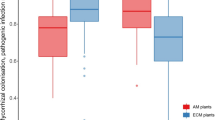
Abstract
Aims
The relationship between functional traits and growth conditions across tree species provides critical information for understanding resource acquisition strategy of each species. This study aims to elucidate the relationship between root tip morphology and growth conditions with considering the phylogenetic difference in a primary lowland dipterocarp forest.
Methods
We surveyed growth conditions for 13 target species of Macaranga and Shorea, i.e., light condition, soil physical properties, and soil nitrogen (N) dynamics, and root tip morphology, including specific root tip length (SRLt), root tip diameter (RDt), and root tip tissue density (RTDt) and analyzed their relationships considering the difference between genera.
Results
The differences in SRLt and RDt between two genera, Macaranga and Shorea, were validated by the difference in environmental factors, i.e., light intensity and soil ammonification rate. With considering the difference between genera, we detected an interspecific variation in SRLt, RDt, and RTDt across all 13 species in response to differences in environmental factors such as light intensity, soil physical properties, soil nitrification, and N mineralization rates. Among the same species, the SRLt decreased with increasing light intensity and increased with increasing nitrification rates, while the RDt exhibited an opposite trend.
Conclusion
We confirmed that the root tip morphology varies according to growth conditions in tropical tree species: shade-tolerant tree species and/or tree species grown in fertile condition had root tips with high efficiency of soil exploration or exploitation per a unit of root biomass. This variation pattern was different than that of temperate tree species.




Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available on TRY plant trait database (https://doi.org/10.17871/TRY.87).
References
Ahmad N (1996) The role of mycorrhizas in the regeneration of some Malaysian lowland rain forest trees of Jengka. J Trop for Sci 8:441–448
Aiba M, Nakashizuka T (2007) Variation in juvenile survival and related physiological traits among dipterocarp species co-existing in a Bornean forest. J Veg Sci 18:379–388. https://doi.org/10.1111/j.1654-1103.2007.tb02550.x
Allbrook RF (1973) The soils of Pasoh forest reserve, Negeri Sembilan. Malay Forester 36:22–33
Amissah L, Mohren GMJ, Kyereh B, Poorter L (2015) The effects of drought and shade on the performance, morphology and physiology of Ghanaian tree species. PLoS ONE 10:e0121004. https://doi.org/10.1371/journal.pone.0121004
Ashton MS (1995) Seedling growth of co-occurring Shorea species in the simulated light environments of a rain forest. For Ecol Manag 72:1–12. https://doi.org/10.1016/0378-1127(94)03452-3
Ashton PS, Okuda T, Manokaran N (2003) Pasoh research, past and present. In: Okuda T, Manokaran N, Matsumoto Y, Niiyama K, Thomas SC, Ashton PS (eds) Pasoh, ecology of a lowland rain forest in southeast Asia. Springer, Tokyo, pp 1–14
Bates D, Maechler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48
Bengough AG, Bransby MF, Hans J, McKenna SJ, Roberts TJ, Valentine TA (2006) Root responses to soil physical conditions; growth dynamics from field to cell. J Exp Bot 57:437–447. https://doi.org/10.1093/jxb/erj003
Bergmann J, Weigelt A, van der Plas F, Laughlin DC, Kuyper TW, Guerrero-Ramirez N, Valverde-Barrantes OJ, Bruelheide H, Freschet GT, Iversen CM, Kattge J, McCormack MC, Meier IC, Rillig MC, Roumet C, Semchenko M, Sweeney CJ, van Ruijven J, York LM, Mommer L (2020) The fungal collaboration gradient dominates the root economics space in plants. Sci Adv 6: eaba3756. https://doi.org/10.1126/sciadv.aba3756
