H. Charmant, A. Kantacha, Chem. Commun. 1037 (2005)
W.-B. Yi, C. Cai, X. Wang, Eur. J. Org. Chem. 3445 (2007)
M. Beauperin, A. Job, H. Cattey, S. Royer, P. Meunier, J.C. Hierso, Organometallics 29, 2815 (2010)
L.-M. Tao, Y. Liang, J.-H. Li, Chin. J. Org. Chem. 27, 1078 (2007)
L. Zani, S. Alesi, P.G. Cozzi, C. Bolm, J. Org. Chem. 71, 1558 (2006)
V.K.Y. Lo, K.K.Y. Kung, M.K. Wong, C.-M. Che, J. Organomet. Chem. 694, 583 (2009)
S. Samai, G.C. Nandi, M.S. Singh, Tetrahedron Lett. 51, 5555 (2010)
S. Murarka, A. Studer, Org. Lett. 13, 2746 (2011)
Acknowledgments
We are grateful to the National Natural Science Foundation of China (No. 20772035) and the Natural Science Foundation of Guangdong Province (No. 5300082) for financial support.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tan, YH., Wang, ZY., Qi, J. et al. Synthesis of N-[5-alkoxy-2(5H)-furanonyl] amino acid propargyl esters. Res Chem Intermed 38, 925–936 (2012). https://doi.org/10.1007/s11164-011-0429-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-011-0429-1