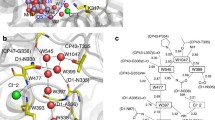
Abstract
The coupling of proton and electron transfers is a key part of the chemistry of photosynthesis. The oxidative side of photosystem II (PS II) in particular seems to involve a number of proton-coupled electron transfer (PCET) steps in the S-state transitions. This mini-review presents an overview of recent studies of PCET model systems in the authors’ laboratory. PCET is defined as a chemical reaction involving concerted transfer of one electron and one proton. These are thus distinguished from stepwise pathways involving initial electron transfer (ET) or initial proton transfer (PT). Hydrogen atom transfer (HAT) reactions are one class of PCET, in which H+ and e − are transferred from one reagent to another: AH+B→A+BH, roughly along the same path. Rate constants for many HAT reactions are found to be well predicted by the thermochemistry of hydrogen transfer and by Marcus Theory. This includes organic HAT reactions and reactions of iron-tris(α-diimine) and manganese-(μ-oxo) complexes. In PS II, HAT has been proposed as the mechanism by which the tyrosine Z radical (YZ) oxidizes the manganese cluster (the oxygen evolving complex, OEC). Another class of PCET reactions involves transfer of H+ and e − in different directions, for instance when the proton and electron acceptors are different reagents, as in AH–B+C+→A–HB++C. The oxidation of YZ by the chlorophyll P680 + has been suggested to occur by this mechanism. Models for this process – the oxidation of phenols with a pendent base – are described. The oxidation of the OEC by YZ could also occur by this second class of PCET reactions, involving an Mn–O–H fragment of the OEC. Initial attempts to model such a process using ruthenium-aquo complexes are described.
Similar content being viewed by others
Abbreviations
- PS II:
-
Photosystem II
- PCET:
-
proton-coupled electron transfer
- ET:
-
electron transfer
- PT:
-
proton transfer
- HAT:
-
hydrogen atom transfer
- OEC:
-
oxygen evolving complex
- YZ or TyrZOH:
-
tyrosine Z, Tyr161 of the D1 subunit (of Thermosynechococcus elongatus)
- YZ or TyrZO:
-
the phenoxyl radical of YZ
- P680+ :
-
oxidized form of the redox-active chlorophyll cluster
- His:
-
histidine
- BDE:
-
bond dissociation enthalpy
- BDFE:
-
bond dissociation free energy
- Fe II (H 2 bim ) :
-
iron(II) tris(2,2′-bi-imidazoline) complex, [Fe(H2bim)3](ClO4)2
- Fe III (Hbim) :
-
the oxidized and singly deprotonated form of Fe II (H 2 bim)
- Fe II (H 2 bip) :
-
iron(II) tris(2,2′-bi-1,4,5,6-tetrahydropyrimidine) complex, [Fe(H2bip)3](ClO4)2
- Fe III (Hbip) :
-
the oxidized and deprotonated form of complex Fe II (H 2 bip)
- TEMPO:
-
2,2,6,6-tetramethyl-1-piperidinyloxy
- TEMPOH:
-
2,2,6,6,-tetramethyl-1-hydroxypiperidine
- HOAr–NH 2 :
-
2-(aminodiphenyl- m ethyl)-4,6-di-tert-butylphenol
- Ar3N:
-
triarylamine
- Cp2Fe:
-
ferrocene
- Ox + :
-
a one-electron oxidant
- tol:
-
tolyl (p-C6H4CH3)
- DFT:
-
density functional theory
- napy:
-
naphthyridine (1,8-diazanaphthalene)
- bpy:
-
2,2′-bipyridine
- binapy:
-
3,3′-binaphthyridine
- trpy:
-
2,2′,6′,2′′-terpyridine
- CV:
-
cyclic voltammogram
References
DV Avila, CE Brown, KU Ingold and J Lusztyk, Solvent effects on the competitive β-scission and hydrogen atom abstraction reactions of the cumyloxyl radical. Resolution of a long-standing problem. J Am Chem Soc 115 (1993) 466-470
