Abstract
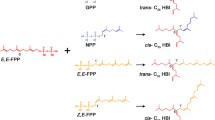
Geranylgeranyl reductase (GGR) encoded by the bchP gene catalyzes the reductions of three unsaturated C = C double bonds (C6 = C7, C10 = C11, and C14 = C15) in a geranylgeranyl (GG) group of the esterifying moiety in 17-propionate residue of bacteriochlorophyll (BChl) molecules. It was recently reported that GGR in Halorhodospira halochloris potentially catalyzes two hydrogenations, yielding BChl with a tetrahydrogeranylgeranyl (THGG) tail. Furthermore, its engineered GGR, in which N-terminal insertion peptides characteristic for H. halochloris were deleted, performed single hydrogenation, producing BChl with a dihydrogeranylgeranyl (DHGG) tail. In some of these enzymatic reactions, it remained unclear in which order the C = C double bond in a GG group was first reduced. In this study, we demonstrated that the (variant) GGR from H. halochloris catalyzed an initial reduction of the C6 = C7 double bond to yield a 6,7-DHGG tail. The intact GGR of H. halochloris catalyzed the further hydrogenation of the C14 = C15 double bonds to give a 6,7,14,15-THGG group, whereas deleting the characteristic peptide region from the GGR suppressed the C14 = C15 reduction. We also verified that in a model bacterium, Blastochloris viridis producing standard BChl-b, the reduction of a GG to phytyl group occurred via 10,11-DHGG and 6,7,10,11-THGG. The high-performance liquid chromatographic elution profiles of BChls-a/b employed in this study are essential for identifying the regioisomeric diterpenoid tails in the BChls of phototrophic bacteria distributed in nature and elucidating GGR enzymatic reactions.






Similar content being viewed by others
Abbreviations
- APCI:
-
Atmospheric pressure chemical ionization
- BChl:
-
Bacteriochlorophyll
- BChlide:
-
Bacteriochlorophyllide
- DHGG:
-
Dihydrogeranylgeranyl
- DHGG-OH:
-
Dihydrogeranylgeraniol
- GC:
-
Gas chromatography
- GG:
-
Geranylgeranyl
- GG-OH:
-
Geranylgeraniol
- GGR:
-
Geranylgeranyl reductase
- HPLC:
-
High-performance liquid chromatography
- LC–MS:
-
Liquid chromatography–mass spectrometry
- LH:
-
Light-harvesting
- MS:
-
Mass spectrometry
- PDA:
-
Photodiode array
- RC:
-
Reaction center
- THGG:
-
Tetrahydrogeranylgeranyl
- THGG-OH:
-
Tetrahydrogeranylgeraniol
- tR:
-
Retention time
- UV–VIS–NIR:
-
Ultraviolet–visible–near infrared
- rt:
-
Room temperature
References
Beale SI (1999) Enzymes of chlorophyll biosynthesis. Photosynth Res 60:43–73. https://doi.org/10.1023/A:1006297731456
Blankenship RE, Olson JM, Miller M (1995) Antenna complexes from green photosynthetic bacteria. In: Madigan MT, Bauer CE (eds) Anoxygenic photosynthetic bacteria, Advanced Photosynthesis, vol 2. Kluwer Academic, Dordrecht, pp 399–435
Bryant DA, Hunter CN, Marren MJ (2020) Biosynthesis of the modified tetrapyrroles—the pigments of life. J Biol Chem 295:6888–6925. https://doi.org/10.1074/jbc.REV120.006194
Chen JH, Wu H, Xu C, Liu XC, Huang Z, Chang S, Wang W, Han G, Kuang T, Shen JR, Zhang X (2020) Architecture of the photosynthetic complex from a green sulfur bacterium. Science 370:abb6350. https://doi.org/10.1126/science.abb6350
Chew AGM, Bryant DA (2007) Chlorophyll biosynthesis in bacteria: the origins of structural and functional diversity. Annu Rev Microbiol 61:113–129. https://doi.org/10.1146/annurev.micro.61.080706.093242
Costas AMG, Tsukatani Y, Rijpstra WIC, Schouten S, Welander PV, Summons RE, Bryant DA (2012) Identification of the bacteriochlorophylls, carotenoids, quinones, lipids, and hopanoids of “Candidatus Chloracidobacterium thermophilum.” J Bacteriol 194:1158–1168. https://doi.org/10.1128/JB.06421-11
