Abstract
Purpose
Arbuscular mycorrhizal fungi that regenerate rapidly after disturbance play a significant role in vegetation resilience. We employed the fungi inhabiting a coastal dune ecosystem to characterize their strategies for survival and regeneration after disturbance.
Methods
Root-soil samples were collected from the seaward and landward slopes that differ in disturbance severity. The native grass Miscanthus sinensis (donor plants) were grown in the soil samples for two months, and from a half of the plants, disturbance-tolerant fungi that regenerate from spores and extraradical hyphae (soil propagule-mediated regenerators, SP) and those that regenerate from the colonized roots (root-direct regenerators, RD) were trapped separately with new seedlings (assessment plants). The other half of the donor plants were further grown without destruction for four months together with assessment plants, during which the fungi trapped by the plants were categorized as disturbance-sensitive slow regenerators (SL). DNA was extracted from the assessment plants for identification based on LSU rDNA sequences.
Results
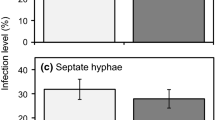
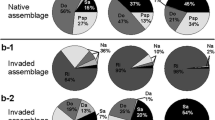
All the fungi that occurred widely in the seaward samples belonged to the Glomeraceae and Paraglomeraceae and preferred the RD strategy to the SP strategy. In the landward samples diverse taxa that employed diverse strategies occurred, and among them, Gigasporaceae fungi showed a distinct preference for the SP strategy.
Conclusion
These observations suggest that not only soil-borne propagules but also colonized roots play a key role in the rapid regeneration of the fungi after disturbance, providing a new insight into the life-history strategies of AM fungi in frequently disturbed environments.






Similar content being viewed by others
Data availability
The sequence reads have been deposited in the Sequence Read Archive of National Center for Biotechnology Information under the accession number PRJNA949820.
References
Atunnisa R, Ezawa T (2019) Nestedness in arbuscular mycorrhizal fungal communities in a volcanic ecosystem: Selection of disturbance-tolerant fungi along an elevation gradient. Microbes Environ 34:327–333. https://doi.org/10.1264/jsme2.ME19073
Avio L, Pellegrino E, Bonari E, Giovannetti M (2006) Functional diversity of arbuscular mycorrhizal fungal isolates in relation to extraradical mycelial networks. New Phytol 172:347–357. https://doi.org/10.1111/j.1469-8137.2006.01839.x
Biermann B, Linderman RG (1983) Use of vesicular-arbuscular mycorrhizal roots, intraradical vesicles and extraradical vesicles as inoculum. New Phytol 95:97–105. https://doi.org/10.1111/j.1469-8137.1983.tb03472.x
Chagnon PL, Bradley RL, Maherali H, Klironomos JN (2013) A trait-based framework to understand life history of mycorrhizal fungi. Trends Plant Sci 18:484–491. https://doi.org/10.1016/j.tplants.2013.05.001
Corkidi L, Rincón E (1997) Arbuscular mycorrhizae in a tropical sand dune ecosystem on the Gulf of Mexico: II. Effects of arbuscular mycorrhizal fungi on the growth of species distributed in different early successional stages. Mycorrhiza 7:17–23. https://doi.org/10.1007/s005720050158
Crisafulli CM, Swanson FJ, Clarkson BD (2015) Volcano Ecology: Disturbance characteristics and assembly of biological communities. In: The Encyclopedia of Volcanoes 1265–1284. https://doi.org/10.1016/B978-0-12-385938-9.00073-0
de la Providencia IE, Fernández F, Declerck S (2007) Hyphal healing mechanism in the arbuscular mycorrhizal fungi Scutellospora reticulata and Glomus clarum differs in response to severe physical stress. FEMS Microbiol Lett 268:120–125. https://doi.org/10.1111/j.1574-6968.2006.00572.x
Declerck S, D’or D, Cranenbrouck S, Le Boulengé E (2001) Modelling the sporulation dynamics of arbuscular mycorrhizal fungi in monoxenic culture. Mycorrhiza 11:225–230. https://doi.org/10.1007/s005720100124
del Moral R (2010) The importance of long-term studies of ecosystem reassembly after the eruption of the Kasatochi Island volcano. Arctic Antarct Alp Res 42:335–341. https://doi.org/10.1657/1938-4246-42.3.335
Delavaux CS, Ramos RJ, Sturmer SL, Bever JD (2022) Environmental identification of arbuscular mycorrhizal fungi using the LSU rDNA gene region: an expanded database and improved pipeline. Mycorrhiza 32:145–153. https://doi.org/10.1007/s00572-022-01068-3