MJ Baldwin and VL Pecoraro, Energetics of proton-coupled electron transfer in high-valent Mn2(μ-O)2 systems: models for water oxidation by the oxygen-evolving complex of Photosystem II. J Am Chem Soc 118 (1996) 11325-11326
J Biesiadka, B Bernhard Loll, J Kern, K-D Irrgang and A Zouni, Crystal structure of cyanobacterial photosystem II at 3.2 Å resolution: a closer look at the Mn-cluster. Phys Chem Chem Phys 6 (2004) 4733-4736
RA Binstead, LK Stultz and TJ Meyer, Proton-coupled electron transfer in acetonitrile solution. Irreversible disproportionation of [RuIII(bpy)2 (py)(OH)]2+. Inorg Chem 34 (1995) 546-551
SJ Blanksby and GB Ellison, Bond dissociation energies of organic molecules. Acc Chem Res 36 (2003) 255-263
FG Bordwell, JP Cheng, GZ Ji, AV Satish and X Zhang, Bond dissociation energies in DMSO related to the gas phase values. J Am Chem Soc 113 (1991) 9790-9795
FG Bordwell and JP Cheng, Substituent effects on the stabilities of phenoxyl radicals and the acidities of phenoxyl radical cations. J Am Chem Soc 113 (1991) 1736-1743
D Borgis and JT Hynes, Curve crossing formulation for proton transfer reactions in solution. J Phys Chem 100 (1996) 1118-1128
M Brookhart, B Grant and AF Volpe Jr., [(3,5-(CF3)2C6H3]4B]−[H(OEt2)2]+: a convenient reagent for generation and stabilization of cationic, highly electrophilic organometallic complexes. Organometallics 11 (1992) 3920-3922
CJ Chang, CYM Chang, NH Damrauer and DG Nocera, Proton-coupled electron transfer: a unifying mechanism for biological charge transport, amino acid radical initiation and propagation, and bond making/breaking reactions of water and oxygen. Biochim Biophys Acta Bioenerg 1655 (2004) 13-28
MT Caudle and VL Pecoraro, Thermodynamic viability of hydrogen atom transfer from water coordinated to the oxygen-evolving complex of Photosystem II. J Am Chem Soc 119 (1997) 3415-3416
K Chen, J Hirst, R Camba, CA Bonagura, CD Stout, BK Burgess and FA Armstrong, Atomically defined mechanism for proton transfer to a buried redox centre in a protein. Nature 405 (2000) 814-817
GK Cook and JM Mayer, C–H bond activation by metal oxo species: chromyl chloride oxidations of cyclooctane, isobutane, and toluene. J Am Chem Soc 117 (1995) 7139-7156
RI Cukier, A theory that connects proton-coupled electron-transfer and hydrogen-atom transfer reaction. J Phys Chem B 106 (2002) 1746-1757
RI Cukier and DG Nocera, Proton-coupled electron transfer. Annu Rev Phys Chem 49 (1998) 337-369
Diner BA, Babcock GT (1996) In: Ort DR, and Yocum CF, (eds), Advances in Photosynthesis: The Light Reactions, Kluwer Academic Publication, Dordrecht, The Netherlands, Vol. 4, pp. 213–247
BA Diner, Amino acid residues involved in the coordination and assembly of the manganese cluster of photosystem II. Proton-coupled electron transport of the redox-active tyrosines and its relationship to water oxidation. Biochim Biophys Acta 1503 (2001) 147-163
KM Doll and RG Finke, A compelling experimental test of the hypothesis that enzymes have evolved to enhance quantum mechanical tunneling in hydrogen transfer reactions: the neopentylcobalamin system combined with prior adocobalamin data. Inorg Chem 42 (2003) 4849-4856
T Dziembowska and Z Rozwadowski, Application of the deuterium isotope effect on NMR chemical shift to study proton transfer equilibrium. Current Org Chem 5 (2001) 289-313
L Eberson, Electron Transfer Reactions in Organic Chemistry. Berlin: Springer-Verlag (1987).
