Abstract
Introduction
Generic or condition-specific Patient-reported Outcome Measures (PROMs) are used to measure physical, mental, and social aspects of health to promote patient-centered care. This scoping review aims to identify and summarize generic and condition-specific PRO domains and PROMs that have been assessed and used in liver transplant (LT) candidates and recipients.
Methods
We searched Medline, Embase, Cochrane Database of Systematic Reviews and Register of Trials, PsychInfo, and CINAHL from inception to 08/26/2020. Included studies addressed a PRO or PROM in LT candidates or recipients.
Results
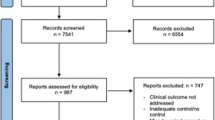
After screening, 341 studies yielded 189 unique PRO domains. Mental health domains (depression, anxiety, and guilt) were most frequently assessed, followed by domains of physical and social health. Fifty-one generic and three condition-specific unique PROMs were identified, with only 13% (n = 45) of studies including condition-specific tools.
Discussion
The most frequent PROMs were the SF-36, Nottingham Health Profile, Hospital Anxiety and Depression Scale, followed by the Liver Disease Quality of Life (LDQoL). Very few studies used transplant-specific PROMs, which may partly be related to the scarcity of LT-specific instruments. We will use these results in future qualitative research to identify PROs and PROMs to build an electronic PROM toolkit to facilitate patient-centered LT care.



Similar content being viewed by others
Data availability
The data underlying this article, including deidentifed participant data and a data dictionary, can be shared upon request to the corresponding author.
Abbreviations
- BADI:
-
Beck anxiety and depression inventory
- CENTRAL:
-
Cochrane central register of controlled trials
- CINAHL:
-
Cumulative index to nursing and allied health literature
- CLDQ:
-
Chronic liver disease questionnaire
- ePROM:
-
Electronic patient-reported outcome measures
- FDA:
-
Food and drug administration
- HADS:
-
Hospital anxiety and depression scale
- HRQoL:
-
Health-related quality of life
- LDQoL:
-
Liver disease quality of life questionnaire
- LT:
-
Liver transplant
- NHP:
-
Nottingham health profile
- pLTQ:
-
Post-liver transplant quality of life
- PRO:
-
Patient-reported outcomes
- PROM:
-
Patient-reported outcome measures
- PROMIS:
-
Patient-reported outcomes measurement information system
- PROMIS CAT:
-
Patient-reported outcomes measurement information system computer adaptive testing
- PSQI:
-
Pittsburgh sleep quality index
- SF-36:
-
36-Item short-form health survey
- WHQOL:
-
World health organization quality of life
References
Aaronson, N., Alonso, J., Burnam, A., Lohr, K. N., Patrick, D. L., Perrin, E., & Stein, R. E. (2002). Assessing health status and quality-of-life instruments: attributes and review criteria. Quality of Life Research, 11(3), 193–205.
Mucsi, I. (2008). Health-related quality of life in chronic kidney disease patients. Primary Psychiatry., 15(1), 46–51.
Juenger, J. (2002). Health related quality of life in patients with congestive heart failure: Comparison with other chronic diseases and relation to functional variables. Heart, 87(3), 235–241.
Kalantar-Zadeh, K., & Unruh, M. (2005). Health Related Quality of Life in Patients with Chronic Kidney Disease. International Urology and Nephrology, 37(2), 367–378.
Orr, J. G., Homer, T., Ternent, L., Newton, J., McNeil, C. J., Hudson, M., & Jones, D. E. J. (2014). Health related quality of life in people with advanced chronic liver disease. Journal of Hepatology, 61(5), 1158–1165.
Cella, D., Yount, S., Rothrock, N., Gershon, R., Cook, K., Reeve, B., Ader, D., Fries, J. F., Bruce, B., & Rose, M. (2007). The patient-reported outcomes measurement information system (PROMIS): Progress of an NIH roadmap cooperative group during its first two years. Medical Care, 45(5 Suppl 1), S3–S11.
Riley, W. T., Rothrock, N., Bruce, B., Christodolou, C., Cook, K., Hahn, E. A., & Cella, D. (2010). Patient-reported outcomes measurement information system (PROMIS) domain names and definitions revisions: further evaluation of content validity in IRT-derived item banks. Quality of Life Research, 19(9), 1311–1321.
Shahabeddin Parizi, A., Krabbe, P. F. M., Buskens, E., Bakker, S. J. L., & Vermeulen, K. M. (2019). A scoping review of key health items in self-report instruments used among solid organ transplant recipients. Patient, 2(12), 171–181.
Kensinger, C. D. F. I., & O’Dell, H. W. (2016). Patient-reported outcomes in liver transplant recipients with hepatocellular carcinoma. Clinical Transplantation, 9(30), 1036–1045.
