Abstract
Purpose
Anchoring vignettes (AVs) are a promising measurement technique to reduce bias in patient-reported outcome (PRO) measures by helping researchers understand differences in how individuals and groups interpret response options. However, little attention has been paid to ensure quality development of AVs, and their performance has not been well assessed in pediatric populations. In this study, we explore the application of a rigorous development process for AVs based upon current standards for PROs, as well as feasibility of AVs when administered to children and adolescents.
Methods
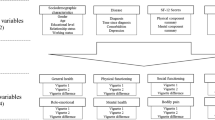
We developed AVs using a rigorous, patient-centered mixed methods process including three phases: (1) development, (2) a pilot study, and (3) a field test. Our proposed process included the generation of a conceptual framework based on the PRO, the Localized Scleroderma Quality of Life Instrument, and numerous vignette-specific considerations. We qualitatively explored readability and comprehension of the AVs (pilot study) and then analyzed ranking patterns within vignette sets (field test).
Results
Four sets of four vignettes were developed. Revisions were suggested at each phase of development. The pilot study demonstrated that children ≥ 10 years had no trouble indicating understanding of the AVs. In the field test, although appropriate rankings of vignettes were generally demonstrated by participants, the percentage of tied rankings was higher than expected in this pediatric group.
Conclusions
This work supports the need for rigorous developmental standards for AVs, as each stage of development suggested revisions. Additionally, AVs showed initial promise for use with pediatric populations; general feasibility and understanding were supported.


Similar content being viewed by others
References
U. S. Department of Health and Human Services, F. a. D. A., Center for Drug Evaluation and Research, Center for Biologics Evaluation and Research, and Center for Devices and Radiological Health. (2006). Guidance for industry: Patient-reported outcome measures: use in medical product development to support labeling claims: Draft guidance. Health Qual Life Outcomes, 4, 79. https://doi.org/10.1186/1477-7525-4-79.
Reeve, B. B., Wyrwich, K. W., Wu, A. W., Velikova, G., Terwee, C. B., Snyder, C. F., et al. (2013). ISOQOL recommends minimum standards for patient-reported outcome measures used in patient-centered outcomes and comparative effectiveness research. Quality of Life Research, 22(8), 1889–1905. https://doi.org/10.1007/s11136-012-0344-y.
Eignor D.R. (2013). The standards for educational and psychological testing: American psychological association.
King, G., Murray, C. J., Salomon, J. A., & Tandon, A. (2004). Enhancing the validity and cross-cultural comparability of measurement in survey research. American Political Science Review, 98(1), 191–207.
Grol-Prokopczyk, H., Freese, J., & Hauser, R. M. (2011). Using anchoring vignettes to assess group differences in general self-rated health. Journal of Health and Social Behavior, 52(2), 246–261. https://doi.org/10.1177/0022146510396713.
King, G., & Wand, J. (2007). Comparing incomparable survey responses: Evaluating and selecting anchoring vignettes. Political Analysis, 15(1), 46–66.
Schwartz, C. E. (2010). Applications of response shift theory and methods to participation measurement: A brief history of a young field. Archives of Physical Medicine Rehabilitation, 91(9), S38–S43.
Bolt, D. M., Lu, Y., & Kim, J.-S. (2014). Measurement and control of response styles using anchoring vignettes: A model-based approach. Journal Psychological Methods, 19(4), 528.
Primi R., Zanon C., Santos D., De Fruyt F., John O.P. (2016). Anchoring vignettes. European Journal of Psychological Assessment.
Vonkova, H., Bendl, S., & Papajoanu, O. (2017). How students report dishonest behavior in school: Self-assessment and anchoring vignettes. The Journal of Experimental Education, 85(1), 36–53.
Vonkova H., Zamarro G., Deberg V., Hitt C. (2015). Comparisons of student perceptions of teacher’s performance in the classroom: Using parametric anchoring vignette methods for improving comparability. EDRE Working Paper No. 2015–01
Marksteiner T., Kuger S., Klieme E. (2018). The potential of anchoring vignettes to increase intercultural comparability of non-cognitive factors. Assessment in Education: Principles, Policy & Practice, 1–21.
Vonkova, H., & Hrabak, J. (2015). The (in) comparability of ICT knowledge and skill self-assessments among upper secondary school students: The use of the anchoring vignette method. Computers Education, 85, 191–202.
Vonkova, H. (2019). Life satisfaction among different groups of children: self-reports, differential scale usage and anchoring vignettes. Child Indicators Research. https://doi.org/10.1007/s12187-019-09629-3.
Primi R., Santos D., John O.P., De Fruyt F., Hauck-Filho N. (2017). Dealing with person differential item functioning in social-emotional skill assessment using anchoring vignettes. In The Annual Meeting of the Psychometric Society, (pp. 275–286): Springer
Grol-Prokopczyk, H., Verdes-Tennant, E., McEniry, M., & Ispány, M. (2015). Promises and pitfalls of anchoring vignettes in health survey research. Demography, 52(5), 1703–1728.
Grol-Prokopczyk, H. (2018). In pursuit of anchoring vignettes that work: evaluating generality versus specificity in vignette texts. The Journals of Gerontology: Series B, 73(1), 54–63. https://doi.org/10.1093/geronb/gbx048.
