Abstract
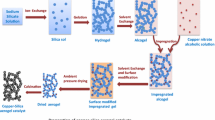
Herein we report on the synthesis and characterization of nano-sized Mn oxide/silica aerogel with low density as a good catalyst toward water oxidation. The composite was synthesized by a simple and low-cost hydrothermal procedure. In the next step, we studied the composite in the presence of cerium(IV) ammonium nitrate and photo-produced Ru(bpy) 3+3 as a water-oxidizing catalyst. The low-density composite is a good Mn-based catalyst with turnover frequencies of ~0.3 and 0.5 (mmol O2/(mol Mn·s)) in the presence of Ru(bpy) 3+3 and cerium(IV) ammonium nitrate, respectively. In addition to the water-oxidizing activities of the composite under different conditions, its self-healing reaction in the presence of cerium(IV) ammonium nitrate was also studied.











Similar content being viewed by others
References
Antczak T, Mrowiec-Bialon J, Bielecki S, Jarzebski AB, Malinowski JJ, Lachowski AI, Galas E (1997) Thermostability and esterification activity in silica aerogel matrix and in organic solvents. Biotechnol Tech 11:9–11
Arnon DI (1959) Conversion of light into chemical energy in photosynthesis. Nature 184:10–20
Begag R, Fesmire JE, Sonn JH (2008) Nonflammable, hydrophobic aerogel composites for cryogenic applications. Thermal Cond 29:323–333
Blakemore JD, Crabtree RH, Brudvig Gary W (2015) Molecular catalysts for water oxidation. Chem Rev 115:2974–13005
Blanchard F, Pommier B, Reymond JP, Teichner SJ (1983) New Fischer-Tropsch catalysts of the aerogel type. Stud. Surf. Sci. Catal 16:395-407. In: Poncelet G, Grange P, Jacobs PA (eds) Studies in surface science and catalysis, preparation of catalysts III. Elsevier, Amsterdam 16:395–407
Blankenship RE (2013) Molecular mechanisms of photosynthesis. Wiley, New York
Bordeepong S, Bhongsuwan D, Pungrassami T, Bhongsuwan T (2011) Characterization of halloysite from Thung Yai District, Nakhon Si Thammarat Province, in Southern Thailand. J Sci Technol 33:599
Chou LY, Liu R, He W, Geh N, Lin Y, Hou EYF, Wang D, Hou HJM (2012) Direct oxygen and hydrogen production by water splitting using a robust bioinspired manganese-oxo oligomer complex/tungsten oxide catalytic system. Int J Hydrogen Energy 37:8889–8896
Fanelli AJ, Burlew JV, Marsh GB (1989) The polymerization of ethylene over titanium tetrachloride supported on alumina aerogels: low-pressure results. J Catal 116:318–324
Glikman TS, Shcheglova IS (1968) Water oxidation by Mn oxide. Kinet Katal 9:461–480
Guenther U, Smirnova I, Neubert RHH (2008) Hydrophilic silica aerogels as dermal drug delivery systems-Dithranol as a model drug. Eur J Pharm Biopharm 69:935–942
Gust D, Moore TA, Moore AL (2009) Solar fuels via artificial photosynthesis. Acc Chem Res 42:1890–1898
He W, Zhao KH, Harvey Hou HJM (2013) Toward solar fuel production using manganese/semiconductor systems to mimic photosynthesis. NanoPhotoBioSciences 1:63–78
Hou HJM (2011) Manganese-based materials inspired by photosynthesis for water-splitting. Materials 4:1693–1704
Huynh M, Bediako DK, Nocera DG (2014) A functionally stable manganese oxide oxygen evolution catalyst in acid. J Am Chem Soc 136:6002–6010
Jiao F, Frei H (2010) Nanostructured manganese oxide clusters supported on mesoporous silica efficient oxygen-evolving catalysts. Chem Commun 46:2920–2922
Julien CM (2003) Lithium intercalated compounds: charge transfer and related properties. Mater Sci Eng R 40:47–102
