Abstract
FGF21 is a peptide hormone that regulates homeostasis of lipid and glucose as well as energy metabolism. It is mainly expressed and secreted in liver and adipose tissues, and it is expressed in lower amounts in the aorta. Recent clinical and preclinical studies indicate increased serum FGF21 levels in atherosclerosis patients. Also, FGF21 therapy has been reported to reduce the initiation and progression of atherosclerosis in animal models and in vitro studies. Moreover, growing evidence indicates that administration of exogenous FGF21 induces anti-atherosclerotic effects, because of its ability to reduce lipid profile, alleviation of oxidative stress, inflammation, and apoptosis. Therefore, FGF21 can not only be considered as a biomarker for predicting atherosclerosis, but also induce protective effects against atherosclerosis. Besides, serum levels of FGF21 increase in various diseases including in diabetes mellitus, hypertension, and obesity, which may be related to initiating and exacerbating atherosclerosis. On the other hand, FGF21 therapy significantly improves lipid profiles, and reduces vascular inflammation and oxidative stress in atherosclerosis related diseases. Therefore, further prospective studies are needed to clarify whether FGF21 can be used as a prognostic biomarker to identify individuals at future risk of atherosclerosis in these atherosclerosis-associated diseases. In this review, we will discuss the possible mechanism by which FGF21 protects against atherosclerosis.



Similar content being viewed by others
References
Hoefer IE, Steffens S, Ala-Korpela M, Bäck M, Badimon L, Bochaton-Piallat M-L, et al. Novel methodologies for biomarker discovery in atherosclerosis. 2015;36(39):2635–42.
Fisher FM, Maratos-Flier E. Understanding the physiology of FGF21. Annu Rev Physiol. 2016;78:223–41.
Nishimura T, Nakatake Y, Konishi M, Itoh N. Identification of a novel FGF, FGF-21, preferentially expressed in the liver1. Biochimica et Biophysica Acta. 2000;1492(1):203–6.
Hondares E, Iglesias R, Giralt A, Gonzalez FJ, Giralt M, Mampel T, et al. Thermogenic activation induces FGF21 expression and release in brown adipose tissue. J Biol Chem. 2011:jbc. M110. 215889.
Fon Tacer K, Bookout AL, Ding X, Kurosu H, John GB, Wang L, et al. Research resource: comprehensive expression atlas of the fibroblast growth factor system in adult mouse. Mol Endocrinol. 2010;24(10):2050–64.
Inagaki T, Dutchak P, Zhao G, Ding X, Gautron L, Parameswara V, et al. Endocrine regulation of the fasting response by PPARα-mediated induction of fibroblast growth factor 21. Cell Metab. 2007;5(6):415–25.
Muise ES, Azzolina B, Kuo DW, El-Sherbeini M, Tan Y, Yuan X, et al. Adipose fibroblast growth factor 21 is up-regulated by peroxisome proliferator-activated receptor γ and altered metabolic states. Mol Pharmacol. 2008;74(2):403–12.
Beenken A, Mohammadi M. The structural biology of the FGF19 subfamily. Endocrine FGFs and Klothos: Springer; 2012. p. 1–24.
Lin Z, Pan X, Wu F, Ye D, Zhang Y, Wang Y, et al. Fibroblast growth factor 21 prevents atherosclerosis by suppression of hepatic sterol regulatory element-binding Protein-2 and induction of adiponectin in mice. Circulation. 2015;131:1861–71.
Xiao Y, Liu L, Xu A, Zhou P, Long Z, Tu Y, et al. Serum fibroblast growth factor 21 levels are related to subclinical atherosclerosis in patients with type 2 diabetes. 2015;14(1):72.
Zhang X, Hu Y, Zeng H, Li L, Zhao J, Zhao J, et al. Serum fibroblast growth factor 21 levels is associated with lower extremity atherosclerotic disease in Chinese female diabetic patients. Cardiovasc Diabetol. 2015;14(1):32.
