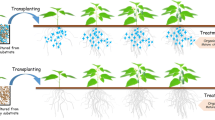
Abstract
Background and Aims
Rhizosphere microorganisms play an important role in promoting plant growth and maintaining soil health. Bacillus altitudinis LZP02 (LZP02) is an efficient plant growth-promoting rhizobacterium that interacts closely with rice radicals. However, the mechanisms mediating the rhizosphere interactions of beneficial strains of microorganisms in native soil are mostly unclear.
Methods
This study analysed the rhizosphere-enriched taxonomic and functional properties of the rhizosphere-associated microbiome and determined how they were affected by LZP02 using metagenome sequencing and ITS sequencing after the inoculation of LZP02 in soil. And microorganisms from the rice rhizosphere were identified and cultivated using high-throughput sequencing.
Results
LZP02 stimulated some beneficial bacteria, such as Novosphingobium, Acidovorax, Sphingomonas, and Devosia, to the rhizosphere. The enrichment of functional attributes, nitrogen metabolism and bacterial chemotaxis demonstrated an increase in active microbe-microbe interactions in the rhizosphere. This study showed that LZP02 stimulated plant growth in three ways: (1) by increasing the relative abundance of most rhizosphere-enriched genus; (2) by increasing the relative abundance of the functional attributes involved in microbe-microbe interactions; and (3) by enriching functional components that are essential for plant fitness, such as nitrogen fixation and bacterial chemotaxis in the rhizosphere microbiome. Finally, inoculation of enriched single strains or synthetic consortia isolated from the radical-associated microbiome using high-throughput cultivation could improve plant growth.
Conclusion
This study proposes a new mechanism by which LZP02 influences the nitrogen metabolism of rhizosphere microorganisms and bacterial chemotaxis for rice growth by reshaping the rhizosphere microbiome.






Similar content being viewed by others
Data availability
Raw data used in this study are available in the NCBI under the bioproject, accession no. PRJNA918597 and accession no. PRJNA919054. The raw data of high-throughput are available in the GSA: CRA007016.
Abbreviations
- PGPR:
-
Plant growth-promoting rhizobacteria
- BCAAs:
-
Branched chain amino acids
- LZP02:
-
Bacillus altitudinis LZP02
- LB:
-
Luria-Bertani broth
- CFU:
-
Colony forming unit
- PCR:
-
Polymerase chain reaction
- ASV:
-
Amplicon sequencing variant
- RDP:
-
Ribosomal database project
- ORFs:
-
Open reading frames
- KEGG:
-
Encyclopedia of genes and genomes
- TSB:
-
Trypticase soy broth
- OD:
-
Optical density
- PCoA:
-
Principal coordinate analysis
- LDA:
-
Linear discriminant analysis
- LEfSe:
-
Linear discriminate effect size
References
Allard-Massicotte R, Tessier L, Lécuyer F, Lakshmanan V, Lucier J, Garneau D, Caudwell L (2016) Bacillus subtilis early colonization of Arabidopsis thaliana roots involves multiple chemotaxis receptors. mBio 7: e01664–01616. https://doi.org/10.1128/mBio.01664-16
Bai B, Liu WD, Qiu XY, Zhang J, Zhang JY, Bai Y (2022) The root microbiome: community assembly and its contributions to plant fitness. J Integr Plant Biol 64:230–243. https://doi.org/10.1111/jipb.13226
Bastian M, Heymann S, Jacomy M (2009) Gephi: an open source software for exploring and manipulating networks. ICWSM 8:361–362. https://doi.org/10.1609/icwsm.v3i1.13937
Berlanga-Clavero MV, Molina-Santiago C, Caraballo-Rodriguez AM, Petras D, Diaz-Martinez L, Perez-Garcia A, de Vicente A (2022) Bacillus subtilis biofilm matrix components target seed oil bodies to promote growth and anti-fungal resistance in melon. Nat Microbiol 7:1001–1015. https://doi.org/10.1038/s41564-022-01134-8