Beudert B, Bernsteinová J Premier J, Bässler C (2018) Natural disturbance by bark beetle offsets climate change effects on streamflow in headwater catchments of the Bohemian Forest. Silva Gabreta 24:21–45
Brundrett MC (2009) Mycorrhizal associations and other means of nutrition of vascular plants: understanding the global diversity of host plants by resolving conflicting information and developing reliable means of diagnosis. Plant Soil 320:37–77. https://doi.org/10.1007/s11104-008-9877-9
Cantarel AAM, Pommier T, Desclos-Theveniau M, Diquélou S, Dumont M, Grassein F, Kastl EM, Grigulis K, Laîné P, Lavorel S, Lemauviel-Lavenant S, Personeni E, Schloter M, Poly F (2015) Using plant traits to explain plant-microbe relationships involved in nitrogen acquisition. Ecology 96:788–799. https://doi.org/10.1890/13-2107.1
Caplan JS, Meiners SJ, Flores-Moreno H, McCormack ML (2019) Fine-root traits are linked to species dynamics in a successional plant community. Ecology 100:e02588. https://doi.org/10.1002/ecy.2588
Chen W, Eissenstat DM, Koide RT (2018) Root diameter predicts the extramatrical hyphal exploration distance of the ectomycorrhizal fungal community. Ecosphere 9:e02202. https://doi.org/10.1002/ecs2.2202
Chen W, Koide RT, Adams TS, DeForest JL, Cheng L, Eissenstat DM (2016) Root morphology and mycorrhizal symbioses together shape nutrient foraging strategies of temperate trees. PNAS 113:8741–8746. https://doi.org/10.1073/pnas.1601006113
Chen W, Zeng H, Eissenstat DM, Guo D (2013) Variation of first-order root traits across climatic gradients and evolutionary trends in geological time. Global Ecol Biogeogr 22:846–856. https://doi.org/10.1111/geb.12048
Comas LH, Bouma TJ, Eissenstat DM (2002) Linking root traits to potential growth rate in six temperate tree species. Oecologia 132:34–43. https://doi.org/10.1007/s00442-002-0922-8
Comas LH, Callahan HS, Midford PE (2014) Patterns in root traits of woody species hosting arbuscular and ectomycorrhizas: implications for the evolution of belowground strategies. Ecol Evol 4:2979–2990. https://doi.org/10.1002/ece3.1147
Comas LH, Eissenstat DM (2004) Linking fine root traits to maximum potential growth rate among 11 mature temperate tree species. Funct Ecol 18:388–397. https://doi.org/10.1111/j.0269-8463.2004.00835.x
Comas LH, Eissenstat DM (2009) Patterns in root trait variation among 25 co-existing North American forest species. New Phytol 182:919–928. https://doi.org/10.1111/j.1469-8137.2009.02799.x
Comas LH, Mueller KE, Taylor LL, Midford PE, Callahan HS, Beerling DJ (2012) Evolutionary patterns and biogeochemical significance of angiosperm root traits. Int J Plant Sci 173:584–595. https://doi.org/10.1086/665823
Davies SJ (2001) Tree mortality and growth in 11 sympatric Macaranga species in Borneo. Ecology 82:920–932. https://doi.org/10.1890/0012-9658(2001)082[0920:TMAGIS]2.0.CO;2
Davies SJ, Palmiotto PA, Ashton PS, Lee HS, Lafrankie JV (1998) Comparative ecology of 11 sympatric species of Macaranga in Borneo: tree distribution in relation to horizontal and vertical resource heterogeneity. J Ecol 86:662–673. https://doi.org/10.1046/j.1365-2745.1998.00299.x
de la Riva EG, Querejeta JI, Villar R, Pérez-Ramos IM, Marañón T, Galán Díaz J, de Tomás Marín S and Prieto I (2021) The economics spectrum drives root trait strategies in Mediterranean vegetation. Front Plant Sci 12: 773118. https://doi.org/10.3389/fpls.2021.773118
Eissenstat DM, Kucharski JM, Zadworny M, Adams TS, Koide RT (2015) Linking root traits to nutrient foraging in arbuscular mycorrhizal trees in a temperate forest. New Phytol 208:114–124. https://doi.org/10.1111/nph.13451