Yan X, Chen J, Zhang C, Zeng J, Zhou S, Zhang Z, et al. Fibroblast growth factor 21 deletion aggravates diabetes-induced pathogenic changes in the aorta in type 1 diabetic mice. Cardiovasc Diabetol. 2015;14(1):77.
Gaich G, Chien JY, Fu H, Glass LC, Deeg MA, Holland WL, et al. The effects of LY2405319, an FGF21 analog, in obese human subjects with type 2 diabetes. Cell Metab. 2013;18(3):333–40.
Talukdar S, Zhou Y, Li D, Rossulek M, Dong J, Somayaji V, et al. A long-acting FGF21 molecule, PF-05231023, decreases body weight and improves lipid profile in non-human primates and type 2 diabetic subjects. Cell Metab. 2016;23(3):427–40.
Kharitonenkov A, Wroblewski VJ, Koester A, Chen Y-F, Clutinger CK, Tigno XT, et al. The metabolic state of diabetic monkeys is regulated by fibroblast growth factor-21. Endocrinology. 2007;148(2):774–81.
Lee CH, Woo YC, Chow WS, Cheung CYY, Fong CHY, Yuen MMA, et al. Role of circulating fibroblast growth factor 21 measurement in primary prevention of coronary heart disease among Chinese patients with type 2 diabetes mellitus. J Am Heart Assoc. 2017;6(6):e005344.
Zhang X, Yeung DC, Karpisek M, Stejskal D, Zhou Z-G, Liu F, et al. Serum FGF21 levels are increased in obesity and are independently associated with the metabolic syndrome in humans. Diabetes. 2008;57:1246–53.
Chou R-H, Huang P-H, Hsu C-Y, Chang C-C, Leu H-B, Huang C-C, et al. Circulating fibroblast growth factor 21 is associated with diastolic dysfunction in heart failure patients with preserved ejection fraction. Sci Rep. 2016;6:33953.
Li H, Horke S, Förstermann U. Vascular oxidative stress, nitric oxide and atherosclerosis. Atherosclerosis. 2014;237(1):208–19.
Husain K, Hernandez W, Ansari RA, Ferder L. Inflammation, oxidative stress and renin angiotensin system in atherosclerosis. World J Biol Chem. 2015;6(3):209–17.
Hamza SM, Dyck JR. Systemic and renal oxidative stress in the pathogenesis of hypertension: modulation of long-term control of arterial blood pressure by resveratrol. Front Physiol. 2014;5:292.
Rahman T, Hosen I, Islam MT, Shekhar HU. Oxidative stress and human health. Adv Biosci Biotechnol. 2012;3(07):997–1019.
Gómez-Sámano MÁ, Grajales-Gómez M, Zuarth-Vázquez JM, Navarro-Flores MF, Martínez-Saavedra M, Juárez-León ÓA, et al. Fibroblast growth factor 21 and its novel association with oxidative stress. Redox Biol. 2017;11:335–41.
Salminen A, Kaarniranta K, Kauppinen A. Integrated stress response stimulates FGF21 expression: systemic enhancer of longevity. Cell Signal. 2017;40:10–21.
Lü Y, Liu J-H, Zhang L-K, Du J, Zeng X-J, Hao G, et al. Fibroblast growth factor 21 as a possible endogenous factor inhibits apoptosis in cardiac endothelial cells. Chin Med J. 2010;123(23):3417–21.
Zhu S, Ma L, Wu Y, Ye X, Zhang T, Zhang Q, et al. FGF21 treatment ameliorates alcoholic fatty liver through activation of AMPK-SIRT1 pathway. Acta Biochim Biophys Sin. 2014;46(12):1041–8.
Zhu W, Wang C, Liu L, Li Y, Li X, Cai J, et al. Effects of fibroblast growth factor 21 on cell damage in vitro and atherosclerosis in vivo. Can J Physiol Pharmacol. 2014;92(11):927–35.
Pober JS, Min W, Bradley JR. Mechanisms of endothelial dysfunction, injury, and death. Annu Rev Pathol. 2009;4:71–95.