Bilal S, Shahzad R, Khan A, Kang S, Imran Q, Al-Harrasi A, Yun B (2018) Endophytic microbial consortia of phytohormones-producing fungus Paecilomyces formosus LHL10 and bacteria Sphingomonas sp. LK11 to glycine max L. regulates physio-hormonal changes to attenuate aluminum and zinc stresses. Front Plant Sci 9: 1273. https://doi.org/10.3389/fpls.2018.01273
Bolyen E, Rideout JR, Dillon MR, Bokulich N, Abnet CC, Al-Ghalith GA, Alexander H (2019) Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat Biotechnol 37:852–857. https://doi.org/10.1038/s41587-019-0209-9
Bulgarelli D, Schlaeppi K, Spaepen S, Loren V, van Themaat E, Schulze-Lefert P (2013) Structure and functions of the bacterial microbiota of plants. Annu Rev Plant Biol 64:807–838. https://doi.org/10.1146/annurev-arplant-050312-120106
Caporaso JG, Kuczynski J, Stombaugh J, Bittinger K, Bushman FD, Costello EK, Fierer N (2010) QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7:335–336. https://doi.org/10.1038/nmeth.f.303
Chen C, Wang M, Zhu J, Tang Y, Zhang H, Zhao Q, Jing M (2022) Long-term effect of epigenetic modification in plant-microbe interactions: modification of DNA methylation induced by plant growth-promoting bacteria mediates promotion process. Microbiome 10:36. https://doi.org/10.1186/s40168-022-01236-9
Chen Q, Ting J, Liu Y, Liu H, Wang G (2019) Recently duplicated sesterterpene (C25) gene clusters in Arabidopsis thaliana modulate root microbiota. Sci China Life Sci 62:947–958. https://doi.org/10.1007/s11427-019-9521-2
Chhetri G, Kim I, Kang MC, Kim J, So Y, Seo T (2022) Devosia rhizoryzae sp. nov., and Devosia oryziradicis sp. nov., novel plant growth promoting members of the genus Devosia, isolated from the rhizosphere of rice plants. J Microbiol 60:1–10. https://doi.org/10.1007/s12275-022-1474-8
Corral-Lugo A, De la Torre J, Matilla M, Fernández M, Morel B, Espinosa-Urgel M, Krell T (2016) Assessment of the contribution of chemoreceptor-based signalling to biofilm formation. Environ Microbiol 18:3355–3372. https://doi.org/10.1111/1462-2920.13170
Delgado-Baquerizo M, Maestre FT, Reich PB, Jeffries TC, Gaitan JJ, Encinar D, Berdugo M (2016) Microbial diversity drives multifunctionality in terrestrial ecosystems. Nat Commun 7:10541. https://doi.org/10.1038/ncomms10541
Deng XH, Zhang N, Shen ZZ, Zhu CZ, Liu HJ, Xu ZH, Li R (2021) Soil microbiome manipulation triggers direct and possible indirect suppression against Ralstonia solanacearum and Fusarium oxysporum. Npj Biofilms Microbiomes 7:33. https://doi.org/10.1038/s41522-021-00204-9
Edgar RC, Haas BJ, Clemente JC, Quince C, Knight R (2011) UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 27:2194–2200. https://doi.org/10.1093/bioinformatics/btr381
Gao M, Xiong C, Gao C, Tsui CKM, Wang MM, Zhou X, Zhang AM (2021) Disease-induced changes in plant microbiome assembly and functional adaptation. Microbiome 9:187. https://doi.org/10.1186/s40168-021-01138-2
Greetatorn T, Hashimoto S, Sarapat S, Tittabutr P, Boonkerd N, Uchiumi T, Teaumroong N (2019) Empowering rice seedling growth by endophytic Bradyrhizobium sp. SUTN 9–2. Lett Appl Microbiol 68:258–266. https://doi.org/10.1111/lam.13114
Gu YA, Banerjee S, Dini-Andreote F, Xu YC, Shen QR, Jousset A, Wei Z (2022) Small changes in rhizosphere microbiome composition predict disease outcomes earlier than pathogen density variations. ISME J 16:2448–2456. https://doi.org/10.1038/s41396-022-01290-z
Gupta V, Kumar GN, Buch A (2020) Colonization by multi-potential Pseudomonas aeruginosa P4 stimulates peanut (Arachis hypogaea L.) growth, defence physiology and root system functioning to benefit the root-rhizobacterial interface. J Plant Physiol 248:153144. https://doi.org/10.1016/j.jplph.2020.153144