Tang, E., Bansal, A., Novák, M., & Mucsi, I. (2017). Patient-reported outcomes in patients with chronic kidney disease and kidney transplant-Part 1. Frontiers in Medicine (Lausanne), 4, 254.
Anugwom, C. M., Parekh, J. R., Hwang, C., MacConmara, M., Lee, W. M., & Leventhal, T. M. (2020). Comparison of clinical outcomes of induction regimens in patients undergoing liver transplantation for acute liver failure. Liver Transplantaion, 27(1), 27–33.
Pelgur, H., Atak, N., & Kose, K. (2009). Anxiety and depression levels of patients undergoing liver transplantation and their need for training. Transplantation Proceedings, 41, 1743–1748.
Miller, L. R., Paulson, D., Eshelman, A., Bugenski, M., Brown, K. A., Moonka, D., & Abouljoud, M. (2013). Mental health affects the quality of life and recovery after liver transplantation. Liver Transplantation, 19, 1272–1278.
Drent, G., De Geest, S., Dobbels, F., Kleibeuker, J. H., & Haagsma, E. B. (2009). Symptom experience, nonadherence and quality of life in adult liver transplant recipients. The Netherlands Journal of Medicine, 67(5), 161–168.
Bababekov, Y. J., Hung, Y. C., Chang, D. C., Rickert, C. G., Adler, J. T., Bethea, E., Pomfret, E. A., Pomposelli, J. J., & Yeh, H. (2020). Do social determinants define “Too Sick” to transplant in patients with end-stage liver disease? Transplantation, 104(2), 280–284.
Dew, M. A., Butt, Z., Humar, A., & DiMartini, A. F. (2017). Long-Term Medical and Psychosocial Outcomes in Living Liver Donors. American Journal of Transplantation, 17(4), 880–892.
DiMartini, A., Dew, M. A., Liu, Q., Simpson, M. A., Ladner, D. P., Smith, A. R., Zee, J., Abbey, S., Gillespie, B. W., Weinrieb, R., & Mandell, M. S. (2017). Social and Financial outcomes of living liver donation: A prospective investigation within the adult-to-adult living donor liver transplantation cohort study 2 (A2ALL-2). American Journal of Transplantation, 17(4), 1081–1096.
Goetzmann, L., Klaghofer, R., Wagner-Huber, R., Halter, J., Boehler, A., Muellhaupt, B., Schanz, U., & Buddeberg, C. (2007). Psychosocial vulnerability predicts psychosocial outcome after an organ transplant: Results of a prospective study with lung, liver, and bone-marrow patients. Journal of Psychosomatic Research, 62(1), 93–100.
Grover, S., & Sarkar, S. (2012). Liver transplant—psychiatric and psychosocial aspects. Journal of Clinical and Experimental Hepatology, 2(4), 382–392.
Mohamed, S., Sabki, Z. A., & Zainal, N. Z. (2014). Depression and psychosocial correlates of liver transplant candidates: A systematic review. Asia-Pacific Psychiatry, 6(4), 447–453.
Fredrik Åberg, M. G., Karlsen, T. H., Ericzon, B.-G., Foss, A., Rasmussen, A., Bennet, W., Olausson, M., Line, P.-D., Nordin, A., & Bergquist, A. (2014). Differences in long-term survival among liver transplant recipients and the general population: A population-based nordic study. Hepatology, 61(2), 668–677.
Basch, E. (2017). Patient-reported outcomes—harnessing patients’ voices to improve clinical care. New England Journal of Medicine, 376, 105–108.
Basch, E. J. X., & Heller, G. (2009). Adverse symptom event reporting by patients vs clinicians: Relationships with clinical outcomes. Journal of the National Cancer Institute, 101, 1624–1632.
Rodrigue, J. N. D., Hanto, D., Reed, A., & Curry, M. (2013). Patient-reported immunosuppression nonadherence 6 to 24 months after liver transplant: Association with pretransplant psychosocial factors and perceptions of health status change. Progress in Transplantation, 23, 319–328.
Barber, K., Blackwell, J., Collett, D., Neuberger, J., UK Transplant Liver Advisory Group. (2007). Life expectancy of adult liver allograft recipients in the UK. Gut, 56, 279–282.
Isa, F., Turner, G. M., Kaur, G., Kyte, D., Slade, A., Pankhurst, T., Kerecuk, L., Keeley, T., Ferguson, J., & Calvert, M. (2018). Patient-reported outcome measures used in patients with primary sclerosing cholangitis: A systematic review. Health and Quality of Life Outcomes. https://doi.org/10.1186/s12955-018-0951-6
Prita Daliya, E. H. G., Lobo, D. N., & Parsons, S. L. (2019). A systematic review of patient reported outcome measures (PROMs) and quality of life reporting in patients undergoing laparoscopic cholecystectomy. HepatoBiliary Surgery and Nutrition., 8(3), 228–245.
