Abstract
Purpose
Atractylodes lancea is a medicinal plant widely used in treating rheumatic diseases, digestive disorders, night blindness, and influenza. Microbes greatly impact plant growth and metabolism. However, the microbiome associated with A. lancea remains unclear. Hence, we aimed at assessing the effect of soil microbe inoculation on A. lancea under heat stress from multiple perspectives, including regulation of growth, valuable secondary metabolites, root endophytic and rhizosphere bacterial communities.
Methods
A. lancea was inoculated with soil microbes, then grown under normal/high temperature. Biomass, chlorophyll contents, production of major medicinal compounds, physiochemical properties of the soil, and in the composition of root bacterial communities of A. lancea were investigated.
Results
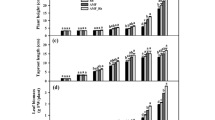
Soil microbe inoculation promoted root sink strength, accumulation of medicinal compounds, and attenuated damage caused by heat stress. A. lancea showed preference for the endophytic bacterial genera Rhodococcus, Ralstonia, Dongia Paenibacillus and Burkholderia-Caballeronia-Paraburkholderia post-inoculation, the latter four genera playing important roles in protection from heat stress, with abundance of the latter two specifically positively correlated to medicinal compound production. A. lancea enriched the bacterial genera Saccharimonadales, Novosphingobium and excluded Chitinophaga in its rhizosphere post-inoculation.
Conclusions
Soil microbes characteristically promoted A. lancea growth, improved heat stress tolerance, and promoted root medicinal compound accumulation. A. lancea selectively enriched particular endophytic and rhizospheric bacterial communities post-inoculation, possibly due to unique aromatic root exudates. The selected bacteria potentially synergistically improved soil available nutrients and uptake by root. Bacterial species selected by A. lancea root have the potential to serve as biological fertilizers for A. lancea farming.






Similar content being viewed by others
Data availability
Publicly available datasets were analyzed in this study. These data can be found in the NCBI database under accession numbers SRR13132034–SRR13132056.
Code availability
The codes in this article are available on the free online platform of Majorbio Cloud Platform (www.majorbio.com).
References
Abedinzadeh M, Etesami H, Alikhani HA (2019) Characterization of rhizosphere and endophytic bacteria from roots of maize (Zea mays L.) plant irrigated with wastewater with biotechnological potential in agriculture. Biotechnol Rep 21:e00305. https://doi.org/10.1016/j.btre.2019.e00305
Afzal I, Shinwari ZK, Sikandar S, Shahzad S (2019) Plant beneficial endophytic bacteria: mechanisms, diversity, host range and genetic determinants. Microbiol Res 221:36–49. https://doi.org/10.1016/j.micres.2019.02.001
Ahemad M (2019) Remediation of metalliferous soils through the heavy metal resistant plant growth promoting bacteria: paradigms and prospects. Arab J Chem 12:1365–1377. https://doi.org/10.1016/j.arabjc.2014.11.020
Bais HP, Weir TL, Perry LG, Gilroy S, Vivanco JM (2006) The role of root exudates in rhizosphere interactions with plants and other organisms. Annu Rev Plant Biol 57:233–266. https://doi.org/10.1146/annurev.arplant.57.032905.105159
Bartikova H, Hanusova V, Skalova L, Ambroz M, Bousova I (2014) Antioxidant, pro-oxidant and other biological activities of sesquiterpenes. Curr Top Med Chem 14:2478–2494. https://doi.org/10.2174/1568026614666141203120833