Jürges, H., & Winter, J. (2013). Are anchoring vignette ratings sensitive to vignette age and sex? Health Economics, 22(1), 1–13. https://doi.org/10.1002/hec.1806.
Zigler, C. K., Ardalan, K., Lane, S., Schollaert, K. L., & Torok, K. S. (2019). A novel patient-reported outcome for paediatric localized scleroderma: A qualitative assessment of content validity. British Journal of Dermatology. https://doi.org/10.1111/bjd.18512.
Zulian, F., Athreya, B. H., Laxer, R., Nelson, A. M., Feitosa de Oliveira, S. K., Punaro, M. G., et al. (2006). Juvenile localized scleroderma: Clinical and epidemiological features in 750 children. An international study. Rheumatology (Oxford), 45(5), 614–620. https://doi.org/10.1093/rheumatology/kei251.
Zigler, C. K., Ardalan, K., Hernandez, A., Calliendo, A. E., Magee, K., Mann, C. M., et al. (2020). Exploring the impact of paediatric localized scleroderma on health-related quality of life: Focus groups with youth and caregivers. British Journal of Dermatology. https://doi.org/10.1111/bjd.18879.
Hopkins, D. J., & King, G. (2010). Improving anchoring vignettes: Designing surveys to correct interpersonal incomparability. Public Opinion Quarterly, 74(2), 201–222.
Collins, D. (2003). Pretesting survey instruments: An overview of cognitive methods. Quality of Life Research, 12(3), 229–238.
Wand, J. (2013). Credible comparisons using interpersonally incomparable data: Nonparametric scales with anchoring vignettes. American Journal of Political Science, 57(1), 249–262.
Wand J., King G., Lau O. (2011). Anchors: Software for anchoring vignette data. Journal of Statistical Software.
Stasiulis, E., Gladstone, B., Boydell, K., O'Brien, C., Pope, E., & Laxer, R. M. (2018). Children with facial morphoea managing everyday life: a qualitative study. British Journal of Dermatology, 179(2), 353–361. https://doi.org/10.1111/bjd.16449.
Murray, K. J., & Laxer, R. M. (2002). Scleroderma in children and adolescents. Rheumatic Diseases Clinics of North America, 28(3), 603–624.
Wu, E. Y., Li, S. C., Torok, K. S., Virkud, Y. V., Fuhlbrigge, R. C., Rabinovich, C. E., et al. (2019). Baseline description of the juvenile localized scleroderma subgroup from the childhood arthritis and rheumatology research alliance legacy registry. ACR Open Rheumatology, 1(2), 119–124. https://doi.org/10.1002/acr2.1019.
Varni, J. W., Limbers, C. A., & Burwinkle, T. M. (2007). How young can children reliably and validly self-report their health-related quality of life? An analysis of 8,591 children across age subgroups with the PedsQL 4.0 Generic Core Scales. Health Qual Life Outcomes, 5, 1–1. https://doi.org/10.1186/1477-7525-5-1.
Riley, A. W. (2004). Evidence that school-age children can self-report on their health. Ambulatory Pediatrics, 4(4 Suppl), 371–376. https://doi.org/10.1367/a03-178r.1.
Rieffe, C., Ketelaar, L., & Wiefferink, C. (2010). Assessing empathy in young children: Construction and validation of an Empathy Questionnaire (EmQue). Personality and Individual Differences, 49, 362–367. https://doi.org/10.1016/j.paid.2010.03.046.
von Davier, M., Shin, H. J., Khorramdel, L., & Stankov, L. (2018). The Effects of vignette scoring on reliability and validity of self-reports. Applied Psychological Measurement, 42(4), 291–306. https://doi.org/10.1177/0146621617730389.
Acknowledgements
The authors would like to thank Rose Cannon, Jennifer Coias, Dafonso Davage, Stephanie Flores-Pollack, Smriti Prasad, and Noelle Teske for their invaluable assistance with this project, as well as Clement Stone for his insight and input into project design. We also would like to acknowledge the vital involvement of the participants and their families, whose experiences drive this work.
Funding
Dr. Torok was supported by The Nancy Taylor Foundation for Chronic Diseases, Inc. (PI: Torok) and NIH-NIAMS (1K23AR059722, PI: Torok). Dr. Jacobe was funded by the James N. Gilliam, M.D. Chair in Dermatology.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. Zigler declares she has no conflicts of interest. Dr. Jacobe declares she has no conflicts of interest. Dr. Ardalan declares he has no conflicts of interest. Ms. Schollaert declares she has no conflicts of interest. Dr. Lane declares she has no conflicts of interest. Dr. Torok declares she has no conflicts of interest. Dr. Coles declares that she has no conflicts of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the University of Pittsburgh and UT Southwestern Institutional Review Boards (IRB) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Specifically, the qualitative pilot study occurred at and was approved by the University of Pittsburgh IRB and the quantitative field test occurred at and was approved by both the University of Pittsburgh and the UT Southwestern IRBs.
Informed consent
Informed consent was obtained from all individual participants included in the study. Adult participant (> 18 yrs old) consent and parental consent/child assent (for minor participants) were obtained in accordance with each institution’s policy.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zigler, C.K., Jacobe, H., Ardalan, K. et al. The importance of development standards for anchoring vignettes: an illustrative example from pediatric localized scleroderma quality of life. Qual Life Res 29, 3263–3272 (2020). https://doi.org/10.1007/s11136-020-02575-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11136-020-02575-6