DC Eisenberg and JR Norton, Hydrogen-atom transfer reactions of transition metal hydrides. Isr J Chem 31 (1991) 55-61
S Ferguson-Miller and GT Babcock, Heme/copper terminal oxidases. Chem Rev 96 (1996) 2889-2908
KN Ferreira, TM Iverson, K Maghlaoui, J Barber and S Iweta, Architecture of the photosynthetic oxygen-evolving center. Science 303 (2004) 1831-1838
Fischer H, (ed) (1984, 1994) Radical Reaction Rates in Liquids, Landolt-Börnstein New Series, Springer-Verlag, New York, 1984, Vol. 13 and 1994–1997, Vol. 18
KA Gardner, LL Kuehnert and JM Mayer, Hydrogen atom abstraction by permanganate: oxidations of arylalkanes in organic solvents. Inorg Chem 36 (1997) 2069-2078
P Gilli, V Bertolasi, L Pretto, V Ferretti and G Gilli, Covalent versus electrostatic nature of the strong hydrogen bond: discrimination among single, double, and asymmetric single-well hydrogen bonds by variable-temperature X-ray crystallographic methods in diketone enol RAHB systems. J Am Chem Soc 126 (2004) 3845-3855
C Goussias, A Boussac and AW Rutherford, Photosystem II and photosynthetic oxidation of water: an overview. Phil Trans R Soc Lond B 357 (2002) 1369-1381
MS Graige, ML Paddock, JM Bruce, G Feher and MY Okamura, Mechanism of proton-coupled electron transfer for quinone (Q B ) reduction in reaction centers of Rb. Sphaeroides. J Am Chem Soc 118 (1996) 9005-9016
MS Graige, ML Paddock, G Feher and MY Okamura, Observation of the protonated semiquinone intermediate in isolated reaction centers from rhodobacter sphaeroides: implications for the mechanism of electron and proton transfer in proteins. Biochemistry 38 (1999) 11465-11473
B Halliwell and JMC Gutteridge, Free Radicals in Biology and Medicine. New York: Oxford University Press (1999).
S Hammes-Schiffer, Theoretical perspectives on proton-coupled electron transfer reactions. Acc Chem Res 34 (2001) 273-281
S Hammes-Schiffer, Impact of enzyme motion on activity. Biochemistry 41 (2002) 13335-13343
TK Harris and GJ Turner, Structural basis of perturbed pK a values of catalytic groups in enzyme active sites. IUBMB Life 53 (2002) 85-98
K Izutsu, Acid-Base Dissociation Constants in Dipolar Aprotic Solvents. Boston, MA: Blackwell Scientific Publications (1990).
KB Jensen, CJ McKenzie and JZ Pedersen, Indication of a hydrogen-atom abstraction reaction relevant to a mechanistic proposal for the oxygen-evolving complex of Photosystem II. Inorg Chem 40 (2001) 5066-5067
W Junge, M Haumann, R Ahlbrink, A Mulkidjanian and J Clausen, Electrostatics and proton transfer in photosynthetic water oxidation. Phil Trans R Soc Lond B 357 (2002) 1407-1418
Free Radicals. New York: Wiley (1973).
A Koll and P Wolschann, Mannich bases as model compounds for intramolecular hydrogen bonding. Part I. Solid state structures and molecular calculations. Montasch Chem 127 (1996) 475-486
H Kuhne and GW Brudvig, Proton-coupled electron transfer involving tyrosine Z in Photosystem II. J Phys Chem B 106 (2002) 8189-8196
AS Larsen, K Wang, MA Lockwood, GL Rice, TJ Won, S Lovell, M Sadílek, F Tureček and JM Mayer, Hydrocarbon oxidation by Bis-μ-Oxo manganese dimers: electron transfer, hydride transfer, and hydrogen atom transfer mechanisms. J Am Chem Soc 124 (2002) 10112-10123
Z-X Liang and JP Klinman, Structural bases of hydrogen tunneling in enzymes: progress and puzzles. Curr Opin Struct Biol 14 (2004) 648-655
EA Mader, AS Larsen and JM Mayer, Hydrogen atom transfer from Iron(II)-tris[2,2′-bi(tetrahydropyrimidine)] to TEMPO: a negative enthalpy of activation. J Am Chem Soc 126 (2004) 8066-8067