Berg G, Rybakova D, Fischer D, Cernava T, Vergès M-CC, Charles T, Chen X, Cocolin L, Eversole K, Corral GH (2020) Microbiome definition re-visited: old concepts and new challenges. Microbiome 8:1–22. https://doi.org/10.1186/s40168-020-00875-0
Bolger AM, Lohse M, Usadel B (2014) Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30:2114–2120. https://doi.org/10.1093/bioinformatics/btu170
Bulgarelli D, Schlaeppi K, Spaepen S, Loren V, van Themaat E, Schulze-Lefert P (2013) Structure and functions of the bacterial microbiota of plants. Annu Rev Plant Biol 64:807–838. https://doi.org/10.1146/annurev-arplant-050312-120106
Cane DE (1999) Sesquiterpene biosynthesis: cyclization mechanisms. Comprehen Natural Prod Chem 2:155–200. https://doi.org/10.1016/B978-0-08-091283-7.00039-4
Caverzan A, Casassola A, Brammer SP (2016) Reactive oxygen species and antioxidant enzymes involved in plant tolerance to stress. Abiotic and biotic stress in plants-recent advances and future perspectives 17:463–480
Chen WM, James EK, Prescott AR, Kierans M, Sprent JI (2003) Nodulation of Mimosa spp. by the beta-proteobacterium Ralstonia taiwanensis. Mol Plant-Microbe Interact 16:1051–1061. https://doi.org/10.1094/MPMI.2003.16.12.1051
Deng J, Wan QY, Gong L, Liu HG, Kun YU (2017) Cloning and analysis of DXS gene from Atractylodes lancea. Chin J Exp Tradit Med Formulae 23:39–44. https://doi.org/10.13422/j.cnki.syfjx.2017160039
Devanathan J, Thiripurasundari T, Selvam K, Selvaraj S, Ramadass L (2021) Isolation and characterization of drought stress tolerant plant growth promoting rhizobacter from chilli crop. Bulletin of Scientific Research 1-12. https://doi.org/10.34256/bsr2111
Dickie IA (2010) Insidious effects of sequencing errors on perceived diversity in molecular surveys. New Phytol 188:916–918. https://doi.org/10.1111/j.1469-8137.2010.03473.x
Edgar RC (2013) UPARSE: highly accurate OTU sequences from microbial amplicon reads. Nat Methods 10:996–998. https://doi.org/10.1038/nmeth.2604
Edgar RC, Haas BJ, Clemente JC, Quince C, Knight R (2011) UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 27:2194–2200. https://doi.org/10.1093/bioinformatics/btr381
Elphinstone JG (2005) The current bacterial wilt situation: a global overview. Bacterial wilt disease and the Ralstonia solanacearum species complex, pp 9–28
Grady EN, MacDonald J, Liu L, Richman A, Yuan ZC (2016) Current knowledge and perspectives of Paenibacillus: a review. Microb Cell Factories 15:203. https://doi.org/10.1186/s12934-016-0603-7
Guo LP, Liu JY, Li JI, Huang LQ (2002) The naphtha composing characteristics of geoherbs of Atractylodes lancea. China J Chin Materia Medica 27:814–819.https://doi.org/10.3321/j.issn:1001-5302.2002.11.004
Guo LP, Huang LQ, Yan H, Lv DM, Jiang YX (2005) Habitat characteristics for the growth of Atractylodes lancea based on GIS. China J Chin Materia Medica 30:565–569. https://doi.org/10.1111/j.1744-7909.2005.00136.x
Hamady M, Walker JJ, Harris JK, Gold NJ, Knight R (2008) Error-correcting barcoded primers for pyrosequencing hundreds of samples in multiplex. Nat Methods 5:235–237. https://doi.org/10.1038/nmeth.1184
Huang J, Cardoza YJ, Schmelz EA, Raina R, Engelberth J, Tumlinson JH (2003) Differential volatile emissions and salicylic acid levels from tobacco plants in response to different strains of Pseudomonas syringae. Planta 217(5):767–775. https://doi.org/10.1007/s00425-003-1039-y
Huang B, Rachmilevitch S, Xu J (2012) Root carbon and protein metabolism associated with heat tolerance. J Exp Bot 63:3455–3465. https://doi.org/10.1093/jxb/ers003
