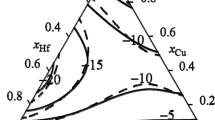
The partial and integral mixing enthalpies of Cu-Yb melts were first determined at 1453 K in the composition range 0 < xCu <0.7 by isoperibolic calorimetry. The Cu–Yb melts were established to form with the release of a small amount of heat: minimum mixing enthalpy ∆H = –9.7 ± 0.8 (at xCu = 0.7), which agrees with the published data for these melts in the composition range 0 <xYb < 0.3 at 1453 K and for other Cu–Ln systems. The model of ideal associated solutions was used to optimize and calculate all thermodynamic properties (Gibbs energies, enthalpies, and entropies of formation) for melts, intermetallic compounds, and associates in the Cu–Yb system. The calculated activities of components in the melts of this system exhibited moderate negative deviations from ideal solutions. The calculations with the ideal associated solution model also showed that \( \varDelta {\overline{H}}_{\textrm{Yb}}^{\infty } \) increased insignificantly and \( \varDelta {\overline{H}}_{\textrm{Cu}}^{\infty } \) more substantially in the Cu–Yb system with higher temperatures. The ideal associated solution model was applied to calculate temperature–composition dependences of the Gibbs energies, enthalpies, and entropies of formation for the melts and intermetallics to determine the coordinates of the liquidus curve in the studied phase diagram. The calculated and experimental data were in good agreement. Full information on the thermodynamic properties of all phases and phase equilibria in the Cu–Yb alloys was obtained.








Similar content being viewed by others
References
N.I. Usenko, M.I. Ivanov, V.M. Petiuh, and V.T. Witusiewicz, “Thermochemistry of binary liquid alloys of copper with barium and lanthanide metals (europium, dysprosium and ytterbium),” J. Alloys Compd., 190, No. 1, 149–155 (1993).
P.R. Subramanian and D.E. Laughtin, “The Cu–Yb (copper–ytterbium) system,” J. Phase Equilib., 9, No. 3a, 398–403 (1988).
L.G. Zhang, L.B. Liu, H.S. Liu, and Z.P. Jin, “Thermodynamic assessment of Cu–Eu and Cu–Yb system,” Calphad, 31, 264–268 (2007).
V.S. Sudavtsova, M.O. Shevchenko, M.I. Ivanov, V.G. Kudin, and N.V. Podoprygora, “Thermodynamic properties and phase equilibria of Nd–Ni alloys,” Powder Metall. Met. Ceram., 58, No. 9–10, 581–590 (2020).
A.T. Dinsdale, “SGTE data for pure elements,” Calphad, 15, Issue 4, 319–427 (1991), DOI: https://doi.org/10.1016/0364-5916(91)90030-N.
I. Prigogine and R. Defay, Chemical Thermodynamics [in German], Leipzig (1962).
M.A. Shevchenko, V.G. Kudin, and V.S. Sudavtsova, “Correctness of the thermodynamic properties of binary alloys calculated with the ideal associated solution model,” in: Current Issues of Physical Materials Science (Collected Papers) [in Russian], Inst. Probl. Materialoved. NAN Ukrainy, Kyiv (2012), Issue 21, pp. 67–77.
M.O. Shevchenko, V.V. Berezutski, M.I. Ivanov, V.G. Kudin, and V.S. Sudavtsova, “Thermodynamic properties of alloys of the binary Al–Sm, Sm–Sn and ternary Al–Sm–Sn systems,” J. Phase Equilib. Diffus., 36, No. 1, 39–52 (2015), DOI: https://doi.org/10.1007/s11669-014-0353-3.
V.S. Sudavtsova, M.A. Shevchenko, M.I. Ivanov, V.V. Berezutski, and V.G. Kudin, “Thermodynamic properties of liquid alloys of copper with lanthanum,” Zr. Fiz. Khim., 91, No. 6, 937–944 (2017), DOI: https://doi.org/10.7868/S0044453717060279.
M.O. Shevchenko, M.I. Ivanov, V.V. Berezutski, and V.S. Sudavtsova, “Thermodynamic properties of alloys in the binary Ca–Ge system,” J. Phase Equilib. Diffus., 36, No. 6, 554–572 (2015), DOI: https://doi.org/10.1007/s11669-015-0408-0.
A. Iandelli and A. Palenzona, “The ytterbium–copper system,” J. Less-Comm. Met., 25, 333–335 (1971).
K. Fitzner and O.J. Kleppa, “Thermochemistry of binary alloys of transition metals: The systems Cu–Ce, Me–Pr, and Me–Nd (Me—Cu, Ag, Au),” Metall. Mater. Trans. A, 25A, 1495–1500 (1994).
K. Fitzner and O.J. Kleppa, “Thermochemistry of binary alloys of transition metals: The systems Me–Gd, Me–Ho, and Me–Lu (Me—Cu, Ag, and Au),” Metall. Mater. Trans. A, 28A, No. 1, 187–190 (1997).
S.V. Meschel and O.J. Kleppa, “Thermochemistry of some of binary alloys of copper with lanthanide metals by high temperature direct synthesis calorimetry,” J. Alloys Compd., 388, No. 1, 91–97 (2005).
C. Colinet, “The thermodynamic properties of rare earth metallic systems,” J. Alloys Compd., 225, 409–422 (1995).
M.A. Turchanin and V.V. Nikolaenko, “Energetics of forming liquid alloys of copper with rare-earth metals,” Teplofiz. Vys. Temp., 27, No. 6, 1090–1096 (1989).
M. Ivanov, V. Berezutski, N. Usenko, and N. Kotova, “Enthalpies of mixing in binary liquid alloys of lutetium with 3d-metals,” Int. J. Mater. Res., 108, No. 1, 29–35 (2017), doi:https://doi.org/10.3139/146.111445.
V.S. Sudavtsova, V.A. Makara, and V.G. Kudin, Thermodynamics of Metallurgy and Welding Melts [in Russian], Logos, Kyiv (2005), Part 3, p. 216.
W.B. Pearson, The Crystal Chemistry and Physics of Metals and Alloys, Wiley-Interscience, New York (1972).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Poroshkova Metallurgiya, Vol. 61, Nos. 5–6 (545), pp. 114–125, 2022.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Dudnyk, A.S., Sudavtsova, V.S., Romanova, L.O. et al. Thermodynamic Properties of Alloys and Phase Equilibria in the Cu–Yb System. Powder Metall Met Ceram 61, 350–359 (2022). https://doi.org/10.1007/s11106-022-00325-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11106-022-00325-6