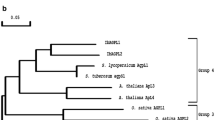
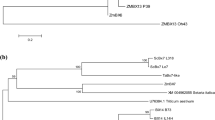
Abstract
Cassava is an important staple food crop, feeding 600 million people worldwide, which produce cyanogenic glycosides. Cyanogenic glycosides in cassava are known to act as a deterrent for herbivores as well as serve as a mobile source of reduced nitrogen. Cassava is also equipped with a cyanide detoxification pathway, mediated by β-cyanoalanine synthase (β-CAS) which converts cyanide into asparagine. β-CAS, belonging to the Bsas family of enzymes, is multi functional and shares sequence homology with cysteine synthase (CS). Using rapid amplification of cDNA end-polymerase chain reaction (RACE-PCR), two cDNA sequences were isolated from cassava. The two clones named MANes;BsasA (accession no. EU350583) and MANes;BsasB (accession no. HQ257219), showed high homology to known β-CAS enzymes (80% and 75% amino acid similarity to Arabidopsis and 76% and 82% similarity to spinach, respectively). The kinetic properties of the two clones were characterized in a Escherichia coli NK3 mutant strain which lacks activity for any of the Bsas proteins. Kinetic studies showed that MANes;BsasB is a β-CAS with a CAS/CS activity ratio of 72 while MANes;BsasA is a CS showing bifunctional capabilities and with a CAS/CS activity ratio of 11. The isolation of cassava β-CAS and CS genes reported here paves the way for their utilization in genetically enhancing the cyanide detoxification potential of cassava and/or increase of the essential amino acid cysteine, which has been found to be low in nutritionally compromised individuals.





Similar content being viewed by others
References
Aregheore EM, Agunbiade OO (1991) The toxic effects of cassava (Manihot esculenta Crantz) diets on humans: a review. Vet Hum Toxicol 33:274–275
Bradford M (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochem 72:248–254
Chenna R, Sugawara H, Koike T, Lopez R, Gibson TJ, Higgins DG, Thompson JD (2003) Multiple sequence alignment with the Clustal series of programs. Nucleic Acids Res 31:3497–3500
Dereeper A, Guignon V, Blanc G, Audic S, Buffet S, Chevenet F, Dufayard JF, Guindon S, Lefort V, Lescot M, Claverie JM, Gascuel O (2008) Phylogeny.fr: robust phylogenetic analysis for the non-specialist. Nucleic Acids Res 36:465–469
Elias M, Sudhakaran P, Nambisan B (1997) Purification and characterization of β-cyanoalanine synthase from cassava tissues. Phytochemistry 46:469–472
Emanuelsson O, Nielsen H, Brunak S, Von Heijne G (2000) Predicting subcellular localization of proteins based on their N-terminal amino acid sequence. J Mol Biol 300:1005–1016
Gaitonde MK (1967) A spectrophotometric method for the direct determination of cysteine in the presence of other naturally occurring amino acids. Biochem J 104:627–633
Hasegawa R, Tomoko T, Yuichiro T, Yohji E (1994) Presence of β-cyanoalanine synthase in unimbibed dry seeds and its activation by ethylene during pregermination. Physiol Plant 91:141–146
Hatzfeld Y, Maruyama A, Schmidt A, Noji M, Ishizawa K, Saito K (2000) β-Cyanoalanine synthase is a mitochondrial cysteine synthase-like protein in spinach and Arabidopsis. Plant Physiol 123:1163–1171
Hegg C, Kruse C, Jost R, Gutensohn M, Ruppert T, Wirtz M, Hell R (2008) Analysis of the Arabidopsis O-acetylserine(thiol)lyase gene family demonstrates compartment-specific differences in the regulation of cysteine synthesis. Plant Cell 20:168–185
Hell R, Bork C, Bogdanova N, Frolov I, Hauschild R (1994) Isolation and characterization of two cDNAs encoding for compartment specific isoforms of O-acetylserine (thiol) lyase from Arabidopsis thaliana. FEBS Lett 351:257–262
Hesse H, Höfgen R (1998) Isolation of cDNAs encoding cytosolic (accession no. AF044172) and plastidic (accession no. AF044173) Cys synthase isoforms from Solanum tuberosum. Plant Physiol 116:1604
Hughes J, Carvalho F, Hughes M (1994) Purification, characterization and cloning of hydroxynitrile lyase from cassava (Manihot esculenta Crantz). Arch Biochem Biophys 311:496–502
Hulanicka MD, Garret C, Jagura-Burdzy G, Kredich NM (1986) Cloning and characterization of the cysAMK region of Salmonella typhimurium. J Bacteriol 168:322–327
Ikegami F, Murakoshi I (1994) Enzymic synthesis of non-protein β-substituted alanines and some higher homologues in plants. Phytochemistry 35:1089–1104
Ikegami F, Kaneko M, Kamiyama H, Murakoshi I (1988) Purification and characterization of cysteine synthases from Citrullus vulgaris. Phytochemistry 27:697–701
Ikegami F, Takayama K, Murakoshi I (1989a) Purification and properties of β-cyano-l-alanine synthase from Lathyrus latifolius. Phytochemistry 27:3385–3389
Ikegami F, Takayama K, Kurihara T, Horiuchi S, Tajima C, Shira R, Murakoshi I (1989b) Purification and properties of β-cyano-l-alanine synthase from Vicia angustifolia. Phytochemistry 28:2285–2291