Jacoby R, Peukert M, Succurro A, Koprivova A, Kopriva S (2017) The role of soil microorganisms in plant mineral nutrition-current knowledge and future directions. Front Plant Sci 8:1617. https://doi.org/10.3389/fpls.2017.01617
Jiang L, Gu W, Chao JG, Sang XH, Xi CC (2017) Gene cloning of farnesyl pyrophosphate synthase in Atractylodes lancea and its expression pattern analysis. Chin Tradit Herb Drugs 48:760–766. https://doi.org/10.7501/j.issn.0253-2670.2017.04.023
Johns C (2017) Living soils: the role of microorganisms in soil health. Fut Direct Intl, pp 1–7. https://apo.org.au/node/96931
Kaur C, Selvakumar G, Ganeshamurthy AN (2017) Burkholderia to Paraburkholderia: the journey of a plant-beneficial-environmental bacterium. In Recent advances in applied microbiology. Springer, Singapore, pp 213–228
Khan MA, Asaf S, Khan AL, Jan R, Kang S-M, Kim K-M, Lee I-J (2020) Thermotolerance effect of plant growth-promoting Bacillus cereus SA1 on soybean during heat stress. BMC Microbiol 20:1–14. https://doi.org/10.1186/s12866-020-01822-7
Kolde R, Kolde MR (2019) pheatmap: Pretty Heatmaps. R package version 1.0. 12. UR: https://CRAN.R-project.org/package=pheatmap
Lebeau A, Daunay MC, Frary A, Palloix A, Wang JF, Dintinger J, Chiroleu F, Wicker E, Prior P (2011) Bacterial wilt resistance in tomato, pepper, and eggplant: genetic resources respond to diverse strains in the Ralstonia solanacearum species complex. Phytopathology 101:154–165. https://doi.org/10.1094/PHYTO-02-10-0048
Li Q,, Zhao XL, Hu CR (2006) Iso10390: 2005 soil quality-determination of ph. Pollution Control Technology 19(1):53–55
Li X, De Boer W, Ding C, Zhang T, Wang X (2018) Suppression of soil-borne fusarium pathogens of peanut by intercropping with the medicinal herb Atractylodes lancea. Soil Biol Biochem 116:120–130. https://doi.org/10.1016/j.soilbio.2017.09.029
Ling Q, Huang W, Jarvis P (2011) Use of a SPAD-502 meter to measure leaf chlorophyll concentration in Arabidopsis thaliana. Photosynth Res 107:209–214. https://doi.org/10.1007/s11120-010-9606-0
Lipiec J, Doussan C, Nosalewicz A, Kondracka K (2013) Effect of drought and heat stresses on plant growth and yield: a review. Int Agrophysics 27:463–477. https://doi.org/10.2478/intag-2013-0017
Liu Q, Cao XY, Jiang JH, Dai CC (2007) Cloning and analysis of HMGR gene conserved fragments in Atractylodes lancea. Chin Tradit Herb Drugs 38:1551–1554. https://doi.org/10.3321/j.issn:0253-2670.2007.10.039
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2− ΔΔCT method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Lu QJ, Chao JG, Gu W, Zhang WM, Sang XH (2019) Effects of copper stress on accumulation of three medicinal compositions and expression of two key enzyme genes in biosynthesis. Chin Herb Med 50:710–715. https://doi.org/10.7501/j.issn.0253-2670.2019.03.026
Magoč T, Salzberg SL (2011) FLASH: fast length adjustment of short reads to improve genome assemblies. Bioinformatics 27:2957–2963. https://doi.org/10.1093/bioinformatics/btr507
Meher SP, Ashok Reddy K, Manohar Rao D (2018) Effect of PEG-6000 imposed drought stress on RNA content, relative water content (RWC), and chlorophyll content in peanut leaves and roots. Saudi J Biol Sci 25:285–289. https://doi.org/10.1016/j.sjbs.2017.04.008
Mendes LW, Kuramae EE, Navarrete AA, van Veen JA, Tsai SM (2014) Taxonomical and functional microbial community selection in soybean rhizosphere. ISME J 8:1577–1587. https://doi.org/10.1038/ismej.2014.17
Motaleb NA, Abd Elhady S, Ghoname A (2020) AMF and bacillus megaterium neutralize the harmful effects of salt stress on bean plants. Gesunde Pflanzen 72:29–39. https://doi.org/10.1007/s10343-019-00480-8