Jiang S, Yan C, Fang Q-C, Zhang Y-L, Liu Y, Deng Y-P, et al. Fibroblast growth factor 21 is regulated by the IRE1α-XBP1 branch of the unfolded protein response and counteracts ER stress-induced hepatic steatosis. J Biol Chem. 2014:jbc. M114. 565960.
Wan X-s, Lu X-h, Xiao Y-c, Lin Y, Zhu H, Ding T, et al. ATF4-and CHOP-dependent induction of FGF21 through endoplasmic reticulum stress. BioMed research international. 2014;2014.
Dong K, Li H, Zhang M, Jiang S, Chen S, Zhou J, et al. Endoplasmic reticulum stress induces up-regulation of hepatic β-klotho expression through ATF4 signaling pathway. Biochem Biophys Res Commun. 2015;459(2):300–5.
Schaap FG, Kremer AE, Lamers WH, Jansen PL, Gaemers IC. Fibroblast growth factor 21 is induced by endoplasmic reticulum stress. Biochimie. 2013;95(4):692–9.
Malhotra JD, Kaufman RJ. Endoplasmic reticulum stress and oxidative stress: a vicious cycle or a double-edged sword? Antioxid Redox Signal. 2007;9(12):2277–94.
Teske BF, Wek SA, Bunpo P, Cundiff JK, McClintick JN, Anthony TG, et al. The eIF2 kinase PERK and the integrated stress response facilitate activation of ATF6 during endoplasmic reticulum stress. Mol Biol Cell. 2011;22(22):4390–405.
Tabas I, Ron D. Integrating the mechanisms of apoptosis induced by endoplasmic reticulum stress. Nat Cell Biol. 2011;13(3):184–90.
Regulation H. The Unfolded Protein Response: From Stress Pathway to. Science. 2011;1209038(1081):334.
Wang X-M, Song S-S, Xiao H, Gao P, Li X-J, Si L-Y. Fibroblast growth factor 21 protects against high glucose induced cellular damage and dysfunction of endothelial nitric-oxide synthase in endothelial cells. Cell Physiol Biochem. 2014;34(3):658–71.
Jia H, Cheng J, Zhou Q, Peng J, Pan Y, Han H. Fibroblast growth factor 21 attenuates inflammation and oxidative stress in atherosclerotic rat via enhancing the Nrf1-ARE signaling pathway. Int J Clin Exp Pathol. 2018;11(3):1308–17.
Juan S, Lee T, Tseng K, Liou J, Shyue S, Wu K, et al. Adenovirus-mediated heme oxygenase-1 gene transfer inhibits the development of atherosclerosis in apolipoprotein E-deficient mice. Circulation. 2001;104(13):1519–25.
Yet S-F, Layne MD, Liu X, Chen Y-H, Ith B, Sibinga NE, et al. Absence of heme oxygenase-1 exacerbates atherosclerotic lesion formation and vascular remodeling. FASEB J. 2003;17(12):1759–61.
Biswas SK, Newby DE, Rahman I, Megson IL. Depressed glutathione synthesis precedes oxidative stress and atherogenesis in Apo-E−/− mice. Biochem Biophys Res Commun. 2005;338(3):1368–73.
Kim S-Y, Jeoung NH, Oh CJ, Choi Y-K, Lee H-J, Kim H-J, et al. Activation of NAD (P) H: quinone oxidoreductase 1 prevents arterial restenosis by suppressing vascular smooth muscle cell proliferation. Circ Res. 2009;104(7):842–50.
Fukai T, Ushio-Fukai M. Superoxide dismutases: role in redox signaling, vascular function, and diseases. Antioxid Redox Signal. 2011;15(6):1583–606.
Zhao Y, Vanhoutte PM, Leung SW. Vascular nitric oxide: Beyond eNOS. J Pharmacol Sci. 2015;129(2):83–94.
Kolluru GK, Bir SC, Kevil CG. Endothelial dysfunction and diabetes: effects on angiogenesis, vascular remodeling, and wound healing. J Vasc Med. 2012;2012:1–30.
Jahanban-Esfahlan R, de la Guardia M, Ahmadi D, Yousefi B. Modulating tumor hypoxia by nanomedicine for effective cancer therapy. J Cell Physiol. 2018;233(3):2019–2031.
