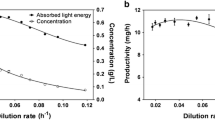
Abstract
In this work, we reconstructed the absorption spectrum of different Synechocystis sp. PCC 6803 optical strains by summing the computed signature of all pigments present in this organism. To do so, modifications to in vitro pigment spectra were first required: namely wavelength shift, curve smoothing, and the package effect calculation derived from high pigment densities were applied. As a result, we outlined a plausible shape for the in vivo absorption spectrum of each chromophore. These are flatter and slightly broader in physiological conditions yet the mean weight-specific absorption coefficient remains identical to the in vitro conditions. Moreover, we give an estimate of all pigment concentrations without applying spectrophotometric correlations, which are often prone to error. The computed cell spectrum reproduces in an accurate manner the experimental spectrum for all the studied wavelengths in the wild-type, Olive, and PAL strain. The gathered pigment concentrations are in agreement with reported values in literature. Moreover, different illumination set-ups were evaluated to calculate the mean absorption cross-section of each chromophore. Finally, a qualitative estimate of light-limited cellular growth at each wavelength is given. This investigation describes a novel way to approach the cell absorption spectrum and shows all its inherent potential for photosynthesis research.









Similar content being viewed by others
References
Aasen AJ, Liaaen Jensen S (1966) Carotenoids of flexibacteria. IV. The carotenoids of two further pigment types. Acta Chem Scand 20(8):2322–2324
Ajlani G, Vernotte C (1998) Construction and characterization of a phycobiliprotein-less mutant of Synechocystis Sp. PCC 6803. Plant Mol Biol 37(3):577–580
Bennett A, Bogobad L (1973) Complementary chromatic adaptation in a filamentous blue-green alga. J Cell Biol 58(2):419–435
Bidigare RR, Ondrusek ME, Morrow JH, Kiefer DA (1990) In-Vivo Absorption Properties of Algal Pigments. In: Ocean Optics X, SPIE, 290. https://spie.org/Publications/Proceedings/Paper/10.1117/12.21451. Accessed 31 March 2020
Bricaud A, Stramski D (1990) Spectral absorption coefficients of living phytoplankton and nonalgal biogenous matter: a comparison between the peru upwelling areaand the Sargasso Sea. Limnol Oceanogr 35(3):562–582
Bricaud A, Claustre H, Ras J, Oubelkheir K (2004) Natural variability of phytoplanktonic absorption in oceanic waters: influence of the size structure of algal populations. J Geophys Res 109:C11010
Bryant DA, Glazer AN, Eiserling FA (1976) Characterization and structural properties of the major biliproteins of Anabaena Sp. Arch Microbiol 110(1):61–75
Buschmann C, Nagel E (1993) In vivo spectroscopy and internal optics of leaves as basis for remote sensing of vegetation. Int J Remote Sens 14(4):711–722
Cerullo G et al (2002) Photosynthetic light harvesting by carotenoids: detection of an intermediate excited state. Science 298(5602):2395–2398
Chábera P et al (2011) Excited-state properties of the 16kDa red carotenoid protein from arthrospira maxima. Biochimica et Biophysica Acta - Bioenergetics 1807(1):30–35
Collins AM et al (2012) Photosynthetic pigment localization and thylakoid membrane morphology are altered in Synechocystis 6803 phycobilisome mutants. Plant Physiol 158(4):1600–1609
de Mooij T et al (2016) Impact of light color on photobioreactor productivity. Algal Res 15:32–42
Eng D, Baranoski GVG (2007) The application of photoacoustic absorption spectral data to the modeling of leaf optical properties in the visible range. In: IEEE transactions on geoscience and remote sensing, pp 4077–4086. https://ieeexplore.ieee.org/document/4378552. Accessed 31 March 2020
Faccio G et al (2014) Tyrosinase-catalyzed site-specific immobilization of engineered C-phycocyanin to surface. Sci Rep 4:5370
Ficek D et al (2004) Spectra of light absorption by phytoplankton pigments in the Baltic; conclusions to be drawn from a Gaussian analysis of empirical data. Oceanologia 46(4):533–555
Fuente D et al (2017) Light distribution and spectral composition within cultures of micro-algae: quantitative modelling of the light field in photobioreactors. Algal Res 23