Nie JH (2018) A study on treating the Neizao syndrome from the spleen. Clin J Chin Med 10:1–4
Nik ZB, Mirza M, Ghaffari M (2008) Effect of drought stress on growth and essential oil contents in Parthenium argentatum gray. J Essent Oil Bearing Plants 11:423–429. https://doi.org/10.1080/0972060X.2008.10643649
Padukkage D, Geekiyanage S, Reparaz JM, Bezus R, Balatti PA, Degrassi G (2020) Bradyrhizobium japonicum, B. elkanii and B. diazoefficiens interact with Rice (Oryza sativa), promote growth and increase yield. Curr Microbiol. https://doi.org/10.1007/s00284-020-02249-z
Pang Z, Chen J, Wang T, Gao C, Li Z, Guo L, Xu J, Cheng Y (2021) Linking plant secondary metabolites and plant microbiomes: a review. Front Plant Sci 12:300. https://doi.org/10.3389/fpls.2021.621276
Parks DH, Tyson GW, Hugenholtz P, Beiko RG (2014) STAMP: statistical analysis of taxonomic and functional profiles. Bioinformatics 30:3123–3124. https://doi.org/10.1093/bioinformatics/btu494
Perez-Jaramillo JE, de Hollander M, Ramirez CA, Mendes R, Raaijmakers JM, Carrion VJ (2019) Deciphering rhizosphere microbiome assembly of wild and modern common bean (Phaseolus vulgaris) in native and agricultural soils from Colombia. Microbiome 7:114. https://doi.org/10.1186/s40168-019-0727-1
Phillips LA, Germida JJ, Farrell RE, Greer CW (2008) Hydrocarbon degradation potential and activity of endophytic bacteria associated with prairie plants. Soil Biol Biochem 40:3054–3064. https://doi.org/10.1016/j.soilbio.2008.09.006
Porter JR, Gawith M (1999) Temperatures and the growth and development of wheat: a review. Eur J Agron 10:23–36. https://doi.org/10.1016/S1161-0301(98)00047-1
Prasad PVV, Staggenborg SA, Ristic Z (2008) Impacts of drought and/or heat stress on physiological, developmental, growth, and yield processes of crop plants. Response of crops to limited water: Understanding and modeling water stress effects on plant growth processes 1:301–355
Qi J, Song CP, Wang B, Zhou J, Kangasjärvi J, Zhu JK, Gong Z (2018) Reactive oxygen species signaling and stomatal movement in plant responses to drought stress and pathogen attack. J Integr Plant Biol 60:805–826. https://doi.org/10.1111/jipb.12654
Qiong W, Fengshan P, Xiaomeng X, Rafiq MT, Xiao’e Y, Bao C, Ying F (2021) Cadmium level and soil type played a selective role in the endophytic bacterial community of hyperaccumulator Sedum alfredii Hance. Chemosphere 263:127986. https://doi.org/10.1016/j.chemosphere.2020.127986
Qu LH, Tu JY, Cao GS, Zhao JW, Pan XL (2018) Study on dryness effect of Atractylodis Rhizoma based on theory of dry-dry and dryness-induced Yin deficiency. China J Chin Materia Medica 43:2705–2712. https://doi.org/10.19540/j.cnki.cjcmm.20180514.012
Reinhold-Hurek B, Bünger W, Burbano CS, Sabale M, Hurek T (2015) Roots shaping their microbiome: global hotspots for microbial activity. Annu Rev Phytopathol 53:403–424. https://doi.org/10.1146/annurev-phyto-082712-102342
Ren C-G, Dai C-C (2012) Jasmonic acid is involved in the signaling pathway for fungal endophyte-induced volatile oil accumulation of Atractylodes lancea plantlets. BMC Plant Biol 12:1–11. https://doi.org/10.1186/1471-2229-12-128
Revelle WR (Photographer) (2017) psych: Procedures for Personality and Psychological Research. Software. URL: https://CRAN.R-project.org/package=psych
Rybakova D, Cernava T, Köberl M, Liebminger S, Etemadi M, Berg G (2016) Endophytes-assisted biocontrol: novel insights in ecology and the mode of action of Paenibacillus. Plant Soil 405:125–140. https://doi.org/10.1007/s11104-015-2526-1
