Abstract
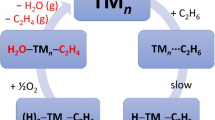
The global optimization of neutral and +1 charged Pt n (n = 2–6) clusters were conducted at the B3LYP/LANL2DZ(f) level of theory. The lowest-energy structures of neutral Pt n (n = 3–6) clusters adopt an equilateral triangle, a tetrahedron, an edge-capped tetrahedron, and a prism structure, respectively. The insertion of Pt n (n = 2–6) into one of the central C–H bonds in propane, Pt n + C3H8 → H–Pt n –CH(CH3)2, was studied subsequently for selected low-energy Pt cluster structures. We found that the Pt n global minimum does not necessary make the most contribution to the catalyzed dehydrogenation of propane. For example, the prism structure in the septet electronic state is the global minimum of Pt6, yet the 5Pt6 edge-capped pyramid structure makes significantly more contribution to the activation of C–H bonds in propane. In sum, the energy barrier for the Pt2 + C3H8 → H–Pt2–CH(CH3)2 reaction is 33 kJ/mol; Pt3–6 have significantly lower barriers of 4–16 kJ/mol. The +1 charged Pt + n (n = 2–6) clusters are much more active towards propane than their neutral counterparts because of the large amount of energy (30–140 kJ/mol) released in the formation of the Pt + n –C3H8 reactant complex. A similar charge effect is also observed for the larger Pt10 + cluster.










Similar content being viewed by others

References
Ferguson GA, Mehmood F, Rankin RB, Greeley JP, Vajda S, Curtiss LA (2012) Exploring computational design of size-specific subnanometer clusters catalysts. Top Catal 55:353–365
Cui Q, Musaev DG, Morokuma K (1998) Molecular orbital study of H2 and CH4 activation on small metal clusters. I. Pt, Pd, Pt2, and Pd2. J Chem Phys 108:8418–8428
Cui Q, Musaev DG, Morokuma K (1998) Molecular orbital study of H2 and CH4 activation on small metal clusters. 2. Pd3 and Pt3. J Phys Chem A 102:6373–6384
Argo AM, Odzak JF, Gates BC (2003) Role of cluster size in catalysis: spectroscopic investigation of γ-Al2O3-supported Ir4 and Ir6 during ethene hydrogenation. J Am Chem Soc 125:7107–7115
Valero MC, Raybaud P, Sautet P (2007) Interplay between molecular adsorption and metal-support interaction for small supported metal clusters: CO and C2H4 adsorption on Pd4/γ-Al2O3. J Catal 247:339–355
Xiao L, Wang LC (2007) Methane activation on Pt and Pt4: a density functional theory study. J Phys Chem B 111:1657–1663
Kulkarni A, Lobo-Lapidus RJ, Gates BC (2010) Metal clusters on supports: synthesis, structure, reactivity, and catalytic properties. Chem Commun (Cambridge, UK) 46:5997–6015
Xu Z, Xiao FS, Purnell SK, Alexeev OSK, Deutsch SE, Gates BC (1994) Size-dependent catalytic activity of supported metal clusters. Nature 372:346–348
Vajda S, Pellin MJ, Greeley JP, Marshall CL, Curtiss LA, Ballentine GA, Elam JW, Catillon-Mucherie S, Redfern PC, Mehmood F, Zapol P (2009) Subnanometre platinum clusters as highly active and selective catalysts for the oxidative dehydrogenation of propane. Nat Mater 8:213–216
Saenz LR, Balbuena PB, Seminario JM (2006) Platinum testbeds: interaction with oxygen. J Phys Chem A 110:11968–11974
Becke AD (1993) Density-functional thermochemistry. III. The role of exact exchange. J Chem Phys 98:5648–5652
Perdew JP, Chevary JA, Vosko SH, Jackson KA, Pederson MR, Singh DJ, Fiolhais C (1992) Atoms, molecules, solids, and surfaces: applications of the generalized gradient approximation for exchange and correlation. Phys Rev B 46:6671–6687
Perdew JP, Wang Y (1992) Accurate and simple analytic representation of the electron-gas correlation energy. Phys Rev B 45:13244–13249
Hay PJ, Wadt WR (1985) Ab initio effective core potentials for molecular calculations. Potentials for K to Au including the outermost core orbitals. J Chem Phys 82:299–310