Förstermann U. Oxidative stress in vascular disease: causes, defense mechanisms and potential therapies. Nat Rev Cardiol. 2008;5(6):338.
Landmesser U, Dikalov S, Price SR, McCann L, Fukai T, Holland SM, et al. Oxidation of tetrahydrobiopterin leads to uncoupling of endothelial cell nitric oxide synthase in hypertension. J Clin Invest. 2003;111(8):1201–9.
Chen D-D, Chen L-Y, Xie J-B, Shu C, Yang T, Zhou S, et al. Tetrahydrobiopterin regulation of eNOS redox function. Curr Pharm Des. 2014;20(22):3554–62.
McCabe TJ, Fulton D, Roman LJ, Sessa WC. Enhanced electron flux and reduced calmodulin dissociation may explain “calcium-independent” eNOS activation by phosphorylation. J Biol Chem. 2000;275(9):6123–8.
Huang PL. eNOS, metabolic syndrome and cardiovascular disease. Trends Endocrinol Metab. 2009;20(6):295–302.
Wu X, Qi Y-F, Chang J-R, Lu W-W, Zhang J-S, Wang S-P, et al. Possible role of fibroblast growth factor 21 on atherosclerosis via amelioration of endoplasmic reticulum stress-mediated apoptosis in apoE−/− mice. Heart Vessel. 2015;30(5):657–68.
Li Y, Guo Y, Tang J, Jiang J, Chen Z. New insights into the roles of CHOP-induced apoptosis in ER stress. Acta Biochim Biophys Sin. 2014;46(8):629–40.
Tsukano H, Gotoh T, Endo M, Miyata K, Tazume H, Kadomatsu T, et al. The endoplasmic reticulum stress-C/EBP homologous protein pathway-mediated apoptosis in macrophages contributes to the instability of atherosclerotic plaques. Arterioscler Thromb Vasc Biol. 2010;30(10):1925–32.
Thorp E, Li G, Seimon TA, Kuriakose G, Ron D, Tabas I. Reduced apoptosis and plaque necrosis in advanced atherosclerotic lesions of Apoe−/− and Ldlr−/− mice lacking CHOP. Cell Metab. 2009;9(5):474–81.
Kutuk O, Basaga H. Bcl-2 protein family: implications in vascular apoptosis and atherosclerosis. Apoptosis. 2006;11(10):1661–75.
Liang P, Zhong L, Gong L, Wang J, Zhu Y, Liu W, et al. Fibroblast growth factor 21 protects rat cardiomyocytes from endoplasmic reticulum stress by promoting the fibroblast growth factor receptor 1-extracellular signal-regulated kinase 1/2 signaling pathway. Int J Mol Med. 2017;40(5):1477–85.
Li E, Wang T, Wang F, Wang T, Sun L-Q, Li L, et al. FGF21 protects against ox-LDL induced apoptosis through suppressing CHOP expression in THP1 macrophage derived foam cells. BMC Cardiovasc Disord. 2015;15(1):80.
Weiss D, Kools JJ, Taylor WR. Angiotensin II–induced hypertension accelerates the development of atherosclerosis in ApoE-deficient mice. 2001.
Weiss D, Taylor WR. Deoxycorticosterone acetate salt hypertension in apolipoprotein E−/− mice results in accelerated atherosclerosis: the role of angiotensin II. Hypertension. 2008;51(2):218–24.
Knowles JW, Maeda N. Genetic modifiers of atherosclerosis in mice. Arterioscler Thromb Vasc Biol. 2000;20(11):2336–45.
Casalnuovo G, Gerdts E, Simone GD, Izzo R, Marco MD, Giudice R, et al. Arterial stiffness is associated with carotid atherosclerosis in hypertensive patients (the Campania Salute Network). Oxford University Press; 2012.
Yamazaki D, Hitomi H, Nishiyama A. Hypertension with diabetes mellitus complications. Hypertens Res. 2018;1.
