Abstract
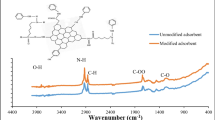
A series of Cu–alginate and Cu–Pd–alginate catalysts were prepared by the ion exchange of Na–alginate, and their structures were well characterized by FT-IR, XPS and ICP-AES measurements. Their catalytic activities for phenol hydroxylation in the fixed-bed flow reactor were investigated. The effects of different reaction conditions, such as molar ratio of phenol/H2O2, reaction temperature, weight hourly space velocity, solution and weight ratio of water/phenol were evaluated. Under the optimum reaction conditions, 17.1 % phenol conversion was obtained with selectivities to catechol and hydroquinone being 67.7 and 31.4 %, respectively. The 16.0–18.0 % phenol conversion and selectivities to HQ 30.0–32.0 %, CAT 66.0–68.0 % and BQ 0.7–2.0 % of the long-run catalytic test confirmed the excellent stable catalytic activity of the catalyst.






Similar content being viewed by others
References
Jian LJ, Chen C, Lan F, Deng SJ, Xiao WM, Zhang N (2011) Catalytic activity of unsaturated coordinated Cu-MOF to the hydroxylation of phenol. Solid State Sci 13:1127–1131
Mugo JN, Mapolie SF, Wyk JL (2010) Cu (II) and Ni (II) complexes based on monofunctional and dendrimeric pyrrole-imine ligands: applications in catalytic liquid phase hydroxylation of phenol. Inorg Chim Acta 363:2643–2651
Wu G, Tan XY, Li GY, Hu CW (2010) Effect of preparation method on the physical and catalytic property of nanocrystalline Fe2O3. J Alloys Comp 504:371–376
Ray S, Mapolie SF, Darkwa J (2007) Catalytic hydroxylation of phenol using immobilized late transition metal salicylaldimine complexes. J Mol Catal A Chem 267:143–148
Zhang GY, Long JL, Wang XX, Zhang ZZ, Dai WX, Liu P, Li ZH, Wu L, Fu XZ (2010) Catalytic Role of Cu Sites of Cu/MCM-41 in phenol hydroxylation. Langmuir 26:1362–1371
Abbo HS, Titinchi SJJ (2010) Synthesis and catalytic activity of Cu (II), Fe(III) and Bi(III) complexes of thio-schiff base encapsulated in zeolite-Y for hydroxylation of phenol. Top Catal 53:254–264
Mirica LM, Vance M, Rudd DJ, Hedman B, Hodgson KO, Solomon EI, Stack TDP (2005) Tyrosinase reactivity in a model complex: an alternative hydroxylation mechanism. Science 308:1890–1892
Palavicini S, Granata A, Monzani E, Casella L (2005) Hydroxylation of phenolic compounds by a peroxodicopper (II) complex: further insight into the mechanism of tyrosinase. J Am Ceram Soc 127:18031–18036
Yang JS, Xie YJ, Wen H (2011) Research progress on chemical modification of alginate: a review. Carbohydr Polym 84:33–39
Stokke BT, Smidsrød O, Bruheim P, Skjak-Bræk G (1991) Distribution of uronate residues in alginate chains in relation to alginate gelling properties. Macromolecules 24:4637–4645
Torres E, Mata YN, Blázquez ML, Munoz JA, González F, Ballester A (2005) Gold and silver uptake and nanoprecipitation on calcium alginate beads. Langmuir 21:7951–7958
Grace M, Chand N, Bajpai SK (2009) Copper alginate-cotton cellulose (CACC) fibers with excellent antibacterial properties. J Eng Fiber Fabr 4:24–35
Kong HJ, Kaigler D, Kim K, Mooney DJ (2004) Controlling rigidity and degradation of alginate hydrogels via molecular weight distribution. Biomacromolecules 5:1720–1727
Gomez CG, Rinaudo M, Villar MA (2007) Oxidation of sodium alginate and characterization of the oxidized derivatives. Carbohydr Polym 67:296–304
Wong TW (2011) Alginate graft copolymers and alginate-co-excipient physical mixture in oral drug delivery. J Pharm Pharmacol 63:1497–1512
Laurienzo P, Malinconico M, Motta A, Vicinanza A (2005) Synthesis and characterization of a novel alginate–poly (ethylene glycol) graft copolymer. Carbohydr Polym 62:274–282