Kistler SS (1931) Coherent expanded aerogels and jellies. Nature 127:741
Lacroix M, Pajonk G, Teichner SJ (1981) Activation for catalytic reactions of the silica gel by hydrogen spillover. Stud. Surf. Sci. Catal 7: 279–290 In: Seiyama T, Tanabe K (eds) Studies in surface science and catalysis, new horizons in catalysis, vol 7. Elsevier, Amsterdam, pp 279–290
Long JW, Fischer AE, McEvoy TM, Bourg ME, Lytle JC, Rolison DR (2008) Self-limiting electropolymerization en route to ultrathin, conformal polymer coatings for energy storage applications. PMSE Prepr 99:772–773
Matis G, Juillet F, Teichner SJ (1976) Catalytic oxidation of paraffins on nickel oxide-based catalysts. I. Selectivity in the partial oxidation of isobutane and propane. Bull Soc Chim Fr:1633–1636
Nagahara H, Suginouchi T, Hashimoto M (2006) Acoustic properties of nanofoam and its applied air-borne ultrasonic transducers. Proc IEEE Ultrason Symp 3:1541–1544
Najafpour MM, Amini E (2015) Nano-sized Mn oxide on halloysite or high surface area montmorillonite as an efficient catalyst for water oxidation with cerium(IV) ammonium nitrate: supports from natural sources. Dalton Trans 44:15441–15449
Najafpour MM, Nemati Moghaddam A (2012a) Amorphous manganese oxide-coated montmorillonite as an efficient catalyst for water oxidation. New J Chem 36:2514–2519
Najafpour MM, Rahimi F, Aro E-M, Lee C-H, Allakhverdiev SI (2012b) Nano-sized manganese oxides as biomimetic catalysts for water oxidation in artificial photosynthesis: a review. J R Soc Interface 9:2383–2395
Najafpour MM, Nemati Moghaddam A, Sakhac Y (2013a) A simple mathematical model for manganese oxide-coated montmorillonite as a catalyst for water oxidation: from nano to macro sized manganese oxide. Dalton Trans 42:11012–11020
Najafpour MM, Kompany-Zareh M, Zahraei A, Jafarian Sedigh D, Jaccard H, Khoshkam M, Britt RD, Casey W (2013b) Mechanism, decomposition pathway and new evidence for self-healing of manganese oxides as efficient water oxidizing catalysts: new insights. Dalton Trans 42:14603–14611
Najafpour MM, Rahimi F, Fathollahzadeh M, Haghighi B, Holynska B, Tomo T, Allakhverdiev SI (2014a) anostructured manganese oxide/carbon nanotubes, graphene and graphene oxide as water-oxidizing composites in artificial photosynthesis. Dalton Trans 43:10866–10876
Najafpour MM, Abasi M, Tomo T, Allakhverdiev SI (2014b) Nanolayered manganese oxide/C60 composite: a good water-oxidizing catalyst for artificial photosynthetic systems. Dalton Trans 43:12058–12064
Najafpour MM, Abasi M, Tomo T, Allakhverdiev SI (2014c) Mn oxide/nanodiamond composite: a new water-oxidizing catalyst for water oxidation. RSC Adv 4:37613–37619
Najafpour MM, Abbasi Isaloo M, Abasi M, Hołyńska M (2014d) Manganese oxide as a water-oxidizing catalyst: from the bulk to Ångström-scale. New J Chem 38:852–858
Najafpour MM, Fekete M, Jafarian Sedigh D, Aro E-M, Carpentier R, Eaton-Rye JJ, Nishihara H, Shen J-R, Allakhverdiev SI, Spiccia L (2015a) Damage management in water-oxidizing catalysts: from Photosystem II to nano-sized metal oxides. ACS Catal 5:1499–1512
Najafpour MM, Khoshkam M, Jafarian Sedigh D, Zahraei A, Kompany-Zareh M (2015b) Self-healing for nanolayered manganese oxides in the presence of cerium (IV) ammonium nitrate: new findings. New J Chem 39:2547–2550
