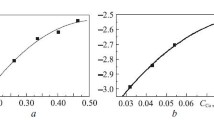
The solubility of molybdenum in copper–silicon melts and the kinetics of growth of Mo5Si3 and MoSi2 layers on the molybdenum–melt interface at 1200°C are studied. At 0.20–0.36 at. parts Si, the solubility of molybdenum in the melts is described by the following equation: lgX Mo = –2.587 + + (3.282)X Si. The dependence of the growth rate constants of the Mo5Si3 layer on the activity of silicon in the 9–15 wt.% Si melt is established: kMo–Si = –0.3806 + (65.191) aSi. It is shown that two phases form at 20 wt.% Si: Mo5Si3 and MoSi2. Their thicknesses are 29.8 ± 2.9 μm and 25.7 ± ± 2.8 μm, respectively. The time dependence of the Mo5Si3 layer growth rate in Cu–Si melts is established to obey a parabolic law. The melt composition in three-phase equilibrium Mo–Mo5Si– melt is found to be (at. parts): XSi = 0.147; XMo = 7.86∙10–3; the rest is copper. At 35 wt.% Si, only the MoSi2 phase is formed at the interface.






Similar content being viewed by others
References
K. Sedlatschek and H. J. Stadler, “Ein Beitragzum Oberflachenschutzvon Molibden bei hohen Temperaturen,” Planseeberichte fur Pulvermetallurgie, 9, 39–43 (1961).
Encyclopedia of Inorganic Materials [in Russian], Vol. 2, Glav. Red. Ukr. Sovet. Éntsiklopedii, Kiev (1977), p. 813.
V. V. Skorokhod, M. M. Churakov, and V. P. Titov, “The growth of disilicide layers on the interface: W/Mo-melt (Cu–Si) of near-equilibrium composition,” J. Mater. Sci. Lett., 16, 1689–1690 (1997).
V. V. Skorokhod, V. P. Titov, and M. M. Churakov, “Interaction of Mo and W silicides with Cu–Si melts,” Ukr. Khim. Zh., 63, No. 5–6, 85–89 (1997).
T. B. Massalski, Binary Alloy Phase Diagrams, Vol. 2, ASM, Metals Park, Ohio (1986), p. 1631.
M. Hansen and K. Anderko, Constitution of Binary Alloys, McGraw-Hill, New York (1958),
T. B. Massalski, Binary Alloy Phase Diagrams, Vol. 1, ASM, Metals Park, Ohio (1986), p. 1100.
G. Ottaviani, “Metallurgical aspects of the formation of silicides,” Thin Sol. Films, 140, No. 1, 3–21 (1986).
M. S. Tsirlin and I. P. Bakhtina, “Texture of diffusion silicide layers on tungsten and molybdenum,” Izv. AN SSSR. Met., No. 2, 141–146 (1989).
V. M. Krivoruchko, V. V. Sagalovich, A. P. Patokin, and V. P. Podtykan, “On participation of Mo–Si system components in reaction diffusion,” Izv. AN SSSR. Neorg. Mater., 7, No. 4, 610–613 (1971).
V. I. Arkharov, “On the kinetics of reaction diffusion in systems with several intermediate phases,” Fiz. Met. Metalloved., 8, No. 2, 193–204 (1959).
K. P. Gurov, B. A. Kartashkin, and Yu. É. Ugaste, Interdiffusion in Multiphase Metal Systems [in Russian], Nauka, Moscow (1981), p. 350.
V. I. Dybkov, Solid-Phase Chemical Kinetics and Reaction Diffusion [in Ukrainian], Inst. Probl. Materialoved. NAN Ukrainy, Kiev (2002), p. 314.
O. Kubaschewski and C. B. Alcock, Metallurgical Thermochemistry, Pergamon Press, New York (1972).
V. N. Yeremenko, G. M. Lukashenko, and V. R. Sidorko, “Thermodynamic properties of vanadium, chromium and manganese silicides at elevated temperatures,” Rev. Intern. Haut. Temp. Refract., 12, No. 3, 237–240 (1975).
S. I. Sidorenko, Yu. M. Makogon, and S. M. Voloshko, “Materials science of thin-film nanostructures,” Diffusion and Reactions [in Ukrainian], Naukova Dumka, Kiev (2000).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Poroshkovaya Metallurgiya, Vol. 51, No. 3–4 (484), pp. 117–125, 2012.
Rights and permissions
About this article
Cite this article
Skorokhod, V.V., Titov, V.P., Golovkova, M.E. et al. Interaction of molybdenum with copper–silicon melts. Powder Metall Met Ceram 51, 222–228 (2012). https://doi.org/10.1007/s11106-012-9421-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11106-012-9421-6