Laith Alrubaiy, H. A. H., & Williams, J. G. (2014). Assessing patient reported outcome measures: A practical guide for gastroenterologists. United European Gastroenterology Journal, 6(2), 463–470.
Younossi, Z. M. G. G., & Kiwi, M. (1995). Development of a disease specific questionnaire to measure health related quality of life in patients with chronic liver disease. Gut, 45, 295–300.
Tapper, E. B., Kanwal, F., Asrani, S. K., Ho, C., Ovchinsky, N., Poterucha, J., Flores, A., Smith, J. E., Ankoma-Sey, V., Luxon, B., & Volk, M. L. (2018). Patient-reported outcomes in cirrhosis: A scoping review of the literature. Hepatology, 67(6), 2375–2383.
Stine, J. G., Stukenborg, G. J., Wang, J., Adkins, A., Niccum, B., Zimmet, A., & Argo, C. K. (2020). Liver transplant candidates have impaired quality of life across health domains as assessed by computerized testing. Annals of Hepatology, 19(1), 62–68.
Aiyegbusi, O. L., Nair, D., Peipert, J. D., Schick-Makaroff, K., & Mucsi, I. (2021). A narrative review of current evidence supporting the implementation of electronic patient-reported outcome measures in the management of chronic diseases. Therapeutic advances in chronic disease, 12, 20406223211015960.
Holzner, B., Giesinger, J. M., Pinggera, J., Zugal, S., Schöpf, F., Oberguggenberger, A. S., Gamper, E. M., Zabernigg, A., Weber, B., & Rumpold, G. (2012). The Computer-based Health Evaluation Software (CHES): A software for electronic patient-reported outcome monitoring. BMC Medical Informatics and Decision Making., 12, 126.
Papuga, M. O. D. C., McIntyre, A., Mitten, D., Kates, S., & Baumhauer, J. F. (2017). Large-scale clinical implementation of PROMIS computer adaptive testing with direct incorporation into the electronic medical record. Health Systems, 64, 1–12.
Aiyegbusi, O. L., Kyte, D., Cockwell, P., Marshall, T., Keeley, T., Gheorghe, A., & Calvert, M. (2016). Measurement properties of patient-reported outcome measures (PROMs) used in adult patients with chronic kidney disease: A systematic review protocol. British Medical Journal Open, 10(6), e012014.
PROM Group AM. (2010). Carolina Casañas i Comabella, Monica Hadi, Elizabeth Gibbons, Ray Fitzpatrick Nia Roberts. PROM Group Construct & Instrument Type Filters.
Cleemput, I. D. F. (2007). Measuring patient-reported outcomes in solid organ transplant recipients. PharmacoEconomics, 4(25), 269–286.
Gralnek, I. M., Hays, R. D., Kilbourne, A., Rosen, H. R., Keeffe, E. B., Artinian, L., Kim, S., Lazarovici, D., Jensen, D. M., Busuttil, R. W., & Martin, P. (2000). Development and evaluation of the Liver Disease Quality of Life instrument in persons with advanced, chronic liver disease-the LDQOL 1.0. The American journal of gastroenterology., 95, 3552–3565.
Saab, S. N. V., & Landaverde, C. (2011). Development of a disease-specific questionnaire to measure health-related quality of life in liver transplant recipients. Liver Transplantation, 5(17), 567–579.
Bajaj, J. S., Thacker, L. R., Wade, J. B., Sanyal, A. J., Heuman, D. M., Sterling, R. K., Gibson, D. P., Stravitz, R. T., Puri, P., Fuchs, M., Luketic, V., Noble, N., White, M., Bell, D., & Revicki, D. A. (2011). PROMIS computerised adaptive tests are dynamic instruments to measure health-related quality of life in patients with cirrhosis. Alimentary Pharmacology & Therapeutics, 34(9), 1123–1132.
Brazier, J.-H.H.R., Jones, N., Ocathain, A., Thomas, K., Usherwood, T., & Westlake, L. (1992). Validating the SF-36 health survey questionnaire: New outcome measure for primary care. British Medical Journal, 305, 60–64.
Hunt, S. M., McKenna, S. P., McEwen, J., Williams, J., & Papp, E. (1981). The Nottingham health profile: Subjective health status and medical consultations. Social Science & Medicine., 15(3), 221–229.
O’Carroll, R. E. S. K., Couston, M., Cossar, J. A., & Hayes, P. C. (2000). A comparison of the WHOQOL-100 and the WHOQOL-BREF in detecting change in quality of life following liver transplantation. Quality of Life Research, 9(1), 121–124.
Herdman, M., Gudex, C., Lloyd, A., Janssen, M. F., Kind, P., Parkin, D., Bonsel, G., & Badia, X. (2011). Development and preliminary testing of the new five-level version of EQ-5D (EQ-5D-5L). Quality of Life Research, 20(10), 1727–1736.