Croce R, van Amerongen H (2020) Light harvesting in oxygenic photosynthesis: Structural biology meets spectroscopy. Science 369:eaay2058. https://doi.org/10.1126/science.aay2058
Eckhardt U, Grimm B, Hörtensteiner S (2004) Recent advances in chlorophyll biosynthesis and breakdown in higher plants. Plant Mol Biol 56:1–14. https://doi.org/10.1007/s11103-004-2331-3
Fiedor L, Kania A, Kurdziel BM, Orzeł Ł, Stochel G (2008) Understanding chlorophylls: Central magnesium ion and phytyl as structural determinants. Biochim Biophys Acta Bioenerg 1777:1491–1500. https://doi.org/10.1016/j.bbabio.2008.09.005
Fiedor L, Zbyradowski M, Pilch M (2019) Tetrapyrrole pigments of photosynthetic antennae and reaction centers of higher plants: Structures, biophysics, functions, biochemistry, mechanisms of regulation, applications. In: Bernhard G (ed) Metabolism, structure and function of plant tetrapyrroles: Introduction, microbial and eukaryotic chlorophyll synthesis and catabolism, Advanced Botany Research, vol 90. Academic, London, pp 1–33
Gutbrod K, Romer J, Dörmann P (2019) Phytol metabolism in plants. Prog Lipid Res 74:1–17. https://doi.org/10.1016/j.plipres.2019.01.002
Hirose M, Harada J, Tamiaki H (2020) In vitro hydrolysis of zinc chlorophyllide a homologues by a BciC enzyme. Biochemistry 59:4622–4626. https://doi.org/10.1021/acs.biochem.0c00850
Imhoff JF, Trüper HG (1997) Ectothiorhodospira halochloris sp. nov., a new extremely halophilic phototrophic bacterium containing bacteriochlorophyll b. Arch Microbiol 114:115–121. https://doi.org/10.1007/BF00410772
Kimura Y, Nojima S, Nakata K, Yamashita T, Wang XP, Takenaka S, Akimoto S, Kobayashi M, Madigan MT, Wang-Otomo ZY, Yu LJ (2021) Electrostatic charge controls the lowest LH1 Qy transition energy in the triply extremophilic purple phototrophic bacterium, Halorhodospira Halochloris. Biochim Biophys Acta Bioenerg 1862:148473. https://doi.org/10.1016/j.bbabio.2021.148473
Liu Y, Zhang S, Lindsey JS (2018) Total synthesis campaigns toward chlorophylls and related natural hydroporphyrins – diverse macrocycles, unrealized opportunities. Nat Prod Rep 35:879–901. https://doi.org/10.1039/C8NP00020D
Matsubara S, Tamiaki H (2020) Supramolecular chlorophyll aggregates inspired from specific light-harvesting antenna “chlorosome”: Static nanostructure, dynamic construction process, and versatile application. J Photochem Photobiol C: Photochem Rev 45:100385. https://doi.org/10.1016/j.jphotochemrev.2020.100385
Miyatake T, Tamiaki H (2010) Self-aggregates of natural chlorophylls and their synthetic analogues in aqueous media for making light-harvesting systems. Coord Chem Rev 254:2593–2602. https://doi.org/10.1016/j.ccr.2009.12.027
Mizoguchi T, Harada J, Tamiaki H (2006) Structural determination of dihydro- and tetrahydrogeranylgeranyl groups at the 17-propionate of bacteriochlorophylls-a. FEBS Lett 580:6644–6648. https://doi.org/10.1016/j.febslet.2006.11.020
Mizoguchi T, Isaji M, Harada J, Watabe K, Tamiaki H (2009) Structural determination of the Δ2,10-phytadienyl substituent in the 17-propionate of bacteriochlorophyll-b from Halorhodospira halochloris. J Porphyr Phthalocyanines 13:41–50. https://doi.org/10.1142/S1088424609000218
Mizoguchi T, Isaji M, Yamano N, Harada J, Fujii R, Tamiaki H (2017) Molecular structures and functions of chlorophylls-a esterified with geranylgeranyl, dihydrogeranylgeranyl, and tetrahydrogeranylgeranyl groups at the 17-propionate residue in a diatom, Chaetoceros calcitrans. Biochemistry 56:3682–3688. https://doi.org/10.1021/acs.biochem.7b00381
