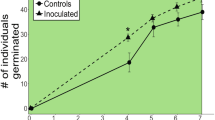
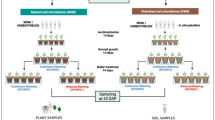
Abstract
Purpose
Rhizosphere bacterial communities (‘rhizobacteria’) can mediate plant-soil feedbacks that enhance plant drought tolerance, but the composition and ecological characteristics of these populations are poorly understood. We investigated the impact of drought on rhizobacteria communities and the effect of propagating drought-impacted communities on the drought tolerance of naïve plants in a two-phase experiment.
Methods
We tested whether propagating rhizosphere soils from drought-stressed shrub willow (Salix purpurea), or continuously watered controls, affected the performance of a subsequent generation of droughted plants. The impacts of drought on rhizobacteria and plant fitness were evaluated using 16S rRNA gene sequencing and measures of plant growth (root mass and stem length etc.) and stress (abscisic acid, osmolality). Uninoculated reference plants were used to isolate the effects of inoculation.
Results
Drought had a significant and lasting impact on the structure of rhizobacterial communities, characterized by increased populations of Actinobacteria (Acidimicrobiia, Thermoleophilia and Micrococcaceae). Foliar osmolality was significantly higher in plants receiving drought-selected communities relative to uninoculated controls, but no significant differences in plant growth were observed. Inoculation with rhizosphere soil from watered controls significantly reduced plant growth. The rhizosphere of watered control plants was dominated by Proteobacteria and Bacteroidetes.
Conclusions
Our findings demonstrate that plants are affected by the legacy effects of drought on the rhizosphere microbiome. This drought legacy was propagated and persisted throughout nine weeks of plant growth, independent of prevailing water stress. Drought-impacted rhizospheres had larger populations of desiccation-tolerant (ex. Arthrobacter) and putatively endophytic taxa (ex. Rhizobium) with established plant growth promoting capabilities.









Similar content being viewed by others
Data availability
Accessioned at the NCBI under BioProject PRJEB41348.
Abbreviations
- 16S rRNA gene:
-
16S small subunit ribosomal RNA gene.
- PCR:
-
Polymerase chain reaction
- DEA buffer:
-
Diethanolamine buffer
- PPFD:
-
Photosynthetic photon flux density
- ABA:
-
Abscisic acid
- E-phase:
-
Establishment phase, where plants are exposed to the first drought cycle.
- T-phase:
-
Testing phase, where plants are inoculated with soils from the E-phase and exposed to a second drought cycle.
- DRY:
-
Plants exposed to drought cycle treatment
- WET:
-
Well-watered, control plants
- REF:
-
Uninoculated sterile treatment
- D:
-
Plants exposed to a second drought cycle in T-phase
- C:
-
Plants continually well-watered in T-phase
References
Angly FE, Dennis PG, Skarshewski A et al (2014) CopyRighter: A rapid tool for improving the accuracy of microbial community profiles through lineage-specific gene copy number correction. Microbiome 2:1–13. https://doi.org/10.1186/2049-2618-2-11
Beckers B, De Beeck MO, Weyens N et al (2017) Structural variability and niche differentiation in the rhizosphere and endosphere bacterial microbiome of field-grown poplar trees. Microbiome 5:1–17. https://doi