Abstract
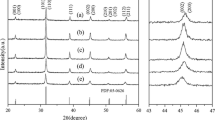
The tetragonal TbPO4 was controlled synthesized via a simple solvothermal route by using glycol as solvent. The structures and micromorphologies of the as-synthesized TbPO4 have been investigated by means of X-ray powder diffraction (XRD) and field-emission scanning electronic microscopy (FE-SEM). The results showed that the tetragonal TbPO4 are composed of square-like particles about 200 nm in diameter. The photoluminescence spectra exhibited that the emission spectra of the sample could be divided into two parts, representing the 5D4 → 7FJ (J = 6, 5, 4, 3) transitions and 5D3 → 7FJ (J = 6, 5, 4, 3) transitions of Tb3+.





Similar content being viewed by others
References
G.Y. Adachi, N. Imanaka, Chem. Rev. 98, 1479 (1998)
R.X. Yan, X.M. Sun, X. Wang, Q. Peng, Y.D. Li, Chem. Eur. J. 11, 2183 (2005)
S.P. Fricker, Chem. Soc. Rev. 35, 524 (2006)
J. Dhanaraj, R. Jagannathan, T.R.N. Kutty, C.-H. Lu, J. Phys. Chem. B 105, 11098 (2001)
H. Mass, A. Currao, G. Calzaferri, Angew. Chem. Int. Ed. 41, 2495 (2002)
F.H. Firsching, S.N. Brune, J. Chem. Eng. Data 36, 93 (1991)
A. Rouanet, J.J. Serra, K. Allaf, V.P. Orlovskii, Neorg. Mater. 17, 104–109 (1981)
Y. Guo, P. Woznicki, A. Barkatt, E.E. Saad, I.G. Talmy, J. Mater. Res. 11, 639 (1996)
O. Lehmann, K. Kompe, M. Haase, J. Am. Chem. Soc. 126, 14935 (2004)
K. Riwotzki, H. Meyssamy, A. Kornowski, M. Haase, J. Phys. Chem. B 104, 2824 (2000)
P. Schuetz, F. Caruso, Chem. Mater. 14, 4509 (2002)
Y.P. Fang, A.W. Xu, R.Q. Song, H.X. Zhang, L.P. You, J.C. Yu, H.Q. Liu, J. Am. Chem. Soc. 125, 16025 (2003)
Y.W. Zhang, Z.G. Yan, L.P. You, R. Si, C.H. Yan, Eur. J. Inorg. Chem. 22, 4099 (2003)
W.B. Bu, Z.L. Hua, H.R. Chen, J.L. Shi, J. Phys. Chem. B 109, 14461 (2005)
J. Bao, R. Yu, J. Zhang, D. Wang, J. Deng, J. Chen, X. Xing, Scripta Mater. 62, 133 (2010)
J. Bao, R. Yu, J. Zhang, X. Yang, D. Wang, J. Deng, J. Chen, X. Xing, Cryst. Eng. Commun. 11, 1630 (2009)
K. Riwotzki, H. Meyssamy, H. Schnablegger, A. Kornowski, M. Haase, Angew. Chem. Int. Ed. 40, 573 (2001)
K. Kompe, H. Borchert, J. Storz, A. Lobo, S. Adam, T. Moller, M. Haase, Angew. Chem. Int. Ed. 42, 5513 (2003)
G. Buhler, C. Feldmann, Angew. Chem. Int. Ed. 45, 4864 (2006)
R. Kijkowska, J. Mater. Sci. 38, 229 (2003)
J.R. Bao, R.B. Yu, J.Y. Zhang, X.D. Yang, D. Wang, J.X. Deng, J. Chen, X.R. Xing, Eur. J. Inorg. Chem. 2388 (2009)
W.H. Di, X.J. Wang, P.P. Zhu, B.J. Chen, J. Solid State Chem. 180, 467 (2007)
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (No. 20871015, 51072020), “Program for New Century Excellent Talents in University” (NCET), Beijing Natural Science Foundation (No. 2092019, 2082022), GF Fundamental Research Foundation (A1320070102), and the National Natural Science Foundation of Inner Mongolia Autonomous Region (No. 09208).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yu, R., Bao, J., Yang, X. et al. Controlled synthesis of tetragonal terbium orthophosphate nanostructures through a solvothermal route. Res Chem Intermed 37, 145–151 (2011). https://doi.org/10.1007/s11164-011-0261-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-011-0261-7