Fujiki T, Taguchi S (2002) Variability in Chlorophyll a specific absorption coefficient in marine phytoplankton as a function of cell size and irradiance. J Plankton Res 24(9):859–874
Gobets B, Van Grondelle R (2001) Energy transfer and trapping in photosystem I. Biochim Biophys Acta 1507(1–3):80–99
Gobets B et al (2003) Excitation wavelength dependence of the fluorescence kinetics in photosystem I particles from Synechocystis PCC 6803 and Synechococcus Elongatus. Biophys J 85(6):3883–3898
Gong N, Li Z, Sun C, Men Z (2018) External field effect on electronic and vibrational properties of carotenoids. In: Progress in carotenoid research. InTech. https://www.intechopen.com/books/progress-in-carotenoid-research/external-field-effect-on-electronic-and-vibrational-properties-of-carotenoids. Accessed 31 March 2020
Green BR, Parson WW (2003) Light-harvesting antennas in photosynthesis. Adv Photosynth Resp 13:513
Greg Mitchell B, Kiefer DA (1988) Chlorophyll α specific absorption and fluorescence excitation spectra for light-limited phytoplankton. Deep Sea Res A 35(5):639–663
Herbert SK, Han T, Vogelmann TC (2000) New applications of photoacoustics to the study of photosynthesis. Photosynth Res 66(1–2):13–31
Hertzberg S, Liaaen-Jensen S, Siegelman HW (1971) The carotenoids of blue-green algae. Phytochemistry 10(12):3121–3127
Hiyama T, Nishimura M, Chance B (1969) Determination of carotenes by thin-layer chromatography. Anal Biochem 29(2):339–342
Hoepffner N, Sathyendranath S (1991) Effect of pigment composition on absorption properties of phytoplankton. Mar Ecol Prog Ser 73:11–23
Jordan P et al (2001) Three-dimensional structure of cyanobaoterial photosystem I at 2.5 Å resolution. Nature 411(6840):909–917
Joshua S, Mullineaux CW (2004) Phycobilisome diffusion is required for light-state transitions in cyanobacteria. Plant Physiol 135(4):2112–2119
Kakitani T, Honig B, Crofts AR (1982) Theoretical studies of the electrochromic response of carotenoids in photosynthetic membranes. Biophys J 39(1):57–63
Kilian O et al (2007) Responses of a thermophilic synechococcus isolate from the microbial mat of octopus spring to light. Appl Environ Microbiol 73(13):4268–4278
Kłodawska K et al (2015) Elevated growth temperature can enhance photosystem I trimer formation and affects xanthophyll biosynthesis in cyanobacterium Synechocystis Sp. PCC6803 Cells. Plant Cell Physiol 56(3):558–571
Knoop H, Zilliges Y, Lockau W, Steuer R (2010) The metabolic network of Synechocystis Sp. PCC 6803: systemic properties of autotrophic growth. Plant Physiol 154(1):410–422
Kondo K, Ochiai Y, Katayama M, Ikeuchi M (2007) The membrane-associated CpcG2-phycobilisome in Synechocystis: a new photosystem I antenna. Plant Physiol 144(2):1200–1210
Kopečná J, Komenda J, Bučinská L, Sobotka R (2012) Long-term acclimation of the cyanobacterium Synechocystis Sp. PCC 6803 to high light is accompanied by an enhanced production of chlorophyll that is preferentially channeled to trimeric photosystem I. Plant Physiol 160(4):2239–2250
Kwon J-H, Rögner M, Rexroth S (2012) Direct approach for bioprocess optimization in a continuous flat-bed photobioreactor system. J Biotechnol 162(1):156–162
Kwon JH et al (2013) Reduced light-harvesting antenna: consequences on cyanobacterial metabolism and photosynthetic productivity. Algal Res 2(3):188–195
Lagarde D, Vermaas W (1999) The zeaxanthin biosynthesis enzyme β-carotene hydroxylase is involved in myxoxanthophyll synthesis in Synechocystis Sp. PCC 6803. FEBS Lett 454(3):247–251
Lauceri R, Bresciani M, Lami A, Morabito G (2018) Chlorophyll a interference in phycocyanin and allophycocyanin spectrophotometric quantification. J Limnol 77(1):169–177
Lichtenthaler HK (1987) Chlorophylls and carotenoids: pigments of photosynthetic biomembranes. Methods Enzymol 148(C):350–382
Lindblad P et al (2019) CyanoFactory, a European Consortium to develop technologies needed to advance cyanobacteria as chassis for production of chemicals and fuels. Algal Res 41:101510
Liu H et al (2019) Phycobilisomes harbor FNRL in cyanobacteria. mBio 10: e00669–19.