Ha-Tran D, Nguyen T, Hung S, Huang E, Huang C (2021) Roles of plant growth-promoting rhizobacteria (PGPR) in stimulating salinity stress defense in plants: a review. Int J Mol Sci 22:3154. https://doi.org/10.3390/ijms22063154
Han Q, Ma Q, Chen Y, Tian B, Xu LX, Bai Y, Chen WF (2020) Variation in rhizosphere microbial communities and its association with the symbiotic efficiency of rhizobia in soybean. ISME J 14:1915–1928. https://doi.org/10.1038/s41396-020-0648-9
Hartmann A, Rothballer M, Schmid M (2008) Lorenz Hiltner, a pioneer in rhizosphere microbial ecology and soil bacteriology research. Plant Soil 312:7–14. https://doi.org/10.1007/s11104-007-9514-z
Hassani MA, Duran P, Hacquard S (2018) Microbial interactions within the plant holobiont. Microbiome 6:58. https://doi.org/10.1186/s40168-018-0445-0
Hida A, Oku S, Kawasaki T, Nakashimada Y, Tajima T, Kato J (2015) Identification of the McpA and McpM genes, encoding methyl-accepting proteins involved in amino acid and l-malate chemotaxis, and involvement of McpM-mediated chemotaxis in plant infection by Ralstonia pseudosolanacearum (formerly Ralstonia solanacearum phylotypes I and III). Appl Environ Microbiol 81:7420–7430. https://doi.org/10.1128/aem.01870-15
Hu J, Yang T, Friman V, Kowalchuk G, Hautier Y, Li M, Wei Z (2021) Introduction of probiotic bacterial consortia promotes plant growth via impacts on the resident rhizosphere microbiome. Proc R Soc B 288:20211396. https://doi.org/10.1098/rspb.2021.1396
Jiao H, Xu W, Chen W, Hu Y, Tian R, Wang Z (2022) Bacillus altitudinis complete genome sequence data of LZP02, a bacterium from the rice rhizosphere, for studying the promotion of plant growth. MPMI 35:428–431. https://doi.org/10.1094/mpmi-01-22-0012-a
Khumairah F, Setiawati M, Fitriatin B, Simarmata T, Alfaraj S, Ansari M, Enshasy H (2022) Halotolerant plant growth-promoting rhizobacteria isolated from saline soil improve nitrogen fixation and alleviate salt stress in rice plants. Front Microbiol 13:905210. https://doi.org/10.3389/fmicb.2022.905210
Klueglein N, Kappler A (2013) Abiotic oxidation of Fe(II) by reactive nitrogen species in cultures of the nitrate-reducing Fe(II) oxidizer Acidovorax sp BoFeN1 - questioning the existence of enzymatic Fe(II) oxidation. Geobiology 11:180–190. https://doi.org/10.1111/gbi.12019
Krishnan R, Menon RR, Likhitha BHJ, Tanaka N, Krishnamurthi S, Rameshkumar N (2017) Novosphingobium pokkalii sp nov, a novel rhizosphere-associated bacterium with plant beneficial properties isolated from saline-tolerant pokkali rice. Res Microbiol 168:113–121. https://doi.org/10.1016/j.resmic.2016.09.001
Li Z, Bai X, Jiao S, Li Y, Li P, Yang Y, Zhang H (2021) A simplified synthetic community rescues Astragalus mongholicus from root rot disease by activating plant-induced systemic resistance. Microbiome 9:217. https://doi.org/10.1186/s40168-021-01169-9
Liu H, Wang Z, Xu W, Zeng J, Li L, Li S, Gao Z (2020) Bacillus pumilus LZP02 promotes rice root growth by improving carbohydrate metabolism and phenylpropanoid biosynthesis. MPMI 33:1222–1231. https://doi.org/10.1094/mpmi-04-20-0106-r
Liu XY, Shi Y, Kong LY, Tong LH, Cao HX, Zhou H, Lv YZ (2022) Long-term application of bio-compost increased soil microbial community diversity and altered its composition and network. Microorganisms 10:462. https://doi.org/10.3390/microorganisms10020462
Luo L, Zhao C, Wang E, Raza A, Yin C (2022) Bacillus amyloliquefaciens as an excellent agent for biofertilizer and biocontrol in agriculture: an overview for its mechanisms. Microbiol Res 259:127016. https://doi.org/10.1016/J.MICRES.2022.127016
Martins DS, Reis VM, Schultz N, Alves BJR, Boddey RM (2020) Both the contribution of soil nitrogen and of biological N2 fixation to sugarcane can increase with the inoculation of diazotrophic bacteria. Plant Soil 454:155–169. https://doi.org/10.1007/s11104-020-04621-1