Alban S, Schauerte A, Franz G (2002) Anticoagulant sulfated polysaccharides: part I. Synthesis and structure- activity relationships of new pullulan sulfates. Carbohydr Polym 47:267–276
Park JN, Wang J, Choi KY, Dong WY, Hong SI, Lee CW (2006) Hydroxylation of phenol with H2O2 over Fe2+ and/or Co2+ ion-exchanged NaY catalyst in the fixed-bed flow reactor. J Mol Catal A Chem 247:73–79
Liu H, Lu GZ, Guo YL, Guo Y (2005) Synthesis of TS-1 using amorphous SiO2 and its catalytic properties for hydroxylation of phenol in fixed-bed reactor. Appl Catal A 293:153–161
Tendulkar SB, Tambe SS, Chandra I, Rao PV, Naik RV, Kulkarni BD (1998) Hydroxylation of phenol to dihydroxybenzenes: development of artificial neural-network-based process identification and model predictive control strategies for a pilot plant scale reactor. Ind Eng Chem Res 37:2081–2085
Liu H, Lu GZ, Guo YL, Guo Y, Wang JS (2006) Chemical kinetics of hydroxylation of phenol catalyzed by TS-1/diatomite in fixed-bed reactor. Chem Eng J 116:179–186
Shams K, Najari S (2011) Dynamics of intraparticle desorption and chemical reaction in fixed-beds using inert core spherical particles. Chem Eng J 172:500–506
Iliuta I, Thyrion FC, Muntean O, Giot M (1996) Residence time distribution of the liquid in gas-liquid cocurrent upflow fixed-bed reactors. Chem Eng Sci 50:4579–4593
Bucalá V, Borio DO, Porras J (1997) Thermal regimes in cocurrently cooled fixed-bed reactors for parallel reactions: application to practical design problems. Chem Eng Sci 52:1577–1587
Dixon AG, Walls G, Stanness H, Nijemeisland M, Stitt EH (2012) Experimental validation of high Reynolds number CFD simulations of heat transfer in a pilot-scale fixed bed tube. Chem Eng J 200–202:344–356
Shi FW, Chen YG, Sun LP, Zhang L, Hu JL (2012) Hydroxylation of phenol catalyzed by different forms of Cu-alginate with hydrogen peroxide as an oxidant. Catal Commun 25:102–105
Shi FW, Zheng JL, Xu K, Zhang L, Hu JL (2012) Synthesis of binary Cu–Pd–alginates dry bead and its high catalytic activity for hydroxylation of phenol. Catal Commun 28:23–26
Papageorgiou SK, Kouvelos EP, Favvas EP, Sapalidis AA, Romanos GE, Katsaros FK (2010) Metal–carboxylate interactions in metal–alginate complexes studied with FTIR spectroscopy. Carbohydr Res 345:469–473
Jiang YJ, Gao QM (2007) Preparation of Cu2+/+–VSB-5 and their catalytic properties on hydroxylation of phenol. Mater Lett 61:2212–2216
Rodrigues A, Costa P, Méthivier C, Dzwigaj S (2011) Controlled preparation of CoPdSiBEA zeolite catalysts for selective catalytic reduction of NO with methane and their characterisation by XRD, DR UV–Vis, TPR, XPS. Catal Today 176:72–76
Clerici MG, Bellussi G, Romano U (1991) Synthesis of propylene oxide from propylene and hydrogen peroxide catalyzed by titanium silicalite. J Catal 129:159–167
Yadav GD, Pujari AA (2000) Epoxidation of styrene to styrene oxide: synergism of heteropoly acid and phase-transfer catalyst under ishii-venturello mechanism. Org Process Res Dev 4:88–93
Acknowledgments
This work was supported by the Natural Science Fund Council of China (21203013) and Undergraduate Scientific and Technological Innovation Project of Changchun University of Technology (2014cxcy156). The authors gratefully acknowledge the Centre of Analysis and Testing of Northeast Normal University and Changchun University of Technology for all of the support that was provided.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shi, F., Luo, Y., Wang, W. et al. Hydroxylation of phenol with H2O2 over binary Cu–Pd–alginate catalyst in the fixed-bed flow reactor. Reac Kinet Mech Cat 115, 187–199 (2015). https://doi.org/10.1007/s11144-015-0836-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11144-015-0836-1