Eissenstat DM, Wells CE, Yanai RD, Whitbeck JL (2000) Building roots in a changing environment: implications for root longevity. New Phytol 147:33–42. https://doi.org/10.1046/j.1469-8137.2000.00686.x
Fox J, Weisberg S (2019) An R Companion to Applied Regression, 3rd edn. SAGE Publications, Los Angeles
Frazer GW, Canham CD, Lertzman KP (1999) Gap Light Analyzer (GLA), Version 2.0: Imaging software to extract canopy structure and gap light transmission indices from true-colour fisheye photographs, users manual and program documentation. Simon Fraser University, Burnaby, British Columbia, and the Institute of Ecosystem Studies, Millbrook, New York
Freschet GT, Valverde-Barrantes OJ, Tucker CM, Craine JM, McCormack ML, Violle C, Fort F, Blackwood CB, Urban-Mead KR, Iversen CM, Bonis A, Comas LH, Cornelissen JHC, Dong M, Guo D, Hobbie SE, Holdaway RJ, Kembel SW, Makita N, Onipchenko VG, Picon-Cochard C, Reich PB, de la Riva EG, Smith SW, Soudzilovskaia NA, Tjoelker MG, Wardle DA, Roumet C (2017) Climate, soil and plant functional types as drivers of global fine-root trait variation. J Ecol 105:1182–1196. https://doi.org/10.1111/1365-2745.12769
Gee GW, Or D (2002) Particle-size analysis. In: Dane JH, Topp GC (eds) Method of soil analysis, part 4, physical methods. Soil Science Society of America, Madison, pp 255–293
Glatthorn J, Beckschäfer P (2014) Standardizing the protocol for hemispherical photographs: accuracy assessment of binarization algorithms. PLoS One 9:e111924. https://doi.org/10.1371/journal.pone.0111924
Guo D, Mitchell RJ, Withington JM, Fan PP, Hendricks JJ (2008) Endogenous and exogenous controls of root life span, mortality and nitrogen flux in a longleaf pine forest: root branch order predominates. J Ecol 96:737–745. https://doi.org/10.1111/j.1365-2745.2008.01385.x
Han M, Chen Y, Li R, Yu M, Fu L, Li S, Su J, Zhu B (2022) Root phosphatase activity aligns with the collaboration gradient of the root economics space. New Phytol (2022) 234: 837–849. https://doi.org/10.1111/nph.17906
Holdaway RJ, Richardson SJ, Dickie IA, Peltzer DA, Coomes DA (2011) Species- and community-level patterns in fine root traits along a 120000-year soil chronosequence in temperate rain forest. J Ecol 99:954–963. https://doi.org/10.1111/j.1365-2745.2011.01821.x
Hothorn T, Bretz F, Westfall P (2008) Simultaneous inference in general parametric models. Biom J 50:346–363
Iversen CM, McCormack ML, Powell AS, Blackwood CB, Freschet GT, Kattge J, Roumet C, Stover DB, Soudzilovskaia NA, Valverde-Barrantes OJ, van Bodegom PM, Violle C (2017) A global fine-root ecology database to address below-ground challenges in plant ecology. New Phytol 215:15–26. https://doi.org/10.1111/nph.14486
Jia S, McLaughlin NB, Gu J, Li X, Wang Z (2013) Relationships between root respiration rate and root morphology, chemistry and anatomy in Larix gmelinii and Fraxinus mandshurica. Tree Physiol 33:579–589. https://doi.org/10.1093/treephys/tpt040
Katabuchi M, Kurokawa H, Davies SJ, Tan S, Nakashizuka T (2012) Soil resource availability shapes community trait structure in a species-rich dipterocarp forest. J Ecol 100:643–651. https://doi.org/10.1111/j.1365-2745.2011.01937.x
Kembel SW, Cahill JF Jr (2011) Independent evolution of leaf and root traits within and among temperate grassland plant communities. PLoS One 6(6):e19992. https://doi.org/10.1371/journal.pone.0019992
Kopáček J, Bače R, Hejzlar J, Kaňa J, Kučera T, Matějka K, Porcal P, Turek J (2020) Changes in microclimate and hydrology in an unmanaged mountain forest catchment after insect-induced tree dieback. Sci Total Environ 720:137518. https://doi.org/10.1016/j.scitotenv.2020.137518