Luimstra VM et al (2018) Blue light reduces photosynthetic efficiency of cyanobacteria through an imbalance between photosystems I and II. Photosynth Res 138(2):177–189
Luimstra VM et al (2019) Exploring the low photosynthetic efficiency of cyanobacteria in blue light using a mutant lacking phycobilisomes. Photosynth Res 141(3):291–301
Ma W, Ogawa T, Shen Y, Mi H (2007) Changes in cyclic and respiratory electron transport by the movement of phycobilisomes in the cyanobacterium Synechocystis Sp. Strain PCC 6803. Biochem Biophys Acta 1767(6):742–749
MacColl R (2004) Allophycocyanin and energy transfer. Biochem Biophys Acta 1657(2–3):73–81
Manodori A, Melis A (1986) Cyanobacterial acclimation to photosystem I or photosystem II light. Plant Physiol 82(1):185–189
McGee D et al (2020) Influence of spectral intensity and quality of LED lighting on photoacclimation, carbon allocation and high-value pigments in microalgae. Photosynth Res 143(1):67–80
Moal G, Lagoutte B (2012) Photo-induced electron transfer from photosystem i to NADP+: characterization and tentative simulation of the in vivo environment. Biochem Biophys Acta 1817(9):1635–1645
Morel A, Bricaud A (1981) Theoretical results concerning light absorption in a discrete medium, and application to specific absorption of phytoplankton. Deep Sea Res Part A 28(11):1375–1393
Münzner P, Voigt J (1992) Blue light regulation of cell division in chlamydomonas reinhardtii. Plant Physiol 99(4):1370–1375
Orr L, Govindjee (2013) Photosynthesis web resources. Photosynth Res 115(2–3):179–214
Porra RJ, Thompson WA, Kriedemann PE (1989) “Determination of Accurate Extinction Coefficients and simultaneous equations for assaying chlorophylls a and b extracted with four different solvents: verification of the concentration of chlorophyll standards by atomic absorption spectroscopy. BBA 975(3):384–394
Rabe AE, Benoit RJ (1962) Mean light intensity—a useful concept in correlating growth rates of dense cultures of microalgae. Biotechnol Bioeng 4(4):377–390
Rakhimberdieva MG, Boichenko VA, Karapetyan NV, Stadnichuk IN (2001) Interaction of phycobilisomes with photosystem II dimers and photosystem I monomers and trimers in the cyanobacterium spirulina platensis. Biochemistry 40(51):15780–15788
Remelli W, Santabarbara S (2018) Excitation and emission wavelength dependence of fluorescence spectra in whole cells of the cyanobacterium Synechocystis Sp. PPC6803: influence on the estimation of photosystem II maximal quantum efficiency. Biochem Biophys Acta 1859(11):1207–1222
Rögner M, Nixon PJ, Diner BA (1990) Purification and characterization of photosystem I and photosystem II core complexes from wild-type and phycocyanin-deficient strains of the cyanobacterium synechocystis PCC 6803. J Biol Chem 265(11):6189–6196
Simis SGH, Kauko HM (2012) In vivo mass-specific absorption spectra of phycobilipigments through selective bleaching. Limnol Oceanogr 10(4):214–226
Singh AK et al (2009) A systems-level analysis of the effects of light quality on the metabolism of a cyanobacterium. Plant Physiol 151(3):1596–1608
Stirbet A, Lazár D, Papageorgiou GC, Govindjee (2019) Chlorophyll a fluorescence in cyanobacteria: relation to photosynthesis. In: Cyanobacteria. Elsevier, pp 79–130
Stramski D, Morel A (1990) Optical properties of photosynthetic picoplankton in different physiological states as affected by growth irradiance. Deep Sea Res A 37(2):245–266
Takaichi S, Maoka T, Masamoto K (2001) Myxoxanthophyll in Synechocystis Sp. PCC 6803 is myxol 2′-dimethyl-fucoside, (3R,2′S)-myxol 2′-(2,4-Di-O-methyl-α-l-fucoside), not rhamnoside. Plant Cell Physiol 42(7):756–762
Thrane J-E et al (2015) Spectrophotometric analysis of pigments: a critical assessment of a high-throughput method for analysis of algal pigment mixtures by spectral deconvolution, ed. Schmitt FG. PLOS ONE 10(9): e0137645
Tian L et al (2011) Site, rate, and mechanism of photoprotective quenching in cyanobacteria. J Am Chem Soc 133(45):18304–18311