Semba RD, Crasto C, Strait J, Sun K, Schaumberg DA, Ferrucci L. Elevated serum fibroblast growth factor 21 is associated with hypertension in community-dwelling adults. J Hum Hypertens. 2013;27(6):397–9.
He J-L, Zhao M, Xia J-J, Guan J, Liu Y, Wang L-Q, et al. FGF21 ameliorates the neurocontrol of blood pressure in the high fructose-drinking rats. Sci Rep. 2016;6:29582.
Pan X, Shao Y, Wu F, Wang Y, Xiong R, Zheng J, et al. FGF21 prevents angiotensin II-induced hypertension and vascular dysfunction by activation of ACE2/angiotensin-(1–7) Axis in mice. Cell Metab. 2018;27:1323–1337.e5.
Montezano AC, Cat AND, Rios FJ, Touyz RM. Angiotensin II and vascular injury. Curr Hypertens Rep. 2014;16(6):431.
Wang Y, Tikellis C, Thomas MC, Golledge J. Angiotensin converting enzyme 2 and atherosclerosis. Atherosclerosis. 2013;226(1):3–8.
Mazzolai L, Duchosal MA, Korber M, Bouzourene K, Aubert JF, Hao H, et al. Endogenous angiotensin II induces atherosclerotic plaque vulnerability and elicits a Th1 response in ApoE−/− mice. Hypertension. 2004;44(3):277–82.
Daugherty A, Manning MW, Cassis LA. Angiotensin II promotes atherosclerotic lesions and aneurysms in apolipoprotein E–deficient mice. J Clin Invest. 2000;105(11):1605–12.
Candido R, Jandeleit-Dahm KA, Cao Z, Nesteroff SP, Burns WC, Twigg SM, et al. Prevention of accelerated atherosclerosis by angiotensin-converting enzyme inhibition in diabetic apolipoprotein E–deficient mice. Circulation. 2002;106(2):246–53.
Daugherty A, Poduri A, Chen X, Lu H, Cassis LA. Genetic variants of the renin angiotensin system: effects on atherosclerosis in experimental models and humans. Curr Atheroscler Rep. 2010;12(3):167–73.
Mazzolai L, Hayoz D. The renin-angiotensin system and atherosclerosis. Curr Hypertens Rep. 2006;8(1):47–53.
Fraga-Silva RA, Ferreira AJ, dos Santos RAS. Opportunities for targeting the angiotensin-converting enzyme 2/angiotensin-(1-7)/mas receptor pathway in hypertension. Curr Hypertens Rep. 2013;15(1):31–8.
Patel VB, Zhong J-C, Grant MB, Oudit GY. Role of the ACE2/angiotensin 1–7 axis of the renin–angiotensin system in heart failure. Circ Res. 2016;118(8):1313–26.
Zhang Y-H, Y-h Z, Dong X-F, Hao Q-Q, Zhou X-M, Yu Q-T, et al. ACE2 and Ang-(1–7) protect endothelial cell function and prevent early atherosclerosis by inhibiting inflammatory response. Inflamm Res. 2015;64(3–4):253–60.
da Silveira KD, Coelho FM, Vieira AT, Sachs D, Barroso LC, Costa VV, et al. Anti-inflammatory effects of the activation of the angiotensin-(1–7) receptor, MAS, in experimental models of arthritis. J Immunol. 2010;1000314.
Zhong J, Guo D, Chen CB, Wang W, Schuster M, Loibner H, et al. Prevention of angiotensin II–mediated renal oxidative stress, inflammation, and fibrosis by angiotensin-converting enzyme 2. Hypertension. 2011;57(2):314–22.
Benter IF, Yousif MH, Dhaunsi GS, Kaur J, Chappell MC, Diz DI. Angiotensin-(1–7) prevents activation of NADPH oxidase and renal vascular dysfunction in diabetic hypertensive rats. Am J Nephrol. 2008;28(1):25–33.
Hsuchou H, Pan W, Kastin AJ. The fasting polypeptide FGF21 can enter brain from blood. Peptides. 2007;28(12):2382–6.
