Abstract
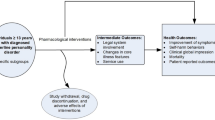
Twenty-three adolescents with psychotic disorders, aged from 13 to 18 years, participated in a 12-week open label trial (17 adolescents completed the study) in order to examine the impact of quetiapine on clinical status and cognitive functions (encompassing processing speed, attention, short-term memory, long-term memory and executive function). An improvement in Clinical Global Impression and Positive and Negative Symptom Scale (P’s ≤ 0.001) was observed. In addition, after controlling for amelioration of symptoms, a significant improvement was observed on one executive function (P = 0.044; Trail Making Part B). The remaining cognitive abilities showed stability. In addition, we observed an interaction between quetiapine doses (>300 mg/day or <300 mg/day) and time, where lower doses showed more improvement in verbal short-term memory (P = 0.048), inhibition abilities (P = 0.038) and positive symptoms (P = 0.020). The neuropsychological functioning of adolescents with psychotic disorders remained mainly stable after 12 weeks of treatment with quetiapine. However, lower doses seemed to have a better impact on two components of cognition (inhibition abilities and verbal short-term memory) and on positive symptoms.

Similar content being viewed by others
References
APA: Diagnostic and Statistical Manual of Mental Disorders (DSM-IV), 4th edn. Washington, DC, American Psychiatric Association, 1994
Asarnow RF, Asamen J, Granholm E, et al.: Cognitive/neuropsychological studies of children with a schizophrenic disorder. Schizophrenia Bulletin 20:647–669, 1994
Holzer L, Chinet L, Jaugey L, et al.: Detection of cognitive impairment with the Repeatable Battery for the Assessment of Neuropsychological Status (RBANS) in adolescents with psychotic symptomatology. Schizophrenia Research 95:48–53, 2007
Kenny JT, Friedman L, Findling RL, et al.: Cognitive impairment in adolescents with schizophrenia. American Journal of Psychiatry 154:1613–1615, 1997
Kumra S, Wiggs E, Bedwell J, et al.: Neuropsychological deficits in pediatric patients with childhood-onset schizophrenia and psychotic disorder not otherwise specified. Schizophrenia Research 42:135–144, 2000
Rhinewine JP, Lencz T, Thaden EP, et al.: Neurocognitive profile in adolescents with early-onset schizophrenia: Clinical correlates. Biological Psychiatry 58:705–712, 2005
Green MF: What are the functional consequences of neurocognitive deficits in schizophrenia? American Journal of Psychiatry 153:321–330, 1996
Hofer A, Baumgartner S, Bodner T, et al.: Patient outcomes in schizophrenia II: The impact of cognition. European Psychiatry 20:395–402, 2005
Robles O, Zabala A, Bombin I, et al.: Cognitive Efficacy of quetiapine and olanzapine in early-onset first-episode psychosis. Schizophrenia Bulletin 37:405–415, 2011
Riedel M, Spellmann I, Strassnig M, et al.: Effects of risperidone and quetiapine on cognition in patients with schizophrenia and predominantly negative symptoms. European Archives of Psychiatry and Clinical Neuroscience 257:360–370, 2007
Velligan DI, Newcomer J, Pultz J, et al.: Does cognitive function improve with quetiapine in comparison to haloperidol? Schizophrenia Research 53:239–248, 2002
Zhong KX, Sweitzer DE, Hamer RM, et al.: Comparison of quetiapine and risperidone in the treatment of schizophrenia: A randomized, double-blind, flexible-dose, 8-week study. Journal of Clinical Psychiatry 67:1093–1103, 2006
Tandon R: Improvement without impairment: A review of clinical data for quetiapine in the treatment of schizophrenia. Journal of Clinical Psychopharmacology 23:S15–S20, 2003
Andersen R, Fagerlund B, Rasmussen H, et al.: Cognitive effects of six months treatment with quetiapine in antipsychotic-naïve first-episode schizophrenia. Psychiatry Research 187:49–54, 2011
Quintana H, Wilson MS 2nd, Purnell W, et al.: An open-label study of olanzapine in children and adolescents with schizophrenia. Journal of Psychiatric Practice 13:86–96, 2007
Sanford M, Keating GM: Aripiprazole: In adolescents with schizophrenia. Paediatric Drugs 9:419–423, 2007
Arango C, Robles O, Parellada M, et al.: Olanzapine compared to quetiapine in adolescents with a first psychotic episode. European Child and Adolescent Psychiatry 18:418–428, 2009
