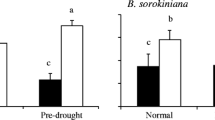
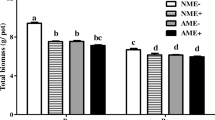
Abstract
Background and aims
According to the nitrogen-disease hypothesis, plant diseases will become more serious with the aggravation of global nitrogen (N) deposition. Numerous studies have indicated that Epichloë endophytes can enhance host plant resistance to pathogens. It is unclear how the competitive ability of endophyte-infected (EI) over endophyte-free (EF) plants changes under the interference of N deposition and plant disease.
Methods
In this study, Achnatherum sibiricum, native to the Inner Mongolia steppe of China, was used as experimental material. We experimentally manipulated N addition and pathogen inoculation and examined the growth and competition between EI and EF A. sibiricum.
Results
The results showed that EI plants had a greater competitive advantage than EF plants under low N conditions, and this advantage decreased with the N supply. When high N and pathogens were both present, pathogen inoculation reversed the adverse effects of high N supply on the competitive advantage of EI A. sibiricum. Epichloë endophytes not only reduced the disease of EI plants but also reduced the disease of neighboring EF plants. Meanwhile, Epichloë endophytes changed the response of the host disease to N. The disease index of EF plants increased with increasing leaf N content, while that of EI plants did not change.
Conclusion
This study highlights that under the dual factors of N deposition and pathogen infection, endophytic fungi improve the competitive ability of host plants. Our results show that considering both biotic and abiotic factors is crucial for predicting the dominance of plant-fungal symbionts in the community.





Similar content being viewed by others

Data availability
Data supporting the findings of this study are available from the corresponding author on the reasonable request.
References
Ampt EA, Francioli D, van Ruijven J, Gomes SIF, Maciá-Vicente JG, Termorshuizen AJ, Bakker LM, Mommer L (2022) Deciphering the interactions between plant species and their main fungal root pathogens in mixed grassland communities. J Ecol 110:3039–3052. https://doi.org/10.1111/1365-2745.14012
Anderson NA (1982) The genetics and pathology of Rhizoctonia solani. Annu Rev Phytopathol 20:329–347. https://doi.org/10.1146/annurev.py.20.090182.001553
Arachevaleta M, Bacon CW, Hoveland CS, Radcliffe DE (1989) Effect of the tall fescue endophyte on plant response to environmental stress. Agron J 81:83–90. https://doi.org/10.2134/agronj1989.00021962008100010015x
Bai W, Guo D, Tian QY, Liu NN, Cheng WX, Li LH, Zhang WH (2015) Differential responses of grasses and forbs led to marked reduction in below-ground productivity in temperate steppe following chronic N deposition. J Ecol 103:1570–1579. https://doi.org/10.1111/1365-2745.12468
Bastías DA, Gianoli E, Gundel PE (2021) Fungal endophytes can eliminate the plant growth–defence trade-off. New Phytol 230:2105–2113. https://doi.org/10.1111/nph.17335
Borer ET, Seabloom EW, Gruner DS et al (2014) Herbivores and nutrients control grassland plant diversity via light limitation. Nature 508:517–520. https://doi.org/10.1038/nature13144
Brem D, Leuchtmann A (2002) Intraspecific competition of endophyte infected vs uninfected plants of two woodland grass species. Oikos 96:281–290. https://doi.org/10.1034/j.1600-0706.2002.960210.x