Cheng WH, Lu PJ, Hsiao M, Hsiao CH, Ho WY, Cheng PW, et al. Renin activates PI3K-Akt-eNOS signalling through the angiotensin AT1 and mas receptors to modulate central blood pressure control in the nucleus tractus solitarii. Br J Pharmacol. 2012;166(7):2024–35.
Tsekouras N, Katsargyris A, Skrapari I, Bastounis E, Georgopoulos S, Klonaris C, et al. The role of carotid plaque echogenicity in baroreflex sensitivity. J Vasc Surg. 2011;54(1):93–9.
Bristow J, Gribbin B, Honour A, Pickering T, Sleight P. Diminished baroreflex sensitivity in high blood pressure and ageing man. J Physiol. 1969;202(1):45P–6P.
Lakka TA, Salonen R, Kaplan GA, Salonen JT. Blood pressure and the progression of carotid atherosclerosis in middle-aged men. Hypertension. 1999;34(1):51–6.
Cuspidi C, Sala C, Tadic M, Rescaldani M, De Giorgi GA, Grassi G, et al. Untreated masked hypertension and carotid atherosclerosis: a meta-analysis. Blood Press. 2015;24(2):65–71.
Lonn E, Yusuf S, Dzavik V, Doris C, Yi Q, Smith S, et al. Effects of ramipril and vitamin E on atherosclerosis: the study to evaluate carotid ultrasound changes in patients treated with ramipril and vitamin E (SECURE). Circulation. 2001;103(7):919–25.
Marfella R, Siniscalchi M, Nappo F, Gualdiero P, Esposito K, Sasso FC, et al. Regression of carotid atherosclerosis by control of morning blood pressure peak in newly diagnosed hypertensive patients. Am J Hypertens. 2005;18(3):308–18.
Chow WS, Xu A, Woo YC, Tso AW, Cheung SC, Fong CH, et al. Serum fibroblast growth factor-21 levels are associated with carotid atherosclerosis independent of established cardiovascular risk factors. Arterioscler Thromb Vasc Biol. 2013;33(10):2454–9.
Lusis AJ. Atherosclerosis. Nature. 2000;407(6801):233–41.
Silverman MG, Ference BA, Im K, Wiviott SD, Giugliano RP, Grundy SM, et al. Association between lowering LDL-C and cardiovascular risk reduction among different therapeutic interventions: a systematic review and meta-analysis. Jama. 2016;316(12):1289–97.
Gofman JW, Lindgren F, Elliott H, Mantz W, Hewitt J, Strisower B, et al. The role of lipids and lipoproteins in atherosclerosis. Science. 1950;111(2877):166–86.
Gaich G, Chien JY, Fu H, Glass LC, Deeg MA, Holland WL, et al. The effects of LY2405319, an FGF21 analog, in obese human subjects with type 2 diabetes. Cell Metab. 2013;18(3):333–40.
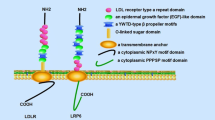
Do HT, Tselykh TV, Mäkelä J, Ho TH, Olkkonen VM, Bornhauser BC, et al. Fibroblast growth factor-21 (FGF21) regulates low-density lipoprotein receptor (LDLR) levels in cells via the E3-ubiquitin ligase Mylip/idol and the Canopy2 (Cnpy2)/Mylip-interacting saposin-like protein (Msap). J Biol Chem. 2012;287(16):12602–11.
Lin X-L, He X-L, Zeng J-F, Zhang H, Zhao Y, Tan J-K, et al. FGF21 increases cholesterol efflux by upregulating ABCA1 through the ERK1/2–PPARγ–LXRα pathway in THP1 macrophage-derived foam cells. DNA Cell Biol. 2014;33(8):514–21.
Shang W, Yu X, Wang H, Chen T, Fang Y, Yang X, et al. Fibroblast growth factor 21 enhances cholesterol efflux in THP-1 macrophage-derived foam cells. Mol Med Rep. 2015;11(1):503–8.
Dong B, Wu M, Cao A, Li H, Liu J. Suppression of idol expression is an additional mechanism underlying statin-induced up-regulation of hepatic LDL receptor expression. Int J Mol Med. 2011;27(1):103–10.