Sailaja B, Anjum N, Prasanth VV, Sarla N, Subrahmanyam D, Voleti S, Viraktamath B, Mangrauthia SK (2014) Comparative study of susceptible and tolerant genotype reveals efficient recovery and root system contributes to heat stress tolerance in rice. Plant Mol Biol Report 32:1228–1240. https://doi.org/10.1007/s11105-014-0728-y
Schimel JP, Bennett J (2004) Nitrogen mineralization: challenges of a changing paradigm. Ecology 85:591–602. https://doi.org/10.1890/03-8002
Schloss PD, Westcott SL, Ryabin T, Hall JR, Hartmann M, Hollister EB, Lesniewski RA, Oakley BB, Parks DH, Robinson CJ (2009) Introducing mothur: open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl Environ Microbiol 75:7537–7541. https://doi.org/10.1128/AEM.01541-09
Segura A, Udaondo Z, Molina L (2021) PahT regulates carbon fluxes in Novosphingobium sp. HR1a and influences its survival in soil and rhizospheres. Environ Microbiol. https://doi.org/10.1111/1462-2920.15509
Shakeel A, Chuansong Z, Yanyan Y, Xuekui W, Tewu Y, Zeying Z, Qiyun Z, Xiaohua L, Xuebo H, Ji-Hong L (2016) The transcript profile of a traditional Chinese medicine, Atractylodes lancea, revealing its Sesquiterpenoid biosynthesis of the major active components. PLoS One 11:e0151975. https://doi.org/10.1371/journal.pone.0151975
Sharma S, Kumar S, Khajuria A, Ohri P, Kaur R, Kaur R (2020) Biocontrol potential of chitinases produced by newly isolated Chitinophaga sp. S167. World J Microbiol Biotechnol 36:90. https://doi.org/10.1007/s11274-020-02864-9
Shi S, Chang J, Tian L, Nasir F, Ji L, Li X, Tian C (2019) Comparative analysis of the rhizomicrobiome of the wild versus cultivated crop: insights from rice and soybean. Arch Microbiol 201:879–888
Shi L, Zhang P, He Y, Zeng F, Xu J, He L (2021) Enantioselective effects of cyflumetofen on microbial community and related nitrogen cycle gene function in acid-soil. Sci Total Environ 771:144831. https://doi.org/10.1177/1747493018778713
Shivakrishna P, Reddy KA, Rao DM (2018) Effect of PEG-6000 imposed drought stress on RNA content, relative water content (RWC), and chlorophyll content in peanut leaves and roots. Saudi J Biol Sci 25:285–289. https://doi.org/10.1016/j.sjbs.2017.04.008
Siciliano SD, Palmer AS, Winsley T, Lamb E, Bissett A, Brown MV, van Dorst J, Ji M, Ferrari BC, Grogan P (2014) Soil fertility is associated with fungal and bacterial richness, whereas pH is associated with community composition in polar soil microbial communities. Soil Biol Biochem 78:10–20. https://doi.org/10.1016/j.soilbio.2014.07.005
Sousa JAJ, Olivares FL (2016) Plant growth promotion by streptomycetes: ecophysiology, mechanisms and applications. Chem Biol Technol Agric 3:24. https://doi.org/10.1186/s40538-016-0073-5
Tambussi EA, Bort J, Araus JL (2007) Water use efficiency in C3 cereals under Mediterranean conditions: a review of physiological aspects. Ann Appl Biol 150:307–321. https://doi.org/10.1111/j.1744-7348.2007.00143.x
Tsusaka T, Makino B, Ohsawa R, Ezura H (2019) Genetic and environmental factors influencing the contents of essential oil compounds in Atractylodes lancea. PloS one 14:e0217522. https://doi.org/10.1371/journal.pone.0151975
Vannier N, Mony C, Bittebiere A-K, Michon-Coudouel S, Biget M, Vandenkoornhuyse P (2018) A microorganisms’ journey between plant generations. Microbiome 6:79. https://doi.org/10.1186/s40168-018-0459-7
Vranová E, Coman D, Gruissem W (2013) Network analysis of the MVA and MEP pathways for isoprenoid synthesis. Annu Rev Plant Biol 64:665–700. https://doi.org/10.1146/annurev-arplant-050312-120116