Matthew, T., Agler, Jonas, Ruhe, Samuel, Kroll (2016) Microbial hub taxa link host and abiotic factors to plant microbiome variation. Plos Biol 14: e1002352 https://doi.org/10.1371/journal.pbio.1002352
Mendes R, Kruijt M, de Bruijn I, Dekkers E, van der Voort M, Schneider JHM, Piceno YM (2011) Deciphering the rhizosphere microbiome for disease-suppressive bacteria. Science 332:1097–1100. https://doi.org/10.1126/science.1203980
Mir M, Hameeda B, Quadriya H, Kumar B, Ilyas N, Kee Zuan A, El Enshasy H (2021) Multifarious indigenous diazotrophic rhizobacteria of rice (Oryza sativa L.) rhizosphere and their effect on plant growth promotion. Front Nutr 8: 781764. https://doi.org/10.3389/fnut.2021.781764
Molina-Favero C, Creus CM, Simontacchi M, Puntarulo S, Lamattina L (2008) Aerobic nitric oxide production by Azospirillum brasilense Sp245 and its influence on root architecture in tomato. MPMI 21:1001–1009. https://doi.org/10.1094/MPMI-21-7-1001
Niu B, Paulson JN, Zheng X, Kolter R (2017) Simplified and representative bacterial community of maize roots. Proc Natl Acad Sci U S A 114:E2450–E2459. https://doi.org/10.1073/pnas.1616148114
Papatheodorou EM, Monokrousos N, Angelina E, Stamou GP (2021) Robustness of rhizosphere microbial communities of L. sativa originated from soils of different legacy after inoculation with Plant Growth Promoting Rhizobacteria. Appl Soil Ecol 167: 104028. https://doi.org/10.1016/j.apsoil.2021.104028
Perez-Rodriguez MM, Piccoli P, Anzuay MS, Baraldi R, Neri L, Taurian T, Ureche MAL (2020) Native bacteria isolated from roots and rhizosphere of Solanum lycopersicum L. increase tomato seedling growth under a reduced fertilization regime. Sci Rep 10: 15642. https://doi.org/10.1038/s41598-020-72507-4
Peterson SB, Dunn AK, Klimowicz AK, Handelsman J (2006) Peptidoglycan from Bacillus cereus mediates commensalism with rhizosphere bacteria from the Cytophaga-Flavobacterium group. Appl Environ Microbiol 72:5421–5427. https://doi.org/10.1128/aem.02928-05
Rognes T, Flouri T, Nichols B, Quince C, Mahé F (2016) VSEARCH: a versatile open source tool for metagenomics. PeerJ Inc 4: e2584. https://doi.org/10.7717/PEERJ.2584
Scharf B, Hynes M, Alexandre G (2016) Chemotaxis signaling systems in model beneficial plant-bacteria associations. Plant Mol Biol 90:549–559. https://doi.org/10.1007/s11103-016-0432-4
Segura A, Udaondo Z, Molina L (2021) PahT regulates carbon fluxes in Novosphingobium sp. HR1a and influences its survival in soil and rhizospheres. Environ Microbiol 23:2969–2991. https://doi.org/10.1111/1462-2920.15509
Sharma R, Gal L, Garmyn D, Bru D, Sharma S, Piveteau P (2022) Plant growth promoting bacterial consortium induces shifts in indigenous soil bacterial communities and controls Listeria monocytogenes in rhizospheres of Cajanus cajan and Festuca arundinacea. Microb Ecol 84:106–121. https://doi.org/10.1007/s00248-021-01837-1
Stopnisek N, Shade A (2021) Persistent microbiome members in the common bean rhizosphere: an integrated analysis of space, time, and plant genotype. ISME J 15:2708–2722. https://doi.org/10.1038/s41396-021-00955-5
Sun X, Xu Z, Xie J, Hesselberg-Thomsen V, Tan T, Zheng D, Strube ML (2022) Bacillus velezensis stimulates resident rhizosphere Pseudomonas stutzeri for plant health through metabolic interactions. ISME J 16:774–787. https://doi.org/10.1038/s41396-021-01125-3
Tao C, Li R, Xiong W, Shen Z, Liu S, Wang B, Ruan Y (2020) Bio-organic fertilizers stimulate indigenous soil Pseudomonas populations to enhance plant disease suppression. Microbiome 8:137. https://doi.org/10.1186/s40168-020-00892-z