Delgado-Fernandez I, Davidson-Arnott R (2011) Meso-scale aeolian sediment input to coastal dunes: The nature of aeolian transport events. Geomorphology 126:217–232. https://doi.org/10.1016/j.geomorph.2010.11.005
Dumbrell AJ, Nelson M, Helgason T, Dytham C, Fitter AH (2010) Idiosyncrasy and overdominance in the structure of natural communities of arbuscular mycorrhizal fungi: Is there a role for stochastic processes? J Ecol 98:419–428. https://doi.org/10.1111/j.1365-2745.2009.01622.x
Ezawa T, Saito K (2018) How do arbuscular mycorrhizal fungi handle phosphate? New insight into fine-tuning of phosphate metabolism. New Phytol 220:1116–1121. https://doi.org/10.1111/nph.15187
Fujiyoshi M, Kagawa A, Nakatsubo T, Masuzawa T (2006) Effects of arbuscular mycorrhizal fungi and soil developmental stages on herbaceous plants growing in the early stage of primary succession on Mount Fuji. Ecol Res 21:278–284. https://doi.org/10.1007/s11284-005-0117-y
Grime JP (1977) Evidence for the existence of three primary strategies in plants and its relevance to ecological and evolutionary theory. Am Nat 111:1169–1194. https://doi.org/10.1086/283244
Hart MM, Reader RJ (2005) The role of the external mycelium in early colonization for three arbuscular mycorrhizal fungal species with different colonization strategies. Pedobiologia 49:269–279. https://doi.org/10.1016/j.pedobi.2004.12.001
Hestrin R, Hammer EC, Mueller CW, Lehmann J (2019) Synergies between mycorrhizal fungi and soil microbial communities increase plant nitrogen acquisition. Commun Biol 2:233. https://doi.org/10.1038/s42003-019-0481-8
Ho J, Tumkaya T, Aryal S, Choi H, Claridge-Chang A (2019) Moving beyond P values: data analysis with estimation graphics. Nat Methods 16:565–566. https://doi.org/10.1038/s41592-019-0470-3
Hodge A, Fitter AH (2010) Substantial nitrogen acquisition by arbuscular mycorrhizal fungi from organic material has implications for N cycling. Proc Natl Acad Sci U S A 107:13754–13759. https://doi.org/10.1073/pnas.1005874107
Hosseini M, Jacob J, Gonzalez O, Alegre S, Ritsema C, Geissen V (2016) Effect of fire frequency on runoff, soil erosion, and loss of organic matter at the micro-plot scale in north-central Portugal. Geoderma 269:126–137. https://doi.org/10.1016/j.geoderma.2016.02.004
Hungr O, Leroueil S, Picarelli L (2014) The Varnes classification of landslide types, an update. Landslides 11:167–194. https://doi.org/10.1007/s10346-013-0436-y
Jansa J, Mozafar A, Anken T, Ruh R, Sanders IR, Frossard E (2002) Diversity and structure of AMF communities as affected by tillage in a temperate soil. Mycorrhiza 12:225–234. https://doi.org/10.1007/s00572-002-0163-z
Jasper DA, Abbott LK, Robson AD (1989) Soil disturbance reduces the infectivity of external hyphae of vesicular-arbuscular mycorrhizal fungi. New Phytol 112:93–99. https://doi.org/10.1111/j.1469-8137.1989.tb00313.x
Jasper DA, Abbott LK, Robson AD (1993) The survival of infective hyphae of vesicular–arbuscular mycorrhizal fungi in dry soil: An interaction with sporulation. New Phytol 124:473–479. https://doi.org/10.1111/j.1469-8137.1993.tb03838.x
Katoh K, Rozewicki J, Yamada KD (2019) MAFFT online service: multiple sequence alignment, interactive sequence choice and visualization. Brief Bioinform 20:1160–1166. https://doi.org/10.1093/bib/bbx108
Kawahara A, Ezawa T (2013) Characterization of arbuscular mycorrhizal fungal communities with respect to zonal vegetation in a coastal dune ecosystem. Oecologia 173:533–543. https://doi.org/10.1007/s00442-013-2622-y
Kikuci Y, Hijikata N, Ohtomo R, Handa Y, Kawaguchi M, Saito K, Masuta C, Ezawa T (2016) Aquaporin-mediated long-distance polyphosphate translocation directed towards the host in arbuscular mycorrhizal symbiosis: Application of virus-induced gene silencing. New Phytol 211:1202–1208. https://doi.org/10.1111/nph.14016
Klironomos JN, Hart MM (2002) Colonization of roots by arbuscular mycorrhizal fungi using different sources of inoculum. Mycorrhiza 12:181–184. https://doi.org/10.1007/s00572-002-0169-6
Kurothe RS, Kumar G, Singh R, Singh HB, Tiwari SP, Vishwakarma AK, Sena DR, Pande VC (2014) Effect of tillage and cropping systems on runoff, soil loss and crop yields under semiarid rainfed agriculture in India. Soil Tillage Res 140:126–134. https://doi.org/10.1016/j.still.2014.03.005
Lanfranco L, Fiorilli V, Gutjahr C (2018) Partner communication and role of nutrients in the arbuscular mycorrhizal symbiosis. New Phytol 220:1031–1046. https://doi.org/10.1111/nph.15230