Xiao L, Wang LC (2004) Structures of platinum clusters: planar or spherical? J Phys Chem A 108:8605–8614
Sebetci A (2009) Does spin–orbit coupling effect favor planar structures for small platinum clusters? Phys Chem Chem Phys 11:921–925
Lee C, Yang W, Parr RG (1988) Development of the Colle–Salvetti correlation-energy formula into a functional of the electron density. Phys Rev B 37:785–789
Stephens PJ, Devlin FJ, Chabalowski CF, Frisch MJ (1994) Ab initio calculation of vibrational absorption and circular dichroism spectra using density functional force fields. J Phys Chem 98:11623–11627
Ross RB, Powers JM, Atashroo T, Ermler WC, LaJohn LA, Christiansen PA (1990) Ab initio relativistic effective potentials with spin-orbit operators. IV. Cs through Rn. J Chem Phys 93:6654–6670
Hariharan PC, Pople JA (1973) The influence of polarization functions on molecular orbital hydrogenation energies. Theor Chim Acta 28:213–222
Hehre WJ, Ditchfield R, Pople JA (1972) Self-consistent molecular-orbital methods.12. Further extensions of Gaussian-type basis sets for use in molecular-orbital studies of organic-molecules. J Chem Phys 56:2257–2261
Frisch MJ et al (2010) Gaussian 09, Revision B.01. Gaussian Inc., Wallingford CT
Bilodeau RC, Scheer M, Haugen HK, Brooks RL (2000) Near-threshold laser spectroscopy of iridium and platinum negative ions: electron affinities and the threshold law. Phys Rev A 61. doi:10.1103/PhysRevA.61.012505
Ho J, Polak ML, Ervin KM, Lineberger WC (1993) Photoelectron spectroscopy of nickel group dimers: Ni2, Pd2, and Pt2. J Chem Phys 99:8542–8551
Jakubek ZJ, Simard B (2000) Rydberg series and the first ionization potential of platinum and yttrium atoms. J Phys B 33:1827–1841
Taylor S, Lemire GW, Hamrick YM, Fu Z, Morese MD (1988) Resonant two-photon ionization spectroscopy of jet-cooled Pt2. J Chem Phys 89:5517–5523
McCarthy MC, Field RW, Engleman R, Bernath PF (1993) Laser and Fourier transform spectroscopy of PtH and PtD. J Mol Spectrosc 158:208–236
Citir M, Metz RB, Belau L, Ahmed M (2008) Direct determination of the ionization energies of PtC, PtO, and PtO2 with VUV radiation. J Phys Chem A 112:9584–9590
Airola MB, Morse MD (2002) Rotationally resolved spectroscopy of Pt2. J Chem Phys 116:1313–1317
Huber KP, Herzberg G (1979) Molecular spectra and molecular structure. IV. Constants of diatomic molecules. Van Nostrand Reinhold, New York
Steimle TC, Jung KY, Li BZ (1995) The permanent electric-dipole moment of PtO, PtS, PtN, and PtC. J Chem Phys 103:1767–1772
Harvey JN (2006) On the accuracy of density functional theory in transition metal chemistry. Annu Rep Prog Chem C 102:203–226
Majumdar D, Dai DG, Balasubramanian K (2000) Theoretical study of the electronic states of platinum trimer (Pt3). J Chem Phys 113:7919–7927
Zhang WQ, Wang LC (2011) The effect of cluster thickness on the adsorption of CH4 on Pdn. Comput Theor Chem 963:236–244
Adlhart C, Uggerud E (2007) Mechanisms for the dehydrogenation of alkanes on platinum: insights gained from the reactivity of gaseous cluster cations, Pt + n , n = 1–21. Chem Eur J 13:6883–6890
Argo AM, Odzak JF, Lai FS, Gates BC (2002) Observation of ligand effects during alkene hydrogenation catalysed by supported metal clusters. Nature 415:623–626
Acknowledgments
Ge thanks the Central Washington University for the startup and SEED grant. Ge also thanks A. L. Diaz and B.-L. Chen for helpful discussions.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shore, T.C., Mith, D., DePrekel, D. et al. A B3LYP study on the C–H activation in propane by neutral and +1 charged low-energy platinum clusters with 2–6 atoms. Reac Kinet Mech Cat 109, 315–333 (2013). https://doi.org/10.1007/s11144-013-0572-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11144-013-0572-3