Qian P, Siebert CA, Wang P, Canniffe DP, Hunter CN (2018) Cryo-EM structure of the Blastochloris viridis LH1–RC complex at 2.9 Å. Nature 556:203–208. https://doi.org/10.1038/s41586-018-0014-5
Qiu NW, Jiang DC, Wang XS, Wang BS, Zhou F (2019) Advances in the members and biosynthesis of chlorophyll family. Photosynthetica 57:974–984. https://doi.org/10.32615/ps.2019.116
Rüdiger W (2003) The last steps of chlorophyll synthesis. In: Kadish KM, Smith KM, Guilard R (eds) The Porphyrin Handbook, vol 13. Elsevier Science, San Diego, pp 71–108
Rüdiger W (2006) Biosynthesis of chlorophylls a and b: The last steps. In: Robert BG, Porra J, Rüdiger W, Scheer H (eds) Chlorophylls and bacteriochlorophylls: Biochemistry, biophysics, functions and applications, Adv Photosynth Respir, vol 25. Springer, Dordrecht, pp 189–200
Ryan AA, Senge MO (2015) How green is green chemistry? Chlorophylls as a bioresource from biorefineries and their commercial potential in medicine and photovoltaics. Photochem Photobiol Sci 14:638–660. https://doi.org/10.1039/C4PP00435C
Saga Y, Yamashita H, Hirota K (2016) Introduction of perfluoroalkyl chain into the esterifying moiety of bacteriochlorophyll c in the green sulfur photosynthetic bacterium Chlorobaculum tepidum by pigment biosynthesis. Bioorg Med Chem 24:4165–4170. https://doi.org/10.1016/j.bmc.2016.07.004
Sattley WM, Madigan MT, Swingley WD, Cheung PC, Clocksin KM, Conrad AL, Dejesa LC, Honchak BM, Jung DO, Karbach LE, Kurdoglu A, Lahiri S, Mastrian SD, Page LE, Taylor HL, Wang ZT, Raymond J, Chen M, Blankenship RE, Touchman JW (2008) The genome of Heliobacterium modesticaldum, a phototrophic representative of the Firmicutes containing the simplest photosynthetic apparatus. J Bacteriol 190:4687–4696. https://doi.org/10.1128/JB.00299-08
Scheer H (1991) Structure and occurrence of chlorophylls. In: Scheer H (ed) Chlorophylls. CRC, Boca Raton, pp 3–30
Scheer H (2003) The pigments. In: Green BR, Parson WW (eds) Light-harvesting antennas in photosynthesis, Adv Photosynth Respir, vol 13. Kluwer Academic, Dordrecht, pp 29–81
Simkin AJ, Kapoor L, Doss CGP, Hofmann TA, Lawson T, Ramamoorthy S (2022) The role of photosynthesis related pigments in light harvesting, photoprotection and enhancement of photosynthetic yield in planta. Photosynth Res 152:23–42. https://doi.org/10.1007/s11120-021-00892-6
Steiner R, Schäfer W, Bios I, Wieschhoff H, Scheer H (1981) 2,10-Phytadienol as esterifying alcohol of bacteriochlorophyll b from Ectothiorhodospira halochloris. Z Naturforsch 36c:417–420. https://doi.org/10.1515/znc-1981-5-613
Steiner R, Wieschhoff H, Scheer H (1982) High-performance liquid chromatography of bacteriochlorophyll b and its derivatives as an aid for structure analysis. J Chromatogr A 242:127–134. https://doi.org/10.1016/S0021-9673(00)87254-4
Takaichi S, Maoka T, Harada S, Imhoff JF (2001) Dihydroxylycopene diglucoside diesters: A novel class of carotenoids from the phototrophic purple sulfur bacteria Halorhodospira abdelmalekii and Halorhodospira halochloris. Arch Microbiol 175:161–167. https://doi.org/10.1007/s002030000247
Tamiaki H, Kunieda M (2011) Photochemistry of chlorophylls and their synthetic analogs. In: Kadish KM, Smith KM, Guilard R (eds) Handbook of Porphyrin Science, vol 11. World Scientific, Singapore, pp 223–290
