Abstract
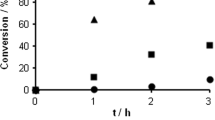
Hydrogen-transfer hydrodehalogenation of brominated anisoles, chlorinated anisoles, and bromochloroanisoles was carried out at 30–40 °C in a solution of NaOH in 2-propanol/methanol (99:1 V/V) in the presence of a Pd/SiO2 catalyst. The dehalogenation reactions proceeded stepwise to afford anisole, and 2-propanol served as a hydrogen source. In single-substrate reactions, 2,4,6-tribromoanisole (TBA) was dehalogenated more slowly than 2,4,6-trichloroanisole, whereas in a competitive reaction, 2,4,6-TBA was preferentially dehalogenated. The analysis of lower-halogenated intermediates revealed that the ortho halogen was less reactive than the para halogen, and the isomer selectivity of the dehalogenation reaction strongly depended on the halogen atom. The conversion of bromochloroanisoles (3-bromo-4-chloroanisole, 3-bromo-5-chloroanisole, and 2-bromo-4-chloroanisole) to anisole occurred exclusively via the corresponding chloroanisoles. The difference in dehalogenation behavior between the brominated and chlorinated anisoles is discussed on the basis of carbon–halogen bond dissociation energies, steric effects, and substrate adsorption ability.







Similar content being viewed by others
References
UNEP, Stockholm convention, The 12 initial POPs. http://chm.pops.int/TheConvention/ThePOPs/The12InitialPOPs/tabid/296/Default.aspx. Accessed Feb 2019
UNEP, Stockholm convention, The new POPs, http://chm.pops.int/TheConvention/ThePOPs/TheNewPOPs/tabid/2511/Default.aspx. Accessed Feb 2019
Hitchman ML, Spackman RA, Ross NC, Agra C (1995) Disposal methods for chlorinated aromatic wastes. Chem Soc Rev 24:423–430
Alonso F, Beletskaya IP, Yus M (2002) Metal-mediated reductive hydrodehalogenation of organic halides. Chem Rev 102:4009–4091
Urbano FJ, Marinas JM (2001) Hydrogenolysis of organohalogen compounds over palladium supported catalysts. J Mol Catal A 173:329–345
Keane MA (2011) Supported transition metal catalysts for hydrodechlorination reactions. ChemCatChem 3:800–821
Johnstone RAW, Wilby AH, Entwistle ID (1985) Heterogeneous catalytic transfer hydrogenation and its relation to other methods for reduction of organic compounds. Chem Rev 85:129–170
Ukisu Y, Miyadera T (1997) Hydrogen-transfer hydrodehalogenation of aromatic halides with alcohols in the presence of noble metal catalysts. J Mol Catal A 125:135–142
Ukisu Y, Miyadera T (2003) Hydrogen-transfer hydrodechlorination of polychlorinated dibenzo-p-dioxins and dibenzofurans catalyzed by supported palladium catalysts. Appl Catal B 40:141–149
Ukisu Y (2008) Highly enhanced hydrogen-transfer hydrodechlorination and hydrogenation reactions in alkaline 2-propanol/methanol over supported palladium catalysts. Appl Catal A 349:229–232
Ukisu Y, Iimura S, Uchida R (1996) Catalytic dechlorination of polychlorinated biphenyls with carbon-supported noble metal catalysts under mild conditions. Chemosphere 33:1523–1530
Ukisu Y, Miyadera T (2004) Dechlorination of dioxins with supported palladium catalysts in 2-propanol solution. Appl Catal A 271:165–170
Ukisu Y (2008) Complete dechlorination of DDT and its metabolites in an alcohol mixture using NaOH and Pd/C catalyst. J Hazard Mater 152:287–292
Ukisu Y, Miyadera T (2005) Dechlorination of hexachlorocyclohexanes with alkaline 2-propanol and a palladium Catalyst. J Hazard Mater 122:1–6
Ukisu Y (2015) Complete catalytic debromination of polybrominated diphenyl ethers over a silica-supported palladium nanoparticle catalyst. Environ Chem Lett 13:211–216
Ukisu Y (2017) Complete catalytic debromination of hexabromocyclododecane using a silica-supported palladium catalyst in alkaline 2-propanol. Chemosphere 179:179–184
Buser HR, Zanier C, Tanner H (1982) Identification of 2,4,6-trichloroanisole as a potent compound causing cork taint in wine. J Agric Food Chem 30:359–362
Chatonnet P, Bonnet S, Boutou S, Labadie MD (2004) Identification and responsibility of 2,4,6-tribromoanisole in musty, corked orders in wine. J Agric Food Chem 52:1255–1262
Jönsson S, Hagberg J, van Bavel B (2008) Determination of 2,4,6-trichloroanisole and 2,4,6-tribromoanisole in wine using microextraction in packed syringe and gas chromatography-mass spectrometry. J Agric Food Chem 56:4962–4967
Gunschera J, Fuhrmann F, Salthammer T, Schulze A, Uhde E (2004) Formation and emission of chloroanisoles as indoor pollutants. Environ Sci Pollut Res 11:147–151