Buckley H, Young CA, Charlton ND, Hendricks WQ, Haley B, Nagabhyru P, Rudgers JA (2019) Leaf endophytes mediate fertilizer effects on plant yield and traits in northern oat grass (Trisetum spicatum). Plant Soil 434:425–440. https://doi.org/10.1007/s11104-018-3848-6
Cao H, Li X, Dong XN (1998) Generation of broad-spectrum disease resistance by overexpression of an essential regulatory gene in systemic acquired resistance. Proc Natl Acad Sci 95:6531–6536. https://doi.org/10.1073/pnas.95.11.6531
Cappelli SL, Pichon NA, Kempel A, Allan E (2020) Sick plants in grassland communities: a growth-defense trade-off is the main driver of fungal pathogen abundance. Ecol Lett 23:1349–1359. https://doi.org/10.1111/ele.13537
Chen YX, Zhang FS, Tang L, Zheng Y, Li YJ, Christie P, Li L (2007) Wheat powdery mildew and foliar N concentrations as influenced by N fertilization and belowground interactions with intercropped faba bean. Plant Soil 291:1–13. https://doi.org/10.1007/s11104-006-9161-9
Chen LY, Cheng CW, Liang JY (2015) Effect of esterification condensation on the Folin-Ciocalteu method for the quantitative measurement of total phenols. Food Chem 170:10–15. https://doi.org/10.1016/j.foodchem.2014.08.038
Chen ZJ, Jin YY, Yao X, Chen TX, Wei XK, Li CJ, White JF, Nan ZB (2020) Fungal endophyte improves survival of Lolium perenne in low fertility soils by increasing root growth, metabolic activity and absorption of nutrients. Plant Soil 452:185–206. https://doi.org/10.1007/s11104-020-04556-7
Cheplick GP, Harrichandra AP, Liu AN (2014) Competitive outcomes depend on host genotype, but not clavicipitaceous fungal endophytes, in Lolium perenne (Poaceae). Am J Bot 101:2068–2078. https://doi.org/10.3732/ajb.1400264
Cheynier V, Comte G, Davies KM, Lattanzio V, Martens S (2013) Plant phenolics: recent advances on their biosynthesis, genetics, and ecophysiology. Plant Physiol Biochem 72:1–20. https://doi.org/10.1016/j.plaphy.2013.05.009
Cibils-Stewart X, Mace WJ, Popay AJ, Lattanzi FA, Hartley SE, Hall CR, Powell JR, Johnson SN (2022) Interactions between silicon and alkaloid defences in endophyte-infected grasses and the consequences for a folivore. Funct Ecol 36:249–261. https://doi.org/10.1111/1365-2435.13916
Clarke BB, White JF, Hurley RH, Torres MS, Sun S, Huff DR (2006) Endophyte-mediated suppression of dollar spot disease in fine fescues. Plant Dis 90:994–998. https://doi.org/10.1094/pd-90-0994
Clay K, Schardl C (2002) Evolutionary origins and ecological consequences of endophyte symbiosis with grasses. Am Nat 160:S99–S127. https://doi.org/10.1086/342161
Crawford MS, Schlägel UE, May F, Wurst S, Grimm V, Jeltsch F (2021) While shoot herbivores reduce, root herbivores increase nutrient enrichment’s impact on diversity in a grassland model. Ecology 102:e03333. https://doi.org/10.1002/ecy.3333
Decunta FA, Perez LI, Malinowski DP, Molina-Montenegro MA, Gundel PE (2021) A systematic review on the effects of Epichloë fungal endophytes on drought tolerance in cool-season grasses. Front Plant Sci 12:644731. https://doi.org/10.3389/fpls.2021.644731
Dirihan S, Helander ML, Saloniemi I, Gundel PE, Saikkonen K (2015) Effects of systemic fungal endophytes on the performance of meadow fescue and tall fescue in mixtures with red clover. Grass Forage Sci 70:465–473. https://doi.org/10.1111/gfs.12145
Ebeling A, Strauss AT, Adler PB, Arnillas CA, Barrio IC, Biederman LA, Borer ET, Bugalho MN, Caldeira MC, Cadotte MW (2021) Nutrient enrichment increases invertebrate herbivory and pathogen damage in grasslands. J Ecol 110:327–339. https://doi.org/10.1111/1365-2745.13801
