Abstract
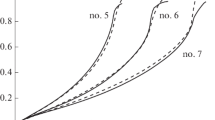
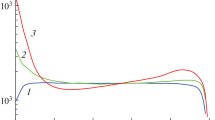
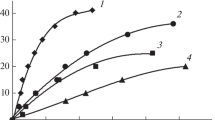
The article presents the results of experiments of the hydrolysis process with a Co/TiO2 catalyst and homogeneous experiments in an aqueous solution of sodium borohydride (NaBH4) aimed to the test the developed model of heterogeneous processes. In data processing of catalytic experiments, we use the method of their correction to exclude the rate of homogeneous hydrolysis. We consider the adsorption/desorption model, in which two types of particles can be adsorbed, i.e. water molecules and BH4−·H+ complexes. As a result of the analysis of possible irreversible heterogeneous reactions and the experimental dependences of the hydrogen generation rates, it is concluded that the main hydrolysis reaction on the Co/TiO2 catalyst occurs upon collisions of BH4− ion from the solution with adsorbed water molecules. We observed the growth of the specific rate of hydrogen generation with an increase in the degree of NaBH4 decomposition in all experiments with this catalyst. Two hypotheses have been suggested to explain this effect.








Similar content being viewed by others
References
Liu BH, Li ZP (2009) A review: hydrogen generation from borohydride hydrolysis reaction. J Power Sources 187:527–534. https://doi.org/10.1016/j.bej.2013.01.012
Walter JC, Zurawski A, Montgomery D et al (2008) Sodium borohydride hydrolysis kinetics comparison for nickel, cobalt, and ruthenium boride catalysts. J Power Sources 179:335–339. https://doi.org/10.1016/j.jpowsour.2007.12.006
Demirci UB, Akdim O, Jerome A et al (2010) Sodium borohydride hydrolysis as hydrogen generator: issues, state of the art and applicability upstream from a fuel cell. Fuel Cells 10:335–350. https://doi.org/10.1002/fuce.200800171
Brack P, Dann SE, Wijayantha KGU (2015) Heterogeneous and homogenous catalysts for hydrogen generation by hydrolysis of aqueous sodium borohydride (NaBH4) solutions. Energy Sci Eng 3:174–188. https://doi.org/10.1002/ese3.67
Abdelhamid HN (2020) A review on hydrogen generation from the hydrolysis of sodium borohydride. Int J Hydrogen Energy 46:726–765. https://doi.org/10.1016/j.ijhydene.2020.09.186
Ruslan N, Yahya MS, Siddique MdNI et al (2022) Review on magnesium hydride and sodium borohydride hydrolysis for hydrogen production. Crystals 12:1376–1390. https://doi.org/10.3390/cryst12101376
Hung AJ, Tsai SF, Hsu YY et al (2008) Kinetics of sodium borohydride hydrolysis reaction for hydrogen generation. Int J Hydrogen Energy 33:6205–6215. https://doi.org/10.1016/j.ijhydene.2008.07.109
Zhang J, Zheng Y, Gore JP et al (2007) 1 kWe sodium borohydride hydrogen generation system Part II: Reactor modeling. J Power Sources 170:150–159. https://doi.org/10.1016/j.jpowsour.2007.03.025
Retnamma R, Novais AQ, Rangel CM (2011) Kinetics of hydrolysis of sodium borohydride for hydrogen production in fuel cell applications: a review. Int J Hydrogen Energy 36:9772–9790. https://doi.org/10.1016/j.ijhydene.2011.04.223
Pena-Alonso R, Sicurelli A, Callone E et al (2007) A picoscale catalyst for hydrogen generation from NaBH4 for fuel cell. J Power Sources 165:315–323. https://doi.org/10.1016/j.jpowsour.2006.12.043
Kaur A, Gangacharyulu D, Bajpai PK (2019) Kinetic studies of hydrolysis reaction of NaBH4 with γ-Al2O3 nanoparticles as catalyst promoter and CoCl2 as catalyst. Braz J Chem Eng 36:929–939. https://doi.org/10.1590/0104-6632.20190362s20180290
Gilani N, Pasikhani JV, Akbari M et al (2019) Hydrogen evolution from catalytic hydrolysis of NaBH4: comparative study between the catalytic activity of TiO2 nanotubes with various arrangements. J Nanostruct 9:587–599. https://doi.org/10.22052/JNS.2019.03.020
Zhang J, Delgass W, Fisher T (2007) Kinetics of Ru-catalyzed sodium borohydride hydrolysis. J Power Sources 164:772–781. https://doi.org/10.1016/j.jpowsour.2006.11.002
Andrieux J, Demirci UB (2011) Langmuir-Hinshelwood kinetic model to capture the cobalt nanoparticles-catalyzed hydrolysis of sodium borohydride over a wide temperature range. Catal Today 170:13–19. https://doi.org/10.1016/j.cattod.2011.01.019
Demirci UB, Miele P (2014) Reaction mechanisms of the hydrolysis of sodium borohydride: a discussion focusing on cobalt-based catalysts. C R Chim 17:707–716. https://doi.org/10.1016/j.crci.2014.01.012
Simagina VI, Netskina OV, Komova OV et al (2012) Progress in the catalysts for H2 generation from NaBH4 fuel. Curr Top Catal 10:147–165
Wei Y, Wang Y, Wei L et al (2018) Highly efficient and reactivated electrocatalyst of ruthenium electrodeposited on nickel foam for hydrogen evolution from NaBH4 alkaline solution. Int J Hydrogen Energy 43:592–600. https://doi.org/10.1016/j.ijhydene.2017.11.010