Jost R, Berkowitz O, Wirtz M, Hopkins L, Hawkesford MJ, Hell R (2000) Genomic and functional characterization of the OAS gene family encoding O-acetylserine(thiol)lyases, enzymes catalyzing the final step in cysteine biosynthesis in Arabidopsis thaliana. Gene 253:237–247
Kibbe WA (2007) OligoCalc: an online oligonucleotide properties calculator. Nucleic Acids Res 35 (webserver issue):May 25
Koch B, Nielsen VS, Halkier BA, Olsen CE, Møller BL (1992) The biosynthesis of cyanogenic glycosides in seedlings of cassava (Manihot esculenta Crantz). Arch Biochem Biophys 292:141–150
López C, Jorge V, Piégu B, Mba C, Cortes D, Restrepo S, Soto M, Laudié M, Berger C, Cooke R, Delseny M, Tohme J, Verdier V (2004) A unigene catalogue of 5700 expressed genes in cassava. Plant Mol Biol 56:541–554
Lunn JE, Droux M, Martin J, Douce R (1990) Localization of ATP sulfurylase and Oacetylserine(thiol)lyase in spinach leaves. Plant Physiol 94:1345–1352
Maruyama A, Ishizawa K, Takaqi T (2000) Purification and characterization of beta-cyanoalanine synthase and cysteine synthases from potato tubers: are beta-cyanoalanine synthase and mitochondrial cysteine synthase same enzyme? Plant Cell Physiol 41:200–208
Maruyama A, Saito K, Ishizawa K (2001) β-Cyanoalanine synthase and cysteine synthase from potato: molecular cloning, biochemical characterization, and spatial and hormonal regulation. Plant Mol Biol 46:749–760
McMahon J, White W, Sayre R (1995) Cyanogenesis in cassava (Manihot esculenta Crantz). J Exp Bot 46:731–741
Morant AV, Jørgensen K, Jørgensen B, Dam W, Olsen CE, Møller BL, Bak S (2007) Lessons learned from metabolic engineering of cyanogenic glucosides. Metabolomics 3:383–398
Nartey F (1969) Studies on cassava, Manihot utilissima: II. Biosynthesis of asparagine-14C from 14C-labelled hydrogen cyanide and its relations with cyanogenesis. Physiol Plant 22:1085–1096
Poulton J (1990) Cyanogenesis in plants. Plant Physiol 94:401–405
Ramanujam T, Indira P (1984) Effect of girdling on the distribution of total carbohydrates and hydrocyanic acid in cassava. Indian J Plant Physiol 27:355–360
Rozen S, Skaletsky H (1998) Primer3. Code available at http://www.genome.wi.mit.edu/genome_software/other/primer3.html. Accessed 21 Sept 2010
Selmar D (1993) Transport of cyanogenic glucosides: linustatin uptake in Hevea cotyledons. Planta 191:191–199
Siritunga D, Sayre R (2003) Generation of cyanogen-free transgenic cassava. Planta 217:367–373
Siritunga D, Sayre R (2004) Engineering cyanogen synthesis and turnover in cassava (Manihot esculenta). Plant Mol Biol 56:661–669
Small I, Peeters N, Legeai F, Lurin C (2004) Predotar: a tool for rapidly screening proteomes for N-terminal targeting sequences. Proteomics 4:1581–1590
Taiz L, Zeiger E (2002) Plant physiology, 3rd edn. Sinauer Associates, Inc., Sunderland, pp 272–275
Vetter J (2000) Plant cyanogenic glycosides. Toxicon 38:11–36
Warrillow A, Hawkesford M (1998) Separation, subcellular location and influence of sulphur nutrition on isoforms of cysteine synthase in spinach. J Exp Bot 49:1625–1636
Warrillow A, Hawkesford M (2000) Cysteine synthase (O-acetylserine (thiol) lyase) substrate specificities classify the mitochondrial isoform as a cyanoalanine synthase. J Exp Bot 51:985–993
Watanabe M, Kusano M, Oikawa A, Kukushima A, Noji M, Saito K (2007) Physiological roles of the β-substituted alanine synthase gene family in Arabidopsis. Plant Physiol 146:310–320
Watanabe M, Mochida K, Kato T, Tabata S, Yoshimoto N, Noji M, Saito K (2008) Comparative genomics and reverse genetics analysis reveal indispensable functions of the serine acetyltransferase gene family in Arabidopsis. Plant Cell 20:2484–2496
White W, McMahon J, Sayre R (1994) Regulation of cyanogenesis in cassava. Acta Hortic 375:69–77
Wirtz M, Droux M, Hell R (2004) O-acetylserine (thiol) lyase: an enigmatic enzyme of plant Cys biosynthesis revisited in Arabidopsis thaliana. J Exp Bot 55:1785–1798
Yamaguchi Y, Nakamura T, Kusano T, Sano H (2000) Three Arabidopsis genes encoding proteins with differential activities for cysteine synthase and beta-cyanoalanine synthase. Plant Cell Physiol 41:465–476
Acknowledgements
The authors would like to thank Professor Kazuki Saito (Chiba University, Chiba, Japan) for providing the NK3 strain and the A. thaliana β-CAS full-length clone used in this study. Special thanks also to Dr. Timothy Porch (USDA-Tropical Agriculture Research Station, Mayaguez, PR, USA) and Dr. Argelia Lawrence (Arkansas State University, Jonesboro, AR, USA) for their critical review of this manuscript. This study was funded by grant to DS from the Research Initiation Program of the National Science Foundation (award number IOS 0641084).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Marrero-Degro, J., Marcano-Velázquez, J. & Siritunga, D. Isolation and Characterization of Novel β-Cyanoalanine Synthase and Cysteine Synthase Genes from Cassava. Plant Mol Biol Rep 29, 514–524 (2011). https://doi.org/10.1007/s11105-010-0255-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11105-010-0255-4