Findling RL, McNamara NK, Youngstrom EA, et al.: A prospective, open-label trial of olanzapine in adolescents with schizophrenia. Journal of the American Academy of Child and Adolescent Psychiatry 42:170–175, 2003
Dittmann RW, Meyer E, Freisleder FJ, et al.: Effectiveness and tolerability of olanzapine in the treatment of adolescents with schizophrenia and related psychotic disorders: Results from a large, prospective, open-label study. Journal of Child and Adolescent Psychopharmacology 18:54–69, 2008
McConville BJ, Arvanitis LA, Thyrum PT, et al.: Pharmacokinetics, tolerability, and clinical effectiveness of quetiapine fumarate: An open-label trial in adolescents with psychotic disorders. Journal of Clinical Psychiatry 61:252–260, 2000
Shaw JA, Lewis JE, Pascal S, et al.: A study of quetiapine: Efficacy and tolerability in psychotic adolescents. Journal of Child and Adolescent Psychopharmacology 11:415–424, 2001
Swadi HS, Craig BJ, Pirwani NZ, et al.: A trial of quetiapine compared with risperidone in the treatment of first onset psychosis among 15- to 18-year-old adolescents. International Clinical Psychopharmacology 25:1–6, 2010
Richelson E: New antipsychotic drugs: How do their receptor-binding profiles compare? Journal of Clinical Psychiatry 71:1243–1244, 2010
Jensen NH, Rodriguiz RM, Caron MG, et al.: N-Desalkylquetiapine, a potent norepinephrine reuptake inhibitor and partial 5-HT1A agonist, as a putative mediator of quetiapine’s antidepressant activity. Neuropsychopharmacology 33:2303–2312, 2008
Yatham LN, Kennedy SH, Schaffer A, et al.: Canadian Network for Mood and Anxiety Treatments (CANMAT) and International Society for Bipolar Disorders (ISBD) collaborative update of CANMAT guidelines for the management of patients with bipolar disorder: Update 2009. Bipolar Disorder 11:225–255, 2009
Chamberlain SR, Muller U, Robbins TW, et al.: Neuropharmacological modulation of cognition. Current Opinion in Neurology 19:607–612, 2006
Nathan PJ, Sitaram G, Stough C, et al.: Serotonin, noradrenaline and cognitive function: A preliminary investigation of the acute pharmacodynamic effects of a serotonin versus a serotonin and noradrenaline reuptake inhibitor. Behavioural Pharmacology 11:639–642, 2000
Holzer L, Preuss U, Baumgartner L, et al.: Quetiapine in adolescents with non-affective psychotic disorders: An open-label trial. Pharmacopsychiatry 44:87–95, 2011
Kay SR, Fiszbein A, Opler LA: The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophrenia Bulletin 13:261–276, 1987
Guy W: ECDEU Assessment Manual. Rockville, MD, US Department of Health, Education and Welfare, 1976
Reitan R, Wolfson D: The Halstead-Reitan Neuropsychological Test Battery: Theory and Clinical Interpretation. Tucson, AZ, Neuropsychology Press, 1985
Asarnow RF, Nuechterlein KH, Subotnik KL, et al.: Neurocognitive impairments in nonpsychotic parents of children with schizophrenia and attention-deficit/hyperactivity disorder: The University of California, Los Angeles Family Study. Archives of General Psychiatry 59:1053–1060, 2002
Nuechterlein KH: Vigilance in Schizophrenia and Related Disorders. In: Steinhauer SR, Gruzelier JH, Zubin J (Eds) Handbook of Schizophrenia: Neuropsychology, Psychophysiology and Information Processing. New York, Elsevier Science Inc., 1991
Hebb DO: Distinctive Features of Learning in the Higher Animal. In: Delafresnaye JE (Ed) Brain Mechanisms and Learning. New York, Oxford University Press, 1961
Corsi PM: Human Memory and the Medial Temporal Region of the Brain. Dissertation Abstracts International 34:891B (University Microfilms No. AAI05-77717), 1972
Rey A: L’examen clinique en psychologie. Paris, Presses Universitaires de France, 1964
Osterrieth, P: Le test de copie d’une figure complexe: Contribution à l’étude de la perception et de la mémoire. Archives de Psychologie, Geneva 30:205–220, 1944
Spreen O, Strauss E: A Compendium of Neuropsychological Tests: Administration, Norms, and Commentary, 2nd edn. New York, Oxford University Press, 1998
Bolognini M, Plancherel B, Laget J, et al.: Evaluation of the adolescent drug abuse diagnosis instrument in a Swiss sample of drug abusers. Addiction 96:1477–1484, 2001