Ukisu Y (2015) Preparation of silica-supported Pd nanoparticle catalysts by an alcohol reduction method and evaluation of their catalytic properties in the dechlorination of an aromatic chloride. React Kinet Mech Cat 114:385–394
Shin EJ, Keane MA (1998) Gas phase catalytic hydrodechlorination of chlorophenols using a supported nickel catalyst. Appl Catal B 18:241–250
Keane MA, Pina G, Tavoularis G (2004) The catalytic hydrodechlorination of mono-, di- and trichlorobenzenes over supported nickel. Appl Catal B 48:275–286
Murena F, Schioppa E (2000) Kinetic analysis of catalytic hydrodechlorination process of polychlorinated biphenyls (PCBs). Appl Catal B 27:257–267
Zhuang Y, Ahn S, Seyfferth AL, Masue-Slowey Y, Fendorf S, Luthy RG (2011) Dehalogenation of polybrominated diphenyl ethers and polychlorinated biphenyl by bimetallic, impregnated, and nanoscale zerovalent iron. Environ Sci Technol 45:4896–4903
Gómez-Quero S, Díaz E, Cárdenas-Lizana F, Keane MA (2010) Solvent effects in the catalytic hydrotreatment of haloaromatics over Pd/Al2O3 in water + organic mixtures. Chem Eng Sci 65:3786–3797
Murthy KV, Patterson PM, Keane MA (2005) C-X bond reactivity in the catalytic hydrodehalogenation of haloarenes over unsupported and silica supported Ni. J Mol Catal A 225:149–160
Keane MA (2004) Hydrodehalogenation of haloarenes over silica supported Pd and Ni: a consideration of catalytic activity/selectivity and haloarene reactivity. Appl Catal A 271:109–118
Tavoularis G, Keane MA (1999) Gas phase catalytic dehydrochlorination and hydrodechlorination of aliphatic and aromatic systems. J Mol Catal A 142:187–199
Park C, Menini C, Valverde JL, Keane MA (2002) Carbon-chlorine and carbon-bromine bond cleavage in the catalytic hydrodehalogenation of halogenated aromatics. J Catal 211:451–463
Oxley JD, Mdleleni MM, Suslick KS (2004) Hydrodehalogenation with sonochemically prepared Mo2C and W2C. Catal Today 88:139–151
Chen ECM, Albyn K, Dussack L, Wentworth WE (1989) Determination of bond dissociation energies from dissociative thermal electron attachment. J Phys Chem 93:6827–6832
Watanabe T, Wang ZY, Takahashi O, Morihashi K, Kikuchi O (2004) Calculation of systematic set of bond dissociation enthalpies of polyhalogenated benzenes. J Mol Struct Theochem 682:63–72
Aramendía MA, Boráu V, García IM, Jiménez C, Marinas A, Marinas JM, Urbano FJ (2003) Liquid-phase hydrodehalogenation of substituted chlorobenzenes over palladium supported catalysts. Appl Catal B 43:71–79
Mackenzie K, Frenzel H, Kopinke FD (2006) Hydrodehalogenation of halogenated hydrocarbons in water with Pd catalysts: reaction rates and surface competition. Appl Catal B 63:161–167
Marques CA, Selva M, Tundo P (1993) Facile Hydrodehalogenation with hydrogen and Pd/C catalyst under multiphase conditions. J Org Chem 58:5256–5260
Aramendía MA, Boráu V, García IM, Jiménez C, Marinas JM, Marinas A, Urbano FJ (2000) Hydrodehalogenation of aryl halides by hydrogen gas and hydrogen transfer in the presence of palladium catalysts. Stud Surf Sci Catal 130:2003–2008
Štěpnička P, Semler M, Demel J, Zukal A, Čejka J (2011) Reductive dehalogenation of aryl halides over palladium catalysts deposited on SBA-15 type molecular sieve modified with amine donor groups. J Mol Catal A 341:97–102
Huang Y, Liu S, Lin Z, Li W, Li X, Cao R (2012) Facile synthesis of palladium nanoparticles encapsulated in amine-functionalized mesoporous metal-organic frameworks and catalytic for dehalogenation of aryl chlorides. J Catal 292:111–117
Kara BY, Yazici M, Kilbas B, Goksu H (2016) A practical and highly efficient reductive dehalogenation of aryl halides using heterogeneous Pd/Al(OH) nanoparticles and sodium borohydride. Tetrahedron 72:5898–5902
Wiener H, Blum J, Sasson Y (1991) Transfer hydrogenolysis of aryl halides and hydrogen acceptors by formate salts in the presence of Pd/C catalyst. J Org Chem 56:6145–6148
Moon J, Lee S (2009) Palladium catalyzed-dehalogenation of aryl chlorides and bromides using phosphite ligands. J Organomet Chem 694:473–477
Acknowledgements
The author gratefully acknowledges financial support from the National Institute of Advanced Industrial Science and Technology, Japan.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ukisu, Y. Hydrogen-transfer hydrodehalogenation of aromatic halides with a silica-supported palladium catalyst in alkaline 2-propanol: comparison between brominated and chlorinated anisoles. Reac Kinet Mech Cat 128, 41–52 (2019). https://doi.org/10.1007/s11144-019-01632-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11144-019-01632-7