Xu J, Du X, Wei Q et al (2020) Efficient hydrolysis of sodium borohydride by Co-B supported on nitrogen-doped carbon. Chem Sel 5:6683–6690. https://doi.org/10.1002/slct.201904818
Farrag M, Ali GAM (2022) Hydrogen generation of single alloy Pd/Pt quantum dots over Co3O4 nanoparticles via the hydrolysis of sodium borohydride at room temperature. Sci Rep 12:17040. https://doi.org/10.1038/s41598-022-21064-z
Raza W, Ahmad K, Kim H (2021) Fabrication of defective graphene oxide for efficient hydrogen production and enhanced 4-nitro-phenol reduction. Nanotechnology 32:495404. https://doi.org/10.1088/1361-6528/ac1dd4
Oshchepkov A, Bonnefont A, Maranzana G et al (2022) Direct borohydride fuel cells: a selected review of their reaction mechanisms, electrocatalysts and influence of operating parameters on their performance. Curr Opinion Electrochem 32:100883. https://doi.org/10.1016/j.coelec.2021.100883
Olu PY, Bonnefont A, Braesch G et al (2017) Influence of the concentration of borohydride towards hydrogen production and escape for borohydride oxidation reaction on Pt and Au electrodes – experimental and modelling insights. J Power Sources 375:300–309. https://doi.org/10.1016/j.jpowsour.2017.07.061
Minkina VG, Shabunya SI, Kalinin VI et al (2022) Hydrogen generation from hydrolysis of concentrated NaBH4 solutions under adiabatic conditions. Int J Hydrogen Energy 47:21772–21781. https://doi.org/10.1016/j.ijhydene.2022.05.006
Xu DY, Dai P, Liu XM et al (2008) Carbon-supported cobalt catalyst for hydrogen generation from alkaline sodium borohydride solution. J Power Sources 182:616–620. https://doi.org/10.1016/j.jpowsour.2008.04.018
Bennici S, Yu H, Obeid E et al (2011) Highly active heteropolyanions supported Co catalysts for fast hydrogen generation in NaBH4 hydrolysis. Int J Hydrogen Energy 36:7431–7442. https://doi.org/10.1016/j.ijhydene.2011.03.148
Marchionni A, Bevilacqua M, Filippi J et al (2015) High volume hydrogen production from the hydrolysis of sodium borohydride using a cobalt catalyst supported on a honeycomb matrix. J Power Sources 299:391–397. https://doi.org/10.1016/j.jpowsour.2015.09.006
Shi LM, Xie W, Jian ZY et al (2019) Graphene modified Co–B catalysts for rapid hydrogen production from NaBH4 hydrolysis. Int J Hydrogen Energy 44:17954–17962. https://doi.org/10.1016/j.ijhydene.2019.05.104
Zhang HM, Feng XL, Cheng LN et al (2019) Non-noble Co anchored on nanoporous graphene oxide, as an efficient and long-life catalyst for hydrogen generation from sodium borohydride. Coll Surf A 563:112–119. https://doi.org/10.1016/j.colsurfa.2018.12.002
Zhang X, Zhang Q, Xu B et al (2020) Efficient hydrogen generation from the NaBH4 hydrolysis by cobalt-based catalysts: positive roles of sulfur-containing salts. ACS Appl Mater Interfaces 12:9376–9386. https://doi.org/10.1021/acsami.9b22645
Xu JN, Du XX, Wei QL et al (2020) Efficient hydrolysis of sodium borohydride by Co-B supported on nitrogen-doped carbon. Chem Sel 5:6683–6690. https://doi.org/10.1002/slct.201904818
Altaf CT, Colak TO, Minkina VG et al (2022) Effect of titanium dioxide support for cobalt nanoparticle catalysts for hydrogen generation from sodium borohydride hydrolysis. Catal Lett. https://doi.org/10.1007/s10562-022-04215-9
Delmas J, Laversenne L, Rougeaux I et al (2011) Improved hydrogen storage capacity through hydrolysis of solid NaBH4 catalyzed with cobalt boride. Int J Hydrogen Energy 36:2145–2153. https://doi.org/10.1016/j.ijhydene.2010.11.036
Li F, Li Q, Kim H (2012) CoB/open-CNTs catalysts for hydrogen generation from alkaline NaBH4 solution. Chem Eng J 210:316–324. https://doi.org/10.1016/j.cej.2012.08.102
Baydaroglu F, Ozdemir E, Hasimoglu A (2014) An effective synthesis route for improving the catalytic activity of carbon-supported CoeB catalyst for hydrogen generation through hydrolysis of NaBH4. Int J Hydrogen Energy 39:1516–1522. https://doi.org/10.1016/j.ijhydene.2013.04.111
Li Y, Zhang Y, Cai Y et al (2015) Effects of composite catalyst Co2B/TiO2 on hydrolysis of NaBH4. Open Mater Sci J 9:60–63. https://doi.org/10.2174/1874088X01509010060
Singh PK, Das T (2017) Generation of hydrogen from NaBH4 solution using metal-boride (CoB, FeB, NiB) catalysts. Int J Hydrogen Energy 42:29360–29369. https://doi.org/10.1016/j.ijhydene.2017.10.030
Xu J, Du X, Wei Q et al (2020) Efficient Hydrolysis of Sodium Borohydride by Co-B Supported on Nitrogen-doped Carbon. ChemistrySelect 5:6683–6690
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shabunya, S.I., Minkina, V.G., Kalinin, V.I. et al. Heterogeneous kinetics of NaBH4 hydrolysis catalyzed by Co/TiO2. Reac Kinet Mech Cat 136, 1839–1857 (2023). https://doi.org/10.1007/s11144-023-02450-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11144-023-02450-8