Najafpour MM, Renger G, Hołyńska M, Nemati Moghaddam A, Aro EM, Carpentier R, Nishihara H, Eaton-Rye JJ, Shen JR, Allakhverdiev SI (2016) Manganese compounds as water-oxidizing catalysts: from the natural water-oxidizing complex to nano-sized manganese oxide structures. Chem Rev. doi:10.1021/acs.chemrev.5b00340
Nakagawa H, Kado R, Obara K, Yano H, Ishikawa O, Hata T, Yokogawa H, Yokoyama M (2007) Equal-spinpairing superfluid phase of 3He in an aerogel acting as an impurity. Phys Rev B 76:172504/1–172504/4
Nocera DG (2012) The Artificial Leaf. Acc Chem Res 45:767–776
Pajonk GM (1991) Aerogel catalysts. Appl Catal 72:217–266
Parent AR, Crabtree RH, Brudvig GW (2013) Comparison of primary oxidants for water-oxidation catalysis. Chem Soc Rev 42:2247–2252
Pierre M, Buisson P, Fache F, Pierre AC (2000) Influence of the drying technique of silica gels on the enzymatic activity of encapsulated lipase. Biocatal Biotransform 18:237–251
Plata DL, Briones YJ, Wolfe RL, Carroll MK, Bakrania SD, Mandel SG, Anderson AM (2004) Aerogelplatform optical sensors for oxygen gas. J Non-Cryst Solids 350:326–335
Power M, Hosticka B, Black E, Daitch C, Norris P (2001) Aerogels as biosensors: viral particle detection by bacteria immobilized on large pore aerogel. J Non-Cryst Solids 285:303–308
Robinson DM, Go YB, Mui M, Gardner G, Zhang Z, Mastrogiovanni D, Garfunkel E, Li J, Greenblatt M, Dismukes GC (2013) Photochemical water oxidation by crystalline polymorphs of manganese oxides: structural requirements for catalysis. J Am Chem Soc 135:3494–3501
Santos A, Ajbary M, Toledo-Fernandez JA, Morales-Florez V, Kherbeche A, Esquivias L (2008) Reactivity of CO2 traps in aerogel-wollastonite composites. J Sol–Gel Sci Technol 48:224–230
Schaefer DW, Keefer KD (1986) Structure of random porous materials: silica aerogel. Phys Rev Lett 56:2199
Soled S (2015) Silica-supported catalysts get a new breath of life. Science 350:1171–1172
Suga M, Akita F, Hirata K, Ueno G, Murakami H, Nakajima Y, Shimizu T, Yamashita K, Yamamoto M, Ago H, Shen JR (2014) Native structure of photosystem II at 1.95 Å resolution viewed by femtosecond X-ray pulses. Nature 517:99–103
Turner JA (1999) A realizable renewable energy future. Science 285:687–689
Umena Y, Kawakami K, Shen JR, Kamiya N (2011) Crystal structure of oxygen-evolving photosystem II at a resolution of 1.9 Å. Nature 473:55–60
Warren BE (1969) X-ray diffraction. Addison-Wesley, Reading, MA
Willey RJ, Lai H, Peri JB (1991) Investigation of iron oxide-chromia-alumina aerogels for the selective catalytic reduction of nitric oxide by ammonia. J Catal 130:319–331
Zhang C, Chen C, Dong H, Shen JR, Dau H, Zhao J (2015) A synthetic Mn4Ca-cluster mimicking the oxygen-evolving center of photosynthesis. Science 348:690–693
Acknowledgments
MMN, SS, and SM are grateful to the Institute for Advanced Studies in Basic Sciences. This work was supported by Grant-in-Aids for Scientific Research from the Ministry of Education of Japan (24370025 and 26220801) to TT. SIA was supported by grant from the Russian Science Foundation (No: 14-14-00039).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Najafpour, M.M., Salimi, S., Madadkhani, S. et al. Nanostructured manganese oxide on silica aerogel: a new catalyst toward water oxidation. Photosynth Res 130, 225–235 (2016). https://doi.org/10.1007/s11120-016-0247-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-016-0247-9