Kosugi Y, Takanashi S, Ohkubo S, Matsuo N, Tani M, Mitani T, Tsutsumi D, Nik AR (2008) CO2 exchange of a tropical rainforest at Pasoh in Peninsular Malaysia. Agric for Meteorol 148:439–452
Kumagai TK, Yoshifuji N, Tanaka N, Suzuki M, Kume T (2009) Comparison of soil moisture dynamics between a tropical rain forest and a tropical seasonal forest in Southeast Asia: Impact of seasonal and year-to-year variations in rainfall. Water Resour Res 45:W04413. https://doi.org/10.1029/2008WR007307
Lion M, Kosugi Y, Takanashi S, Noguchi S, Itoh M, Katsuyama M, Matsuo N, Shamsuddin SA (2017) Evapotranspiration and water source of a tropical rainforest in peninsular Malaysia. Hydrol Process 31:4338–4353. https://doi.org/10.1002/hyp.11360
Liu B, Li H, Zhu B, Koide RT, Eissenstat DM, Guo D (2015) Complementarity in nutrient foraging strategies of absorptive fine roots and arbuscular mycorrhizal fungi across 14 coexisting subtropical tree species. New Phytol 208:125–136. https://doi.org/10.1111/nph.13434
Liu C, Xiang W, Zou L, Lei P, Zeng Y, Ouyang S, Deng X, Fang X, Liu Z, Peng C (2019) Variation in the functional traits of fine roots is linked to phylogenetics in the common tree species of Chinese subtropical forests. Plant Soil 436:347–364. https://doi.org/10.1007/s11104-019-03934-0
Lu B, Qian J, Hu J, Wang P, Jin W, Tang S, He Y, Zhang C (2022) The role of fine root morphology in nitrogen uptake by riparian plants. Plant Soil 472:527–542. https://doi.org/10.1007/s11104-021-05270-8
Ma A, Guo D, Xu X, Lu M, Bardgett RD, Eissenstat DM, McCormack ML, Hedin LO (2018) Evolutionary history resolves global organization of root functional traits. Nature 555:94–97. https://doi.org/10.1038/nature25783
Makita N, Kosugi Y, Dannoura M, Takanashi S, Niiyama K, Kassim AR, Nik AR (2012) Patterns of root respiration rates and morphological traits in 13 tree species in a tropical forest. Tree Physiol 32:303–312. https://doi.org/10.1093/treephys/tps008
Manokaran N, Quah ES, Ashton PS, LaFrankie JV, Nur Supardi MNW, Ahmad WMS, Okuda T (2004) Pasoh forest dynamics plot, Malaysia. In: Losos EC, Leigh EG (eds) Forest diversity and dynamism: findings from a network of largescale tropical forest plots. University of Chicago Press, Chicago, pp 585–598
Markesteijn L, Poorter L (2009) Seedling root morphology and biomass allocation of 62 tropical tree species in relation to drought- and shade-tolerance. J Ecol 97:311–325. https://doi.org/10.1111/j.1365-2745.2008.01466.x
McCormack ML, Adams TS, Smithwick EA, Eissenstat DM (2012) Predicting fine root lifespan from plant functional traits in temperate trees. New Phytol 195:823–831. https://doi.org/10.1111/j.1469-8137.2012.04198.x
McCormack ML, Dickie IA, Eissenstat DM, Fahey TJ, Fernandez CW, Guo D, Helmisaari HS, Hobbie EA, Iversen CM, Jackson RB, Leppäalammi-Kujansuu J, Norby RJ, Phillips RP, Pregitzer KS, Pritchard SG, Rewald B, Zadworny M (2015) Redefining fine roots improves understanding of below-ground contributions to terrestrial biosphere processes. New Phytol 207:505–518. https://doi.org/10.1111/nph.13363
Modrzyński J, Chmura DJ, Tjoelker MG (2015) Seedling growth and biomass allocation in relation to leaf habit and shade tolerance among 10 temperate tree species. Tree Physiol 35:879–893. https://doi.org/10.1093/treephys/tpv053
Niiyama K, Kajimoto T, Matsuura Y, Yamashita T, Matsuo M, Yashiro Y, Ripin A, Kassim AR, Noor NS (2010) Estimation of root biomass based on excavation of individual root systems in a primary dipterocarp forest in Pasoh Forest Reserve, Peninsular Malaysia. J Trop Ecol 26:1–14. https://doi.org/10.1017/S0266467410000040
Nobis M (2005) SideLook 1.1 -Imaging software for the analysis of vegetation structure with true-colour photographs. Available via web. http://www.appleco.ch. Accessed 28 Jan 2016.