Buchanan RW, Davis M, Goff D, et al.: A summary of the FDA-NIMH-MATRICS workshop on clinical trial design for neurocognitive drugs for schizophrenia. Schizophrenia Bulletin 31:5–19, 2005
Gothelf D, Apter A, Reidman J, et al.: Olanzapine, risperidone and haloperidol in the treatment of adolescent patients with schizophrenia. Journal of Neural Transmission 110:545–560, 2003
Miyake A, Friedman NP, Emerson MJ, et al.: The unity and diversity of executive functions and their contributions to complex “Frontal Lobe” tasks: A latent variable analysis. Cognitive Psychology 41:49–100, 2000
Good KP, Kiss I, Buiteman C, et al.: Improvement in cognitive functioning in patients with first-episode psychosis during treatment with quetiapine: An interim analysis. British Journal of Psychiatry Supplement 43:s45–s49, 2002
Kivircik Akdede BB, Alptekin K, Kitis A, et al.: Effects of quetiapine on cognitive functions in schizophrenia. Progress in Neuro-Psychopharmacology & Biological Psychiatry 29:233–238, 2005
Arvanitis LA, Miller BG: Multiple fixed doses of “Seroquel” (quetiapine) in patients with acute exacerbation of schizophrenia: A comparison with haloperidol and placebo. The Seroquel Trial 13 Study Group. Biological Psychiatry 42:233–246, 1997
Small JG, Hirsch SR, Arvanitis LA, et al.: Quetiapine in patients with schizophrenia. A high- and low-dose double-blind comparison with placebo. Seroquel Study Group. Archives of General Psychiatry 54:549–557, 1997
Kahn RS, Fleischhacker WW, Boter H, et al.: Effectiveness of antipsychotic drugs in first-episode schizophrenia and schizophreniform disorder: An open randomised clinical trial. Lancet 371:1085–1097, 2008
Robinson DG, Woerner MG, Alvir JM, et al.: Predictors of treatment response from a first episode of schizophrenia or schizoaffective disorder. American Journal of Psychiatry 156:544–549, 1999
Robinson DG, Woerner MG, Delman HM, et al.: Pharmacological treatments for first-episode schizophrenia. Schizophrenia Bulletin 31:705–722, 2005
Suhara T, Okubo Y, Yasuno F, et al.: Decreased dopamine D2 receptor binding in the anterior cingulate cortex in schizophrenia. Archives of General Psychiatry 59:25–30, 2002
Riedel M, Muller N, Strassnig M, et al.: Quetiapine in the treatment of schizophrenia and related disorders. Neuropsychiatric Disease and Treatment 3:219–235, 2007
Kapur S, Zipursky R, Jones C, et al.: Relationship between dopamine D(2) occupancy, clinical response, and side effects: A double-blind PET study of first-episode schizophrenia. American Journal of Psychiatry 157:514–520, 2000
Kapur S, Zipursky R, Jones C, et al.: A positron emission tomography study of quetiapine in schizophrenia: A preliminary finding of an antipsychotic effect with only transiently high dopamine D2 receptor occupancy. Archives of General Psychiatry 57:553–559, 2000
Tandon R, Milner K, Jibson MD: Antipsychotics from theory to practice: Integrating clinical and basic data. Journal of Clinical Psychiatry 60(Suppl 8):21–28, 1999
Nikisch G, Baumann P, Wiedemann G, et al.: Quetiapine and norquetiapine in plasma and cerebrospinal fluid of schizophrenic patients treated with quetiapine: Correlations to clinical outcome and HVA, 5-HIAA, and MHPG in CSF. Journal of Clinical Psychopharmacology 30:496–503, 2010
Holzer L, Halfon O: Normalization of impaired cognitive functions failed to improve clinical symptomatology in a schizophrenic patient. European Psychiatry 19:384–386, 2004
Conway Greig T, Nicholls SS, Wexler BE, et al.: Test–retest stability of neuropsychological testing and individual differences in variability in schizophrenia outpatients. Psychiatry Research 129:241–247, 2004
Acknowledgment
This study was financially supported by AstraZeneca, Switzerland, by an unrestricted grant.
Conflict of interest
PB and UP have received honoraries or sponsorships from almost all pharmaceutical companies in Switzerland selling psychotropic drugs. In particular, they received honoraries from the manufacturer of quetiapine (AstraZeneca). Others authors reported no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Urben, S., Baumann, P., Barcellona, S. et al. Cognitive Efficacy of Quetiapine in Early-Onset First-Episode Psychosis: A 12-Week Open Label Trial. Psychiatr Q 83, 311–324 (2012). https://doi.org/10.1007/s11126-011-9201-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11126-011-9201-3