RA Marcus and N Sutin, Electron transfers in chemistry and biology. Biochim Biophys Acta 811 (1985) 265-322
Markle TF, Rhile IJ, Lam OP, DiPasquale AD and Mayer JM (2005) work in progress
JM Mayer, Hydrogen atom abstraction by metal-oxo complexes: understanding the analogy with organic radical reactions. Acc Chem Res 31 (1998) 441-450
Mayer JM (2000) Thermodynamic Influences on C–H Bond Oxidation in Biomimetic Oxidations Catalyzed by Transition Metal Complexes, In: Meunier B., (ed.), Imperial College, Chapter 1
JM Mayer, Proton-coupled electron transfer: a reaction chemist’s view. Annu Rev Phys Chem 55 (2004) 363-390
Meyer TJ and Huynh MHV (2003) The remarkable reactivity of high oxidation state Ruthenium and Osmium Polypyridyl complexes. 42: 8140–8160
BA Moyer and TJ Meyer, Properties of the oxo/aqua system (bpy)2(py)RuO2+/(bpy)2(py)Ru(OH2)2+. Inorg Chem 20 (1981) 436-444
Nakajima H, Nagao H and Tanaka K (1996) Control of the coordination mode of 1,8-naphthyridine ligated to ruthenium(II) bipyridine complexes. J Chem Soc Dalton Trans 1405–1409
SF Nelsen and JR Pladziewicz, Intermolecular electron transfer reactivity determined from cross-rate studies. Acc Chem Res 35 (2002) 247-254
Nemes A and Bakac A (2001) A Disproportionation of Aquachromyl(IV) Ion by Hydrogen Abstraction from coordinated water, Inorg Chem 40: 2720–2704
NIST (2003) NIST Chemistry WebBook, NIST Standard Reference Database Number 69 – March 2003 Release, http://webbook.nist.gov/chemistry/
JHA Nugent, RJ Ball and MCW Evans, Photosynthetic water oxidation: the role of tyrosine radicals. Biochim Biophys Acta Bioenerg 1655 (2004) 217-221
Parker VD, Handoo KL, Roness F and Tilset M (1991) Electrode potentials and the thermodynamics of isodesmic reactions. 113: 7493–7498
RJ Pace and KA Åhrling, Water oxidation in PS II–H atom abstraction revisited. Biochim Biophys Acta Bioenerg 1655 (2004) 172-178
RP Pesavento and WA Donk van der, Tyrosyl radical cofactors. Adv Prot Chem 58 (2001) 317-385
I Pujols-Ayala and BA Barry, Tyrosyl radicals in Photosystem II. Biochim Biophys Acta Bioenerg 1655 (2004) 205-216
DL Reger, TD Wright, CA Little, JJS Lamba and MD Smith, Control of the stereochemical impact of the lone pair in Lead(II) Tris(pyrazolyl)methane complexes. Improved preparation of Na {B[3,5-(CF3)2C6H3]4}. Inorg Chem 40 (2001) 3810-3814
G Renger, Coupling of electron and proton transfer in oxidative water cleavage in photosynthesis. Biochim Biophys Acta Bioenerg 1655 (2004) 195-204
IJ Rhile and JM Mayer, Thermodynamics and kinetics of proton-coupled electron transfer: stepwise vs.concerted pathways. Biochim Biophys Acta 1655 (2004a) 51-58
IJ Rhile and JM Mayer, One-electron oxidation of a hydrogen-bonded phenol occurs by concerted proton-coupled electron transfer. J Am Chem Soc 126 (2004b) 12718-12719
IJ Rhile and JM Mayer, Comment on “how single and bifurcated hydrogen bonds influence proton-migration rate constants, redox, and electronic properties of phenoxyl radicals” by Thomas et al. Angew Chem Int Ed 44 (2005) 2-3
JP Roth, JC Yoder, TJ Won and JM Mayer, Application of the Marcus cross relation to hydrogen atom transfer reactions. Science 294 (2001) 2524-2526
W Ruttinger and GC Dismukes, Synthetic water-oxidation catalysts for artificial photosynthetic water oxidation. Chem Rev 97 (1997) 1-24
M Sjödin, S Styring, B Åkermark, L Sun and L Hammarström, Proton-coupled electron transfer from Tyrosine in a Tyrosine-Ruthenium-tris-bipyridine complex: comparison with TyrosineZ oxidation in Photosystem II. J Am Chem Soc 122 (2000) 3932-3936