Nobis M, Hunziker U (2005) Automatic thresholding for hemispherical canopy-photographs based on edge detection. Agric For Meteorol 128:243–250. https://doi.org/10.1016/j.agrformet.2004.10.002
Noguchi S, Kosugi Y, Takanashi S, Tani M, Niiyama K, Siti Aisah S, Marryanna L (2016) Long term variation in soil moisture in the Pasoh Forest reserve, a lowland tropical rain forest in Malaysia. J Trop for Sci 28:324–333
Oksanen J, Simpson GL, Blanchet GF, Kindt R, Legendre P, Minchin PR, O’Hara RB, Solymos P, Stevens MHH, Szoecs E, Wagner H, Barbour M, Bedward M, Bolker B, Borcard D, Carvalho G, Chirico M, De Caceres M, Durand S, Antoniazi Evangelista HBA, FitzJohn R, Friendly M, Furneaux B, Hannigan G, Hill MO, Lahti L, McGlinn D, Ouellette MH, Cunha ER, Smith T, Stier A, Braak CJFT, Weedon J (2022) vegan: Community Ecology Package. R package version 2.6–2. https://CRAN.R-project.org/package=vegan
Olsson PA, Rahm J, Aliasgharzad N (2010) Carbon dynamics in mycorrhizal symbioses is linked to carbon costs and phosphorus benefits. FEMS Microbiol Ecol 72:123–131. https://doi.org/10.1111/j.1574-6941.2009.00833.x
Palmiotto PA, Davies SJ, Vogt KA, Ashton MS, Vogt DJ, Ashton PS (2004) Soil-related habitat specialization in dipterocarp rain forest tree species in Borneo. J Ecol 92:609–623. https://doi.org/10.1111/j.0022-0477.2004.00894.x
Paoli GD, Curran LM, Zak DR (2006) Soil nutrients and beta diversity in the Bornean Dipterocarpaceae: evidence for niche partitioning by tropical rain forest trees. J Ecol 94:157–170. https://doi.org/10.1111/j.1365-2745.2005.01077.x
Poorter L, Arets EJMM (2003) Light environment and tree strategies in a Bolivian tropical moist forest: an evaluation of the light partitioning hypothesis. Plant Ecol 166:295–306. https://doi.org/10.1023/A:1023295806147
Pregitzer KS, DeForest JL, Burton AJ, Allen MF, Ruess RW, Hendrick RL (2002) Fine root architecture of nine North American trees. Ecol Monogr 72:293–309. https://doi.org/10.1890/0012-9615(2002)072[0293:FRAONN]2.0.CO;2
R Core Team (2021) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria.
Reich PB (2014) The world-wide “fast-slow” plant economics spectrum: a traits manifesto. J Ecol 102:275–301. https://doi.org/10.1111/1365-2745.12211
Rewald B, Ephrath JE, Rachmilevitch S (2011) A root is a root is a root? Water uptake rates of Citrus root orders. Plant Cell Environ 34:33–42. https://doi.org/10.1111/j.1365-3040.2010.02223.x
See Chung C (1995) Rain forest in the city: Bukit Timah Nature Reserve, Singapore. Singapore Botanic Gardens, Singapore.