Tamiaki H, Shibata R, Mizoguchi T (2007) The 17-propionate function of (bacterio)chlorophylls: Biological implication of their long esterifying chains in photosynthetic systems. Photochem Photobiol 83:152–162. https://doi.org/10.1562/2006-02-27-IR-819
Tamiaki H, Teramura M, Tsukatani Y (2016) Reduction processes in biosynthesis of chlorophyll molecules: Chemical implication of enzymatically regio- and stereoselective hydrogenations in the late stages of their biosynthetic pathway. Bull Chem Soc Jpn 89:161–173. https://doi.org/10.1246/bcsj.20150307
Tamiaki H, Nomura K, Mizoguchi T (2017) Preparation of regio- and stereoisomeric di- and tetrahydrogeranylgeraniols and identification of esterifying groups in natural (bacterio)chlorophylls. Bioorg Med Chem 25:6361–6370. https://doi.org/10.1016/j.bmc.2017.10.002
Tani K, Kanno R, Ji XC, Hall M, Yu LJ, Kimura Y, Madigan MT, Mizoguchi A, Humbel BM, Otomo ZYW (2021) Cryo-EM structure of the photosynthetic LH1-RC complex from Rhodospirillum rubrum. Biochemistry 60:2483–2491. https://doi.org/10.1021/acs.biochem.1c00360
Thornber JP, Cogdell RJ, Seftor REB, Webster GD (1980) Further studies on the composition and spectral properties of the photochemical reaction centers of bacteriochlorophyll b-containing bacteria. Biochim Biophys Acta Bioenerg 593:60–75. https://doi.org/10.1016/0005-2728(80)90008-0
Tsukatani Y, Tamiaki H (2019) In vitro and in vivo synthesis of bacteriochlorophyll absorbing near-infrared light. In: Réglier M (ed) Bioinspired chemistry: From enzymes to synthetic models, series on chemistry, energy and the environment, vol 5. World Scientific, Singapore, pp 1–17
Tsukatani Y, Hirose Y, Harada J, Yonekawa C, Tamiaki H (2019) Unusual features in the photosynthetic machinery of Halorhodospira halochloris DSM 1059 revealed by complete genome sequencing. Photosynth Res 140:311–319. https://doi.org/10.1007/s11120-019-00613-0
Tsukatani Y, Harada J, Kurosawa K, Tanaka K, Tamiaki H (2022) Incomplete hydrogenation by geranylgeranyl reductase from a proteobacterial phototroph Halorhodospira halochloris, resulting in the production of bacteriochlorophyll with a tetrahydrogeranylgeranyl tail. J Bacteriol 204:e00605-e621. https://doi.org/10.1128/jb.00605-21
Xiong J, Fischer WM, Inoue K, Nakahara M, Bauer CE (2000) Molecular evidence for the early evolution of photosynthesis. Science 289:1724–1730. https://doi.org/10.1126/science.289.5485.1724
Zakhidov EA, Zakhidova MA, Kasymdzhanov MA, Kurbanov SS, Nematov SK, Norris JR, Ponomarenko NS, Khabibullaev PK (2004) The Qy band of bacteriochlorophyll as an indicator of interactions between structural functional elements of the purple bacterium Blastochloris viridis. Doklady Biochem Biophys 398:294–296. https://doi.org/10.1023/B:DOBI.0000046641.19675.f8
Acknowledgements
We would like to thank Prof. Kazuhito Inoue of Kanagawa University for his kind provision of a purple photosynthetic bacterium, B. viridis DSM 133. This work was partially supported by the Japan Society for the Promotion of Science (JSPS) KAKENHI Grant Numbers, 21J22995 (to M.H.), 19H02018 (to Y.T.), 18H03743 (to Y.T.), 22H02203 (to H.T.), and 17H06436 in the Scientific Research on Innovative Areas “Innovation for Light-Energy Conversion (I4LEC)” (to H.T.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hirose, M., Tsukatani, Y., Harada, J. et al. Characterization of regioisomeric diterpenoid tails in bacteriochlorophylls produced by geranylgeranyl reductase from Halorhodospira halochloris and Blastochloris viridis. Photosynth Res 154, 1–12 (2022). https://doi.org/10.1007/s11120-022-00938-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-022-00938-3