Farrer EC, Suding KN (2016) Teasing apart plant community responses to N enrichment: the roles of resource limitation, competition and soil microbes. Ecol Lett 19:1287–1296. https://doi.org/10.1111/ele.12665
Fu YW (2020) Responses and mechanisms of ecosystem carbon fluxes to multiple nutrient addition and drought in an alpine meadow. Agricultural and Animal Husbandry College of Tibet University (in Chinese). https://doi.org/10.27735/d.cnki.gxzdx.2020.000319
Galloway JN, Townsend AR, Erisman JW, Bekunda M, Cai Z, Freney JR, Martinelli LA, Seitzinger SP, Sutton MA (2008) Transformation of the nitrogen cycle: recent trends, questions, and potential solutions. Science 320:889–892. https://doi.org/10.1126/science.1136674
Graff P, Gundel PE, Salvat A, Cristos D, Chaneton EJ (2020) Protection offered by leaf fungal endophytes to an invasive species against native herbivores depends on soil nutrients. J Ecol 108:1592–1604. https://doi.org/10.1111/1365-2745.13371
Greaver TL, Clark CM, Compton JE, Vallano D, Talhelm AF, Weaver CP, Band LE, Baron JS, Davidson EA, Tague CL, Felker-Quinn E, Lynch JA, Herrick JD, Liu L, Goodale CL, Novak KJ, Haeuber RA (2016) Key ecological responses to nitrogen are altered by climate change. Nat Clim Change 6:836–843. https://doi.org/10.1038/nclimate3088
Guo JQ, McCulley RL, McNear DH Jr (2015) Tall fescue cultivar and fungal endophyte combinations influence plant growth and root exudate composition. Front Plant Sci 6:183. https://doi.org/10.3389/fpls.2015.00183
Guo ZP, Luo CS, Dong Y, Dong K, Zhu JH, Ma LK (2021) Effect of nitrogen regulation on the epidemic characteristics of intercropping faba bean rust disease primarily depends on the canopy microclimate and nitrogen nutrition. Field Crops Res 274:108339. https://doi.org/10.1016/j.fcr.2021.108339
Hahn PG, Keefover-Ring K, Nguyen LMN, Maron JL (2021) Intraspecific correlations between growth and defence vary with resource availability and differ within and among populations. Funct Ecol 35:2387–2396. https://doi.org/10.1111/1365-2435.13878
Hill NS, Belesky DP, Stringer WC (1991) Competitiveness of tall fescue as influenced by Acremonium coenophialum. Crop Sci 31:185–190. https://doi.org/10.2135/cropsci1991.0011183X003100010042x
Hill NS, Belesky DP, Stringer WC (1998) Encroachment of endophyte-infected on endophyte-free tall fescue. Ann Bot: 483–488. https://doi.org/10.1006/anbo.1998.0583
Hou WP, Wang JF, Christensen MJ, Liu J, Zhang YQ, Liu YL, Cheng C (2021) Metabolomics insights into the mechanism by which Epichloë gansuensis endophyte increased Achnatherum inebrians tolerance to low nitrogen stress. Plant Soil 463:487–508. https://doi.org/10.1007/s11104-021-04930-z
Iannone LJ, Vignale MV, Pinget AD, Re A, Mc Cargo PD, Novas MV (2017) Seed-transmitted Epichloë sp. endophyte alleviates the negative effects of head smut of grasses (Ustilago bullata) on Bromus auleticus. Fungal Ecol 29:45–51. https://doi.org/10.1016/j.funeco.2017.06.001
IRRI (1980) Standard evaluation system for rice. International Rice Research Institute, Philippines
Kneitel JM, Chase JM (2004) Trade-offs in community ecology: linking spatial scales and species coexistence. Ecol Lett 7:69–80. https://doi.org/10.1046/j.1461-0248.2003.00551.x
Latch G, Christensen M, Samuels G (1984) Five endophytes of Lolium and Festuca in New Zealand [Fungi, description, new taxa]. Mycotaxon 20:535–550