Touloupakis E, Cicchi B, Torzillo G (2015) A bioenergetic assessment of photosynthetic growth of Synechocystis Sp. PCC 6803 in continuous cultures. Biotechnol Biofuels 8(1):133
Tsunoyama Y et al (2009) Multiple rieske proteins enable short- and long-term light adaptation of Synechocystis Sp. PCC 6803. J Biol Chem 284(41):27875–27883
Tyystjärvi T et al (2002) Action spectrum of PsbA gene transcription is similar to that of photoinhibition in Synechocystis Sp. PCC 6803. FEBS Lett 516(1–3):167–171
Umena Y, Kawakami K, Shen JR, Kamiya N (2011) Crystal structure of oxygen-evolving photosystem II at a resolution of 1.9Å. Nature 473(7345):55–60
Vajravel S et al (2016) β-Carotene influences the phycobilisome antenna of cyanobacterium Synechocystis Sp. PCC 6803. Photosynth Res 130(1–3):403–415
van Amerongen H, van Grondelle R, Valkunas L (2000) Photosynthetic excitons. World Scientific. https://www.worldscientific.com/worldscibooks/10.1142/3609. Accessed 28 March 2020
von Wobeser EA et al (2011) Concerted changes in gene expression and cell physiology of the cyanobacterium Synechocystis sp. strain PCC 6803 during transitions between nitrogen and light-limited growth. Plant Physiol 155(3):1445–1457
Warren CK, Weedon BCL (1958) 804. Carotenoids and related compounds. Part VII. Synthesis of canthaxanthin and echinenone. J Chem Soc (Resumed) 3986–393.
Westermark S, Steuer R (2016) Toward multiscale models of cyanobacterial growth: a modular approach. Front Bioeng Biotechnol 4(DEC)
Woźniak B et al (2003) Modelling light and photosynthesis in the marine environment. Oceanologia 45(2):171–245
Wright SW, Jeffrey SW, Mantoura RFC (1997) Phytoplankton pigments in oceanography: guidelines to modern methods. UNESCO Publishing, Paris
Yacobi YZ, Köhler J, Leunert F, Gitelson A (2015) Phycocyanin-specific absorption coefficient: eliminating the effect of chlorophylls absorption. Limnol Oceanogr 13(4):e10015
Zakar T et al (2017) Lipid and carotenoid cooperation-driven adaptation to light and temperature stress in Synechocystis Sp PCC6803. Biochem Biophys Acta 1858(5):337–350
Zavřel T et al (2015) Characterization of a model cyanobacterium Synechocystis Sp. PCC 6803 autotrophic growth in a flat-panel photobioreactor. Eng Life Sci 15(1):122–132
Zavřel T, Očenášová P, Červený J (2017) Phenotypic characterization of Synechocystis Sp. PCC 6803 substrains reveals differences in sensitivity to abiotic stress, ed. Jacobs JM. PLOS ONE 12(12): e0189130
Zhang Y et al (2017) An extended PROSPECT: advance in the leaf optical properties model separating total chlorophylls into chlorophyll a and B. Sci Rep 7:6429
Zhao W, Xie J, Xiuling Xu, Zhao J (2015) State transitions and fluorescence quenching in the cyanobacterium Synechocystis PCC 6803 in response to changes in light quality and intensity. J Photochem Photobiol B 142:169–177
Zlenko DV et al (2019) Role of the PB-LOOP in ApcE and phycobilisome core function in cyanobacterium Synechocystis Sp PCC 6803. Biochim Biophys Acta 1860(2):155–166
Acknowledgements
We kindly acknowledge Alberto Conejero for his suggestions on the modeling part.
Funding
DF was supported by an internal grant of Palacký University Olomouc (no. IGA_PrF_2020_028) and DL by the ERDF project “Plants as a tool for sustainable global development” (no. CZ.02.1.01/0.0/0.0/16–019/0000827)."
Author information
Authors and Affiliations
Contributions
DF and JFU conceived the research; DF and JVO-V performed the simulations; DF, DL, and JFU analyzed the data; DF did the experiments, wrote the manuscript with contributions from DL, JVO-V, and JFU, and agreed to serve as the author responsible for contact and ensured communication.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Fuente, D., Lazar, D., Oliver-Villanueva, J.V. et al. Reconstruction of the absorption spectrum of Synechocystis sp. PCC 6803 optical mutants from the in vivo signature of individual pigments. Photosynth Res 147, 75–90 (2021). https://doi.org/10.1007/s11120-020-00799-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-020-00799-8