Shipley B, Bello FD, Cornelissen JHC, Laliberté E, Laughlin DC, Reich PB (2016) Reinforcing loose foundation stones in trait-based plant ecology. Oecologia 180:923–931. https://doi.org/10.1007/s00442-016-3549-x
Soil Survey Staff (2006) Keys to soil taxonomy, 10th edn. USDA Natural Resources Conservation Service, Washington, DC
Tianxiang L, Ronald NP, Hanqin T, Charles VJ, Huazhong Z, Shirong L (2002) A model for seasonality and distribution of leaf area index of forests and its application to China. J Veg Sci 13:817–830. https://doi.org/10.1111/j.1654-1103.2002.tb02111.x
The Angiosperm Phylogeny Group (2016) An update of the Angiosperm Phylogeny Group classification for the orders and families of flowering plants: APG IV. Bot J Linn Soc 181:1–20. https://doi.org/10.1111/boj.12385
Ugawa S, Inagaki Y, Karibu F, Tateno R (2020) Effects of soil compaction by a forestry machine and slash dispersal on soil N mineralization in Cryptomeria japonica plantations under high precipitation. New for 51:887–907. https://doi.org/10.1007/s11056-019-09768-z
Valverde-Barrantes OJ, Blackwood CB (2016) Root traits are multidimensional: specific root length is independent from root tissue density and the plant economic spectrum: Commentary on Kramer-Walter et al. (2016). J Ecol 104:1311–1313. https://doi.org/10.1111/1365-2745.12605
Valverde-Barrantes OJ, Freschet GT, Roumet C, Blackwood CB (2017) A worldview of root traits: the influence of ancestry, growth form, climate and mycorrhizal association on the functional trait variation of fine-root tissues in seed plants. New Phytol 215:1562–1573. https://doi.org/10.1111/nph.14571
van der Sande MT, Peña-Claros M, Ascarrunz N, Arets EJMM, Licona JC, Toledo M, Poorter L (2017) Abiotic and biotic drivers of biomass change in a Neotropical forest. J Ecol 105:1223–1234. https://doi.org/10.1111/1365-2745.12756
Vleminckx J, Fortunel C, Valverde-Barrantes O, Paine CET, Engel J, Petronelli P, Dourdain AK, Guevara J, Béroujon S, Baraloto C (2021) Resolving whole-plant economics from leaf, stem and root traits of 1467 Amazonian tree species. Oikos 130:1193–1208. https://doi.org/10.1111/oik.08284
Wang Y, Gao G, Wang N, Wang Z, Gu J (2019) Effects of morphology and stand structure on root biomass and length differed between absorptive and transport roots in temperate trees. Plant Soil 442:355–367. https://doi.org/10.1007/s11104-019-04206-7
Weemstra M, Kiorapostolou N, van Ruijven J, Mommer L, de Vries J, Sterck F (2020) The role of fine-root mass, specific root length and life span in tree performance: A whole-tree exploration. Funct Ecol 34:575–585. https://doi.org/10.1111/1365-2435.13520
Whitmore TC (1984) Tropical Rain Forests of the Far East. Clarendon Press, Oxford, UK
Yamada T, Tomita A, Itoh A, Yamakura T, Ohkubo T, Kanzaki M, Tan S, Ashton PS (2006) Habitat associations of Sterculiaceae trees in a Bornean rain forest plot. J Veg Sci 17:559–566. https://doi.org/10.1111/j.1654-1103.2006.tb02479.x
Yamada T, Yamada Y, Okuda T, Fletcher C (2013) Soil-related variations in the population dynamics of six dipterocarp tree species with strong habitat preferences. Oecologia 172:713–724. https://doi.org/10.1007/s00442-012-2529-z
Yamashita T, Kasuya N, Wan-Rasidah K, Suhaimi-Wan C, Quah-Eng S, Okuda T (2003) Soil and belowground characteristics of Pasoh Forest Reserve. In: Okuda T, Manokaran Y, Matsumoto Y, Niiyama K, Thomas SC, Ashton PS (eds) Pasoh: Ecology of a lowland rain forest in Southeast Asia. Springer, Tokyo, pp 89–109
Acknowledgements
We thank the members of the Soil Management Branch in Forest Research Institute of Malaysia and the Silviculture Laboratory in Faculty of Agriculture, Kagoshima University for helping with this research. We wish to thank the editor and the reviewers for their constructive comments on our manuscript.
Funding
This work was supported by JSPS KAKENHI Grant Number 25870569.
Author information
Authors and Affiliations
Contributions
Shin Ugawa designed this experiment, analyzed the collected data, and prepared this manuscript; Wataru Kuninaka, Keisuke Hayata, Naoko Maruta, and Syota Ohashi collected the field data and soil samples, and measured light and edaphic environmental factors; Victoria Rika Kubota collected the field data and identified tree species; Ahmad Rozita and Kadir Wan Rashidah measured soil N dynamics and supported to get a permission for this survey in the Pasoh Forest Reserve. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Responsible Editor: Amandine Erktan.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ugawa, S., Kuninaka, W., Hayata, K. et al. Relationship between root tip morphology and growth conditions across Macaranga and Shorea species in a tropical lowland forest of Peninsula Malaysia. Plant Soil 481, 621–639 (2022). https://doi.org/10.1007/s11104-022-05665-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-022-05665-1