Lecompte F, Abro MA, Nicot PC (2013) Can plant sugars mediate the effect of nitrogen fertilization on lettuce susceptibility to two necrotrophic pathogens: Botrytis cinerea and Sclerotinia sclerotiorum? Pant Soil 369:387–401. https://doi.org/10.1007/s11104-012-1577-9
Leuchtmann A, Bacon CW, Schardl CL, White JF, Tadych M (2014) Nomenclatural realignment of Neotyphodium species with genus Epichloë. Mycologia 106:202–215. https://doi.org/10.3852/106.2.202
Lewis GC (2004) Effects of biotic and abiotic stress on the growth of three genotypes of Lolium perenne with and without infection by the fungal endophyte Neotyphodium lolii. Ann Appl Biol 144:53–63. https://doi.org/10.1111/j.1744-7348.2004.tb00316.x
Li X, Ren AZ, Han R, Yin LJ, Wei MY, Gao YB (2012) Endophyte-mediated effects on the growth and physiology of Achnatherum sibiricum are conditional on both N and P availability. PLoS One 7:e48010. https://doi.org/10.1371/journal.pone.0048010
Li X, Zhou Y, Mace W, Qin JH, Liu H, Chen W, Ren AZ, Gao YB (2016) Endophyte species influence the biomass production of the native grass Achnatherum sibiricum (L.) Keng under high nitrogen availability. Ecol Evol 6:8595–8606. https://doi.org/10.1002/ece3.2566
Lind EM, Borer E, Seabloom E et al (2013) Life-history constraints in grassland plant species: a growth-defence trade-off is the norm. Ecol Lett 16:513–521. https://doi.org/10.1111/ele.12078
Liu H, Chen J, Qin TZ, Shi XJ, Gao YB, Ren AZ (2020a) Removal of soil microbes alters interspecific competitiveness of Epichloë endophyte-infected over endophyte-free Leymus chinensis. Microorganisms 8:219. https://doi.org/10.3390/microorganisms8020219
Liu X, Lu Y, Zhang Z, Zhou S (2020b) Foliar fungal diseases respond differently to nitrogen and phosphorus additions in tibetan alpine meadows. Ecol Res 35:162–169. https://doi.org/10.1111/1440-1703.12064
Ma Y, Fu X, Chen S (1985) Flora Inner Mongolia. Inner Mongolia People’s Press, China
Malinowski D, Leuchtmann A, Schmidt D, Nösberger J (1997) Growth and water status in meadow fescue is affected by Neotyphodium and Phialophora species endophytes. Agron J 89:673–678. https://doi.org/10.2134/agronj1997.00021962008900040021x
Malinowski DP, Alloush GA, Belesky DP (1998a) Evidence for chemical changes on the root surface of tall fescue in response to infection with the fungal endophyte neotyphodium coenophialum. Plant Soil 205:1–12. https://doi.org/10.1023/A:1004331932018
Malinowski DP, Belesky DP, Hill NS, Baligar VC, Fedders JM (1998b) Influence of phosphorus on the growth and ergot alkaloid content of Neotyphodium coenophialum-infected tall fescue (Festuca arundinacea Schreb.). Plant Soil 198:53–61. https://doi.org/10.1023/A:1004279401196
McGilchrist CA, Trenbath BR (1971) A revised analysis of plant competition experiments. Biometrics 27:659–677. https://doi.org/10.2307/2528603
Mitchell CE, Reich PB, Tilman D, Groth JV (2003) Effects of elevated CO2, nitrogen deposition, and decreased species diversity on foliar fungal plant disease. Glob Change Biol 9:438–451. https://doi.org/10.1046/j.1365-2486.2003.00602.x
Pánka D, Piesik D, Jeske M, Baturo-Ciésniewska A (2013) Production of phenolics and the emission of volatile organic compounds by perennial ryegrass (Lolium perenne L.)/Neotyphodium lolii association as a response to infection by Fusarium poae. J Plant Physiol 170:1010–1019. https://doi.org/10.1016/j.jplph.2013.02.009