Yvan-Charvet L, Wang N, Tall AR. Role of HDL, ABCA1, and ABCG1 transporters in cholesterol efflux and immune responses. Arterioscler Thromb Vasc Biol. 2010;30(2):139–43.
Tall AR, Yvan-Charvet L, Terasaka N, Pagler T, Wang N. HDL, ABC transporters, and cholesterol efflux: implications for the treatment of atherosclerosis. Cell Metab. 2008;7(5):365–75.
Goldstein BJ, Scalia R. Adiponectin: a novel adipokine linking adipocytes and vascular function. J Clin Endocrinol Metab. 2004;89(6):2563–8.
Villarreal-Molina M, Antuna-Puente B. Adiponectin: anti-inflammatory and cardioprotective effects. Biochimie. 2012;94(10):2143–9.
Liang B, Wang X, Guo X, Yang Z, Bai R, Liu M, et al. Adiponectin upregulates ABCA1 expression through liver X receptor alpha signaling pathway in RAW 264.7 macrophages. Int J Clin Exp Pathol. 2015;8(1):450.
Wolf AM, Wolf D, Rumpold H, Enrich B, Tilg H. Adiponectin induces the anti-inflammatory cytokines IL-10 and IL-1RA in human leukocytes. Biochem Biophys Res Commun. 2004;323(2):630–5.
Holland WL, Adams AC, Brozinick JT, Bui HH, Miyauchi Y, Kusminski CM, et al. An FGF21-adiponectin-ceramide axis controls energy expenditure and insulin action in mice. Cell Metab. 2013;17(5):790–7.
Lin Z, Tian H, Lam KS, Lin S, Hoo RC, Konishi M, et al. Adiponectin mediates the metabolic effects of FGF21 on glucose homeostasis and insulin sensitivity in mice. Cell Metab. 2013;17(5):779–89.
Maeda N, Takahashi M, Funahashi T, Kihara S, Nishizawa H, Kishida K, et al. PPARγ ligands increase expression and plasma concentrations of adiponectin, an adipose-derived protein. Diabetes. 2001;50(9):2094–9.
Holland WL, Adams AC, Brozinick JT, Bui HH, Miyauchi Y, Kusminski CM, et al. An FGF21-adiponectin-ceramide axis controls energy expenditure and insulin action in mice. Cell Metab. 2013;17(5):790–7.
Lin Z, Tian H, Lam KS, Lin S, Hoo RC, Konishi M, et al. Adiponectin mediates the metabolic effects of FGF21 on glucose homeostasis and insulin sensitivity in mice. Cell Metab. 2013;17(5):779–89.
Li G, Yin J, Fu J, Li L, Grant SFA, Li C, et al. FGF21 deficiency is associated with childhood obesity, insulin resistance and hypoadiponectinaemia: the BCAMS study. Diabetes Metab. 2017;43(3):253–60.
Hui E, Xu A, Chow WS, Lee PC, Fong CH, Cheung SC, et al. Hypoadiponectinemia as an independent predictor for the progression of carotid atherosclerosis: a 5-year prospective study. Metab Syndr Relat Disord. 2014;12(10):517–22.
Dieplinger B, Poelz W, Haltmayer M, Mueller T. Hypoadiponectinemia is associated with symptomatic atherosclerotic peripheral arterial disease. Clin Chem Lab Med. 2006;44(7):830–3.
Barseghian A, Gawande D, Bajaj M. Adiponectin and vulnerable atherosclerotic plaques. J Am Coll Cardiol. 2011;57(7):761–70.
Singhal G, Fisher FM, Chee MJ, Tan TG, El Ouaamari A, Adams AC, et al. Fibroblast growth factor 21 (FGF21) protects against high fat diet induced inflammation and islet hyperplasia in pancreas. PLoS One. 2016;11(2):e0148252.
Zhang C, Shao M, Yang H, Chen L, Yu L, Cong W, et al. Attenuation of hyperlipidemia- and diabetes-induced early-stage apoptosis and late-stage renal dysfunction via administration of fibroblast growth factor-21 is associated with suppression of renal inflammation. PLoS One. 2013;8(12):e82275.