Trivedi P, Leach JE, Tringe SG, Sa TM, Singh BK (2020) Plant-microbiome interactions: from community assembly to plant health. Nat Rev Microbiol 18:607–621. https://doi.org/10.1038/s41579-020-0412-1
Vessey JK (2003) Plant growth promoting rhizobacteria as biofertilizers. Plant Soil 255:571–586. https://doi.org/10.1023/a:1026037216893
Wang X, Wang M, Xie X, Guo S, Zhou Y, X Z (2020) An amplification-selection model for quantified rhizosphere microbiota assembly. Sci Bull 65: 983-986https://doi.org/10.1016/j.scib.2020.03.005
Wang Z, Chen Z, Kowalchuk G, Xu Z, Fu X, Kuramae E (2021) Succession of the resident soil microbial community in response to periodic inoculations. Appl Environ Microbiol 87:e00046-e21. https://doi.org/10.1128/aem.00046-21
Yahya M, ul Islam E, Rasul M, Farooq I, Mahreen N, Tawab A, Irfan M (2021) Differential root exudation and architecture for improved growth of wheat mediated by phosphate solubilizing bacteria. Front Microbiol 12: 744094 https://doi.org/10.3389/fmicb.2021.744094
Yan S, Ren T, Wan Mahari W, Feng H, Xu C, Yun F, Waiho K (2022) Soil carbon supplementation: improvement of root-surrounding soil bacterial communities, sugar and starch content in tobacco (N. tabacum). Sci Total Environ 802: 149835. https://doi.org/10.1016/j.scitotenv.2021.149835
Zhang J, Hussain S, Zhao F, Zhu L, Cao X, Yu S, Jin Q (2018) Effects of Azospirillum brasilense and Pseudomonas fluorescens on nitrogen transformation and enzyme activity in the rice rhizosphere. J Soils Sediments 18:1453–1465. https://doi.org/10.1007/s11368-017-1861-7
Zhang J, Liu Y, Zhang N, Hu B, Jin T, Xu H, Qin Y (2019) NRT1.1B is associated with root microbiota composition and nitrogen use in field-grown rice. Nat Biotechnol 37:676–684. https://doi.org/10.1038/s41587-019-0104-4
Zhang JY, Liu YX, Guo XX, Qin Y, Garrido-Oter R, Schulze-Lefert P, Bai Y (2021) High-throughput cultivation and identification of bacteria from the plant root microbiota. Nat Protoc 16:988–1012. https://doi.org/10.1038/s41596-020-00444-7
Zhang YZ, Xu J, Riera N, Jin T, Li JY, Wang N (2017) Huanglongbing impairs the rhizosphere-to-rhizoplane enrichment process of the citrus root-associated microbiome. Microbiome 5:97. https://doi.org/10.1186/s40168-017-0304-4
Zhao ML, Zhao J, Yuan J, Hale L, Wen T, Huang QW, Vivanco JM (2021) Root exudates drive soil-microbe-nutrient feedbacks in response to plant growth. Plant Cell Environ 44:613–628. https://doi.org/10.1111/pce.13928
Acknowledgements
The authors give thanks to Wang Zhigang and Chen Wenjing for their excellent technical assistance. Sequencing support was provided by Shanghai Majorbio Bio-pharm Technology Co., Ltd and Sangon Biotech (Shanghai) Co., Ltd.
Funding
This work was funded by the Outstanding Youth Fund project of Heilongjiang Province in China (JQ2023D001), the Key Research and Development Projects of Heilongjiang in China (GA21B007).
Author information
Authors and Affiliations
Contributions
W.N.C. wrote and edited the paper. W.N.C., W.J.C., and Z.G.W. conceived the overall study design. W.J.C. and Y.L.H. performed soil and radical sampling. W.N.C. conducted the experiments and prepared the figures and bioinformatics analysis. W.J.C. and Y.L.H. provided critical guidance in the analysis. Z.G.W. was involved in revising the manuscript and approving the final version.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Responsible Editor: Wen-Hao Zhang.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chang, W., Chen, W., Hu, Y. et al. Bacillus altitudinis LZP02 improves rice growth by reshaping the rhizosphere microbiome. Plant Soil 498, 279–294 (2024). https://doi.org/10.1007/s11104-023-06435-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-023-06435-3