Pérez LI, Gundel PE, Zabalgogeazcoa I, Omacini M (2020) An ecological framework for understanding the roles of Epichloë endophytes on plant defenses against fungal diseases. Fungal Biol Rev 34:115–125. https://doi.org/10.1016/j.fbr.2020.06.001
R Core Team (2021) R: a language and environment for statistical computing. R Foundation for Statistical Computing
Ravel C, Courty C, Coudret A, Charmet G (1997) Beneficial effects of Neotyphodium lolii on the growth and the water status in perennial ryegrass cultivated under nitrogen deficiency or drought stress. Agronomie 17:173–181. https://doi.org/10.1051/agro:19970304
Ren AZ, Li X, Han R, Yin LJ, Wei MY, Gao YB (2011) Benefits of a symbiotic association with endophytic fungi are subject to water and nutrient availability in Achnatherum sibiricum. Plant Soil 346:363–373. https://doi.org/10.1007/s11104-011-0824-9
Rho H, Hsieh M, Kandel SL, Cantillo J, Doty SL, Kim SH (2018) Do endophytes promote growth of host plants under stress? A meta-analysis on plant stress mitigation by endophytes. Microb Ecol 75:407–418. https://doi.org/10.1007/s00248-017-1054-3
Rojas X, Guo J, Leff JW, McNear DH Jr, Fierer N, McCulley RL (2016) Infection with a shoot-specific fungal endophyte (Epichloë) alters tall fescue soil microbial communities. Microb Ecol 72:197–206. https://doi.org/10.1007/s00248-016-0750-8
Saari S, Faeth SH (2012) Hybridization of Neotyphodium endophytes enhances competitive ability of the host grass. New Phytol 195:231–236. https://doi.org/10.1111/j.1469-8137.2012.04140.x
Saikkonen K, Lehtonen P, Helander M, Koricheva J, Faeth SH (2006) Model systems in ecology: dissecting the endophyte-grass literature. Trends Plant Sci 11:428–433. https://doi.org/10.1016/j.tplants.2006.07.001
Sampedro L, Moreira X, Zas R (2011) Costs of constitutive and herbivore-induced chemical defences in pine trees emerge only under low nutrient availability. J Ecol 99:818–827. https://doi.org/10.1111/j.1365-2745.2011.01814.x
Shi XJ, Qin TZ, Liu H, Wu M, Li JJ, Shi YS, Gao YB, Ren AZ (2020) Corrigendum: endophytic fungi activated similar defense strategies of Achnatherum sibiricum host to different trophic types of pathogens. Front Microbiol 11:1607. https://doi.org/10.3389/fmicb.2020.611649
Shi XJ, Qin TZ, Qu YB, Zhang JZ, Hao G, Zhao NX, Gao YB, Ren AZ (2022) Infection by endophytic Epichloë sibirica was associated with activation of defense hormone signal transduction pathways and enhanced pathogen resistance in the grass Achnatherum sibiricum. Phytopathology 112:2310–2320. https://doi.org/10.1094/PHYTO-12-21-0521-R
Song H, Nan ZB, Song QY, Xia C, Li XZ, Yao X, Xu WB, Kuang Y, Tian P, Zhang QP (2016) Advances in research on Epichloë endophytes in chinese native grasses. Front Microbiol 7:1399. https://doi.org/10.3389/fmicb.2016.01399
Tian QY, Liu NN, Bai WM, Li LH, Chen JQ, Reich PB, Yu Q, Guo DL, Smith MD, Knapp AK, Cheng WX, Lu P, Gao Y, Yang A, Wang TZ, Li X, Wang ZW, Ma YB, Han XG, Zhang WH (2016) A novel soil manganese mechanism drives plant species loss with increased nitrogen deposition in a temperate steppe. Ecology 97:65–74. https://doi.org/10.1890/15-0917.1
van der Heijden MGA, Klironomos JN, Ursic M, Moutoglis P, Streitwolf-Engel R, Boller T, Wiemken A, Sanders IR (1998) Mycorrhizal fungal diversity determines plant biodiversity, ecosystem variability and productivity. Nature 396:69–72. https://doi.org/10.1038/23932
Vázquez-de-Aldana BR, Zabalgogeazcoa I, García-Ciudad A, García-Criado B (2013) An Epichloë endophyte affects the competitive ability of Festuca rubra against other grassland species. Plant Soil 362:201–213. https://doi.org/10.1007/s11104-012-1283-7