Wang WF, Li SM, Ren GP, Zheng W, Lu YJ, Yu YH, et al. Recombinant murine fibroblast growth factor 21 ameliorates obesity-related inflammation in monosodium glutamate-induced obesity rats. Endocrine. 2015;49(1):119–29.
Yu Y, Li S, Liu Y, Tian G, Yuan Q, Bai F, et al. Fibroblast growth factor 21 (FGF21) ameliorates collagen-induced arthritis through modulating oxidative stress and suppressing nuclear factor-kappa B pathway. Int Immunopharmacol. 2015;25(1):74–82.
Li JY, Wang N, Khoso MH, Shen CB, Guo MZ, Pang XX, et al. FGF-21 elevated IL-10 production to correct LPS-induced inflammation. Inflammation. 2018;41(3):751–9.
Liu T, Zhang L, Joo D, Sun S-C. NF-κB signaling in inflammation. Signal Transduct Target Ther. 2017;2:17023.
Yang SJ, Hong HC, Choi HY, Yoo HJ, Cho GJ, Hwang TG, et al. Effects of a three-month combined exercise programme on fibroblast growth factor 21 and fetuin-A levels and arterial stiffness in obese women. 2011;75(4):464–9.
Lenart-Lipińska M, Matyjaszek-Matuszek B, Gernand W, Nowakowski A, Solski JJDr, practice c. Serum fibroblast growth factor 21 is predictive of combined cardiovascular morbidity and mortality in patients with type 2 diabetes at a relatively short-term follow-up. 2013;101(2):194–200.
Díaz-Delfín J, Hondares E, Iglesias R, Giralt M, Caelles C, Villarroya FJE. TNF-α represses β-Klotho expression and impairs FGF21 action in adipose cells: involvement of JNK1 in the FGF21 pathway. 2012;153(9):4238–45.
Shen Y, Ma X, Zhou J, Pan X, Hao Y, Zhou M, et al. Additive relationship between serum fibroblast growth factor 21 level and coronary artery disease. 2013;12(1):124.
Ong K-L, Januszewski AS, O’Connell R, Jenkins AJ, Xu A, Sullivan DR, et al. The relationship of fibroblast growth factor 21 with cardiovascular outcome events in the Fenofibrate Intervention and Event Lowering in Diabetes study. 2015;58(3):464–73.
Tomiyama H, Koji Y, Yambe M, Shiina K, Motobe K, Yamada J, et al. Brachial-ankle pulse wave velocity is a simple and independent predictor of prognosis in patients with acute coronary syndrome 2005;69(7):815–22.
Paraskevas KI, Kotsikoris I, Koupidis SA, Giannoukas AD, Mikhailidis DP. Ankle—brachial index: a marker of both peripheral arterial disease and systemic atherosclerosis as well as a predictor of vascular events. Los Angeles: Sage Publications Sage CA; 2010.
Gaich G, Chien JY, Fu H, Glass LC, Deeg MA, Holland WL, et al. The effects of LY2405319, an FGF21 analog, in obese human subjects with type 2 diabetes. 2013;18(3):333–40.
Chui PC, Antonellis PJ, Bina HA, Kharitonenkov A, Flier JS, Maratos-Flier EJD. Obesity is a fibroblast growth factor 21 (FGF21)-resistant state. 2010;59(11):2781–9.
Ding X, Boney-Montoya J, Owen BM, Bookout AL, Coate KC, Mangelsdorf DJ, et al. βKlotho is required for fibroblast growth factor 21 effects on growth and metabolism. 2012;16(3):387–93.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Farzane Shanebandpour Tabari and Ansar Karimian equally first author
Rights and permissions
About this article
Cite this article
Tabari, F.S., Karimian, A., Parsian, H. et al. The roles of FGF21 in atherosclerosis pathogenesis. Rev Endocr Metab Disord 20, 103–114 (2019). https://doi.org/10.1007/s11154-019-09488-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11154-019-09488-x