Wan W, Qin Y, Wu H, Zuo W, He H, Tan J, Wang Y, He D (2020) Isolation and characterization of phosphorus solubilizing bacteria with multiple phosphorus sources utilizing capability and their potential for lead immobilization in soil. Front Microbiol 11:752. https://doi.org/10.3389/fmicb.2020.00752
Wang HX, Liu CM, Liu Q, Gao K (2008) Three types of sesquiterpenes from rhizomes of Atractylodes lancea. Phytochemistry 69:2088–2094. https://doi.org/10.1016/j.phytochem.2008.04.008
Wang TL, Guo LP, Zhang Y, Chen ML, Guan W (2016) Pathogen identification, regularity of development and control measures of diseases on Atractylodes lancea. Zhongguo Zhong Yao Za Zhi 41:2411–2415. https://doi.org/10.4268/cjcmm20161307
Wang J, Wang C, Li J, Bai P, Li Q, Shen M, Li R, Li T, Zhao J (2018) Comparative genomics of degradative Novosphingobium strains with special reference to microcystin-degrading Novosphingobium sp. THN1. Front Microbiol 9:2238. https://doi.org/10.3389/fmicb.2018.02238
Wang G, Jin Z, Wang X, George TS, Feng G, Zhang L (2022) Simulated root exudates stimulate the abundance of Saccharimonadales to improve the alkaline phosphatase activity in maize rhizosphere. Appl Soil Ecol 170:104274. https://doi.org/10.1177/1747493018778713
Wu Y-S, Yang C-Y (2019) Ethylene-mediated signaling confers thermotolerance and regulates transcript levels of heat shock factors in rice seedlings under heat stress. Bot Stud 60:1–12. https://doi.org/10.1186/s40529-019-0272-z
Xu N, Tan GC, Wang HY, Gai XP (2016) Effect of biochar additions to soil on nitrogen leaching, microbial biomass and bacterial community structure. Eur J Soil Biol 74:1–8. https://doi.org/10.1016/j.ejsobi.2016.02.004
Yan Z, Guo LP, Chen B, Li H, Lin S, Wu Z (2010) The effects of different temperature on the growth and components of essential oil of Atractylodes lancea (Thunb.) DC. World Sci Technol (Modern Tradit Chin Med Materia Medica) 12:773–778. https://doi.org/10.11842/wst.2010.5.[sequence]
Yang HR, Yuan J, Liu LH, Zhang W, Chen F, Dai CC (2019) Endophytic Pseudomonas fluorescens induced sesquiterpenoid accumulation mediated by gibberellic acid and jasmonic acid in Atractylodes macrocephala Koidz plantlets. Plant Cell Tissue Organ Cult 138:445–457. https://doi.org/10.1007/s11240-019-01640-4
Yao M, Rui J, Niu H, Heděnec P, Li J, He Z, Wang J, Cao W, Li X (2017) The differentiation of soil bacterial communities along a precipitation and temperature gradient in the eastern Inner Mongolia steppe. Catena 152:47–56. https://doi.org/10.1016/j.catena.2017.01.007
Yin C, Hulbert SH, Schroeder KL, Mavrodi O, Mavrodi D, Dhingra A, Schillinger WF, Paulitz TC (2013) Role of bacterial communities in the natural suppression of Rhizoctonia solani bare patch disease of wheat (Triticum aestivum L.). Appl Environ Microbiol 79:7428–7438. https://doi.org/10.1128/AEM.01610-13
Yuan L, Liu S, Zhu S, Chen G, Liu F, Zou M, Wang C (2016) Comparative response of two wucai (Brassica campestris L.) genotypes to heat stress on antioxidative system and cell ultrastructure in root. Acta Physiol Plant 38:1–8. https://doi.org/10.1007/s11738-016-2246-z
Yuan J, Zhang W, Sun K, Tang MJ, Chen PX, Li X, Dai CC (2019) Comparative transcriptomics and proteomics of Atractylodes lancea in response to endophytic fungus Gilmaniella sp. AL12 Reveals Regulation in Plant Metabolism. Front Microbiol 10:1208. https://doi.org/10.3389/fmicb.2019.01208
Zai X, Zhu S, Qin P, Wang X, Che L, Luo F (2012) Effect of Glomus mosseae on chlorophyll content, chlorophyll fluorescence parameters, and chloroplast ultrastructure of beach plum (Prunus maritima) under NaCl stress. Photosynthetica 50:323–328. https://doi.org/10.1007/s11099-012-0035-5