Wang XY, Qin JH, Chen W, Zhou Y, Ren AZ, Gao YB (2016) Pathogen resistant advantage of endophyte-infected over endophyte-free Leymus chinensis is strengthened by pre-drought treatment. Eur J Plant Pathol 144:477–486. https://doi.org/10.1007/s10658-015-0788-3
Wang JJ, Zhou YP, Lin WH, Li MM, Wang MN, Wang ZG, Kuang Y, Tian P (2017) Effect of an Epichloë endophyte on adaptability to water stress in Festuca sinensis. Fungal Ecol 30:39–47. https://doi.org/10.1016/j.funeco.2017.08.005
Wang JF, Nan ZB, Christensen MJ, Zhang XX, Tian P, Zhang ZX, Niu XL, Gao P, Chen T, Ma LX (2018) Effect of Epichloë gansuensis endophyte on the nitrogen metabolism, nitrogen use efficiency, and stoichiometry of Achnatherum inebrians under nitrogen limitation. J Agric Food Chem 66:4022–4031. https://doi.org/10.1021/acs.jafc.7b06158
Wei YK, Gao YB, Xu H, Su D, Zhang X, Wang YH, Lin F, Chen L, Nie LY, Ren AZ (2006) Occurrence of endophytes in grasses native to northern China. Grass Forage Sci 61:422–429. https://doi.org/10.1111/j.1365-2494.2006.00551.x
Zang XP, Xu YP, Cai XZ (2010) Establishment of an inoculation technique system for Sclerotinia sclerotiorum based on mycelial suspensions. J Zhejiang Univ (Agric. Life Sci) 36:381–386
Zhang X, Ren AZ, Wei YK, Lin F, Li C, Liu ZJ, Gao YB (2009) Taxonomy, diversity and origins of symbiotic endophytes of Achnatherum sibiricum in the Inner Mongolia Steppe of China. FEMS Microbiol Lett 301:12–20. https://doi.org/10.1111/j.1574-6968.2009.01789.x
Zhang JZ, Deng YK, Ge XY, Shi XJ, Fan XW, Dong K, Chen L, Zhao NX, Gao YB, Ren AZ (2022) The beneficial effect of Epichloë endophytes on the growth of host grasses was affected by arbuscular mycorrhizal fungi, pathogenic fungi and nitrogen addition. Environ Exp Bot 201:104979. https://doi.org/10.1016/j.envexpbot.2022.104979
Zhou Y, Li X, Gao Y, Liu H, Gao YB, van der Heijden MG, Ren AZ (2018) Plant endophytes and arbuscular mycorrhizal fungi alter plant competition. Funct Ecol 32:1168–1179. https://doi.org/10.1111/1365-2435.13084
Zhou Y, Li X, Liu H, Gao YB, Mace WJ, Card SD, Ren AZ (2019) Effects of endophyte infection on the competitive ability of Achnatherum sibiricum depend on endophyte species and nitrogen availability. J Plant Ecol 12:815–824. https://doi.org/10.1093/jpe/rtz017
Acknowledgements
This work was supported by International Cooperation and Exchange of the National Science Foundation (32061123004) and the National Natural Science Foundation of China (32271586 and 31971425).
Author information
Authors and Affiliations
Contributions
Junzhen Zhang and Anzhi Ren conceived the ideas and designed methodology; Junzhen Zhang, Xinhe Yu, Yaobing Qu, Xinjian Shi and Luoyang He collected the data; Junzhen Zhang, Xianqin Wei and Nianxi Zhao analyzed the data; Junzhen Zhang, Lei Chen and Anzhi Ren led the writing of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that we have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Responsible Editor: Hans Lambers.
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, J., Yu, X., Qu, Y. et al. The decreasing trend of the competitive advantage of endophyte-infected Achnatherum sibiricum over endophyte-free plants under high nitrogen conditions was reversed by pathogenic fungi inoculation. Plant Soil 493, 427–439 (2023). https://doi.org/10.1007/s11104-023-06239-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-023-06239-5