M Sjödin, S Styring, H Wolpher, Y Xu, L Sun and L Hammarström, Switching the redox mechanism: models for proton-coupled electron transfer from tyrosine and tryptophan. J Am Chem Soc 122 (2005) 3932-3936
DW Snelgrove, J Lusztyk, JT Banks, P Mulder and KU Ingold, Kinetic solvent effects on hydrogen-atom abstractions: reliable, quantitative predictions via a single empirical equation. J Am Chem Soc 123 (2001) 469-477
JD Soper and JM Mayer, Slow hydrogen atom self-exchange between Os(IV) Anilide and Os(III) Aniline complexes: relationships with electron and proton transfer self exchange. J Am Chem Soc 125 (2003) 12217-12229
SP Sorensen and WH Bruning, Ion pairing effects on electron transfer rate constants by electron spin resonance fast exchange line-width studies. Application to Tri-p-tolylamine and phenothiazine cations. J Am Chem Soc 95 (1973) 2445-2451
J Stubbe, DG Nocera, CS Yee and MCY Chang, Radical initiation in the class I ribonucleotide reductase: long-range proton-coupled electron transfer?. Chem Rev 103 (2003) 2167-2202
J Stubbe and WA Donk van der, Protein radicals in enzyme catalysis. Chem Rev 98 (1998) 705-762
H-F Suen, SW Wilson, M Pomerantz and JL Walsh, Photosubstitution reactions of terpyridine complexes of ruthenium(II). Inorg Chem 28 (1989) 786-791
N Sutin, Theory of electron transfer reactions: insights and hindsights. Prog Inorg Chem 30 (1983) 441
JM Tedder, Which factors determine the reactivity and regioselectivity of free radical substitution and addition reactions?. Ang Chem Int Ed Engl 21 (1982) 401-410
Thomas F, Olivier JO, Jamet H, Hamman S, Saint-Aman E, Duboc C and Pierre JL (2004) How single and bifurcated hydrogen bonds influence proton-migration rate constants, redox, and electronic properties of phenoxyl radicals. Angew Chem Int Ed 43: 594–597; Angew Chem 116: 604–607
RP Thummel, F Lefoulon, D Cantu and R Mahadevan, Annelated derivatives of 2-(2′-pyridyl)-1,8-naphthyridine and 2,2′-bi-1,8-naphthyridine. J Org Chem 49 (1984) 2208-2212
C Tommos and GT Babcock, Oxygen production in nature: a light-driven metalloradical enzyme process. Acc Chem Res 31 (1998) 18-25
K Wang and JM Mayer, Oxidation of hydrocarbons by [(phen)2Mn(μ-O)2Mn(phen)2]3+ via hydrogen atom abstraction. J Am Chem Soc 119 (1997) 1470-1471
SC Weatherly, IV Yang, PA Armistead and HH Thorp, Proton-coupled electron transfer in guanine oxidation: effects of isotope solvent, and chemical modification. J Phys Chem B 107 (2003) 372-378
C Tommos, Electron, proton and hydrogen-atom transfers in photosynthetic water oxidation. Phil Trans R Soc Lond B 357 (2002) 1383-1394
JS Vrettos and GW Brudvig, Water oxidation chemistry of photosystem II. Phil Trans R Soc Lond B 357 (2002) 1395-1405
R Zong, F Naud, C Segal, J Burke, F Wu and R Thummel, Design and study of bi[1,8]naphthyridine ligands as potential photooxidation mediators in Ru(II) polypyridyl aquo complexes. Inorg Chem 43 (2004) 6195-6202
Author information
Authors and Affiliations
Corresponding author
Additional information
An erratum to this article can be found at http://dx.doi.org/10.1007/s11120-006-9053-0
Rights and permissions
About this article
Cite this article
Mayer, J.M., Rhile, I.J., Larsen, F.B. et al. Models for Proton-coupled Electron Transfer in Photosystem II. Photosynth Res 87, 3–20 (2006). https://doi.org/10.1007/s11120-005-8164-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-005-8164-3