Zhang H, Xie X, Kim MS, Kornyeyev DA, Holaday S, Paré PW (2008) Soil bacteria augment Arabidopsis photosynthesis by decreasing glucose sensing and abscisic acid levels in planta. Plant J 56:264–273. https://doi.org/10.1111/j.1365-313X.2008.03593.x
Zhao CL, Cui XM, Chen YP, Liang Q (2010) Key enzymes of triterpenoid saponin biosynthesis and the induction of their activities and gene expressions in plants. Nat Prod Commun 5:1147–1158. https://doi.org/10.1002/minf.201000055
Zheng B-X, Bi Q-F, Hao X-L, Zhou G-W, Yang X-R (2017) Massilia phosphatilytica sp. nov., a phosphate solubilizing bacteria isolated from a long-term fertilized soil. Int J Syst Evol Microbiol 67:2514–2519. https://doi.org/10.1099/ijsem.0.001916
Zhong YJ, Yang YQ, Liu P, Xu RN, Rensing C, Fu XD, Liao H (2019) Genotype and rhizobium inoculation modulate the assembly of soybean rhizobacterial communities. Plant Cell Environ 42:2028–2044. https://doi.org/10.1111/pce.13519
Zhou JY, Li X, Zheng JY, Dai CC (2016) Volatiles released by endophytic Pseudomonas fluorescens promoting the growth and volatile oil accumulation in Atractylodes lancea. Plant Physiol Biochem 101:132–140. https://doi.org/10.1016/j.plaphy.2016.01.026
Acknowledgments
We acknowledge Stefana-Catrinel Catana BSc of University College London for polishing the English of this manuscript. This study was supported by the National Key Research and Development Program of China (No.2017YFC1700701, No.2017YFC1700704), the National Natural Science Foundation of China (No.81891014), the Ministry of Finance Central Level of the Special (No.2060302), the Fundamental Research Funds for the Central public welfare research institutes (ZZ13-036-2), and the Special fund for the construction of modern agricultural industrial technology system (CARS-21).
Funding
This study was supported by the National Key Research and Development Program of China (No.2017YFC1700701, No.2017YFC1700704), the National Natural Science Foundation of China (No.81891014), the Ministry of Finance Central Level of the Special (No.2060302), the Fundamental Research Funds for the Central public welfare research institutes (ZZ13–036-2), and the Special fund for the construction of modern agricultural industrial technology system (CARS-21).
Author information
Authors and Affiliations
Contributions
All authors contributed to the conception and design of this study. Material preparation, data measurements and analyses were performed by HW, YW, DJ, and ZX. The first draft of the manuscript was written by HW. The latest version of the manuscript was written by YW. HW and YW made the figures. All authors gave valuable suggestions on each version of the manuscript; all authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
All authors approve the publication of this work.
Conflict of interest
There are no moral and ethical issues or conflicts to declare in this paper.
Open access
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Additional information
Responsible Editor: Stijn Spaepen.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 11762 kb)
Rights and permissions
About this article
Cite this article
Wang, H., Wang, Y., Jiang, D. et al. Soil microbe inoculation alters the bacterial communities and promotes root growth of Atractylodes lancea under heat stress. Plant Soil 478, 371–389 (2022). https://doi.org/10.1007/s11104-022-05369-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-022-05369-6