Lekberg Y, Schnoor T, Kjøller R, Gibbons SM, Hansen LH, Al-Soud WA, Sørensen SJ, Rosendahl S (2012) 454-sequencing reveals stochastic local reassembly and high disturbance tolerance within arbuscular mycorrhizal fungal communities. J Ecol 100:151–160. https://doi.org/10.1111/j.1365-2745.2011.01894.x
Neary DG (2019) Forest soil disturbance: Implications of factors contributing to the wildland fire nexus. Soil Sci Soc Am J 83:S228–S243. https://doi.org/10.2136/sssaj2018.12.0471
Niwa R, Koyama T, Sato T, Adachi K, Tawaraya K, Sato S, Hirakawa H, Yoshida S, Ezawa T (2018) Dissection of niche competition between introduced and indigenous arbuscular mycorrhizal fungi with respect to soybean yield responses. Sci Rep 8:2–5. https://doi.org/10.1038/s41598-018-25701-4
Oehl F, Sieverding E, Ineichen K, Mader P, Wiemken A, Boller T (2009) Distinct sporulation dynamics of arbuscular mycorrhizal fungal communities from different agroecosystems in long-term microcosms. Agric Ecosyst Environ 134:257–268. https://doi.org/10.1016/j.agee.2009.07.008
Oksanen J, Simpson G, Blanchet F, Kindt R, Legendre P, Minchin P, O'Hara R, Solymos P, Stevens M, Szoecs E, Wagner H, Barbour M, Bedward M, Bolker B, Borcard D, Carvalho G, Chirico M, De Caceres M, Durand S, Evangelista H, FitzJohn R, Friendly M, Furneaux B, Hannigan G, Hill M, Lahti L, McGlinn D, Ouellette M, Ribeiro Cunha E, Smith T, Stier A, Ter Braak C, Weedon J (2022) vegan: Community Ecology Package. R package version 2.6-4. http://CRAN.R-project.org/package=vegan
Rey F (2003) Influence of vegetation distribution on sediment yield in forested marly gullies. CATENA 50:549–562. https://doi.org/10.1016/S0341-8162(02)00121-2
Schnoor TK, Lekberg Y, Rosendahl S, Olsson PA (2011) Mechanical soil disturbance as a determinant of arbuscular mycorrhizal fungal communities in semi-natural grassland. Mycorrhiza 21:211–220. https://doi.org/10.1007/s00572-010-0325-3
Silvestro D, Michalak I (2012) raxmlGUI: a graphical front-end for RAxML. Org Divers Evol 12:335–337. https://doi.org/10.1007/s13127-011-0056-0
Smith VH (2003) Eutrophication of freshwater and coastal marine ecosystems: A global problem. Environ Sci Pollut Res 10:126–139. https://doi.org/10.1065/espr2002.12.142.10.18637/jss.v059.c03
Smith SE, Read DJ (2008) Mycorrhizal Symbiosis, 3rd edn. Academic Press, London
Suescún D, Villegas JC, León JD, Flórez CP, García-Leoz V, Correa-Londoño GA (2017) Vegetation cover and rainfall seasonality impact nutrient loss via runoff and erosion in the Colombian Andes. Reg Environ Chang 17:827–839. https://doi.org/10.1007/s10113-016-1071-7
Tanaka S, Hashimoto K, Kobayashi Y, Yano K, Maeda T, Kameoka H, Ezawa T, Saito K, Akiyama K, Kawaguchi M (2022) Asymbiotic mass production of the arbuscular mycorrhizal fungus Rhizophagus clarus. Communications Biology 5:43. https://doi.org/10.1038/s42003-021-02967-5
Van Eynde E, Dondeyne S, Isabirye M, Deckers J, Poesen J (2017) Impact of landslides on soil characteristics: Implications for estimating their age. CATENA 157:173–179. https://doi.org/10.1016/j.catena.2017.05.003
Varela-Cervero S, López-García Á, Barea JM, Azcón-Aguilar C (2016) Differences in the composition of arbuscular mycorrhizal fungal communities promoted by different propagule forms from a Mediterranean shrubland. Mycorrhiza 26:489–496. https://doi.org/10.1007/s00572-016-0687-2
Weber SE, Diez JM, Andrews LV, Goulden ML, Aronson EL, Allen MF (2019) Responses of arbuscular mycorrhizal fungi to multiple coinciding global change drivers. Fungal Ecol 40:62–71. https://doi.org/10.1016/j.funeco.2018.11.008
Acknowledgements
This work was conducted at the open research facility of Hokkaido Agricultural Research Center, NARO. We also acknowledge Ministry of Education, Culture, Sports, Science and Technology, Japan for the scholarship to AC.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception, design, and writing the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Responsible Editor: Jan Jansa.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cahyaningtyas, A., Ezawa, T. Disturbance tolerance of arbuscular mycorrhizal fungi: characterization of life-history strategies along a disturbance gradient in a coastal dune ecosystem. Plant Soil 495, 535–549 (2024). https://doi.org/10.1007/s11104-023-06337-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-023-06337-4