Zigmond, A. S. S. R. (1983). The Hospital Anxiety and Depression Scale. Acta Psychiatrica Scand., 67, 361–370.
Beck, A. T., Epstein, N., Brown, G., & Steer, R. A. (1988). An inventory for measuring clinical anxiety: Psychometric properties. Journal of Consulting and Clinical Psychology., 56(6), 893–897.
Buysse, D. J. R. C., Monk, T. H., Berman, S. R., & Kupfer, D. J. (1989). The Pittsburgh Sleep Quality Index: A new instrument for psychiatric practice and research. Psychiatry Research, 28, 193–213.
Tong, A., Gill, J., Budde, K., Marson, L., Reese, P. P., Rosenbloom, D., Rostaing, L., Wong, G., Josephson, M. A., Pruett, T. L., Warrens, A. N., Craig, J. C., Sautenet, B., Evangelidis, N., Ralph, A. F., Hanson, C. S., Shen, J. I., Howard, K., Meyer, K., … Ro, C. (2017). Toward establishing core outcome domains for trials in kidney transplantation: Report of the standardized outcomes in nephrology—Kidney Transplantation (SONG-Tx) Consensus workshops. Transplantation, 101(8), 1887–1896.
Cella, D., Riley, W., Stone, A., Rothrock, N., Reeve, B., Yount, S., Amtmann, D., Bode, R., Buysse, D., Choi, S., Cook, K., DeVellis, R., DeWalt, D., Fries, J. F., Gershon, R., Hahn, E. A., Lai, J.-S., Pilkonis, P., Revicki, D., … Hays, R. (2010). The patient-reported outcomes measurement information system (PROMIS) developed and tested its first wave of adult self-reported health outcome item banks: 2005–2008. Journal of Clinical Epidemiology, 63(11), 1179–1194.
Gershon, R., Rothrock, N. E., Hanrahan, R. T., Jansky, L. J., Harniss, M., & Riley, W. (2010). The development of a clinical outcomes survey research application: Assessment Center. Quality of Life Research, 19(5), 677–685.
Bajaj, J. S., White, M. B., Unser, A. B., Ganapathy, D., Fagan, A., Gavis, E. A., Sterling, R. K., Heuman, D. M., Matherly, S., Puri, P., Sanyal, A. J., Luketic, V., Fuchs, M., Siddiqui, M. S., Todd Stravitz, R., John, B., Thacker, L. R., & Wade, J. B. (2016). Cirrhotic patients have good insight into their daily functional impairment despite prior hepatic encephalopathy: Comparison with PROMIS norms. Metabolic Brain Disease, 31(5), 1199–1203.
Younossi, Z. H. L. (2015). Overall health-related quality of life in patients with end-stage liver disease. Clinical Liver Disease, 6(1), 9–14.
Sabatino, M. J., Gans, C. V., Zynda, A. J., Chung, J. S., Miller, S. M., Wilson, P. L., Jo, C. H., & Ellis, H. B. (2019). An electronic patient-reported outcomes measurement system in paediatric orthopaedics. J Child Ortho, 4(13), 1–15.
Kyte, D., Bishop, J., Brettell, E., Calvert, M., Cockwell, P., Dutton, M., Eddington, H., Hadley, G., Ives, N. J., Jackson, L. J., Stringer, S., & Valente, M. (2018). Use of an electronic patient-reported outcome measure in the management of patients with advanced chronic kidney disease: The RePROM pilot trial protocol. British Medical Journal Open. https://doi.org/10.1136/bmjopen-2018-026080
Patricia Holch, K. A., Brooke, C., & Wang, X. (2020). Advances in patient reported outcomes: Integration and innovation. Journal of Patient Reported Outcomes. https://doi.org/10.1186/s41687-020-00193-x
Jensen, R. E. S. C., & Abernethy, A. P. (2014). Review of electronic patient-reported outcomes systems used in cancer clinical care. Journal of Oncology Practice/American Society of Clinical Oncology, 10, e215–e222.
Funding
None.
Author information
Authors and Affiliations
Contributions
IM, AV, SB, NS-M, and AO-C participated in study design. AV, RK, AC, AN, MS, AA, AW, AO-C, and SM participated in data abstraction and analysis. AV, IM, SB, and NS-M contributed to the interpretation of the data. IM, AV, SB, and NS-M contributed to the writing of the paper. All authors participated in drafting and finalizing the article.
Corresponding author
Ethics declarations
Conflict of interest
The authors of this manuscript have no conflicts of interest to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Vedadi, A., Khairalla, R., Che, A. et al. Patient-reported outcomes and patient-reported outcome measures in liver transplantation: a scoping review. Qual Life Res 32, 2435–2445 (2023). https://doi.org/10.1007/s11136-023-03405-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11136-023-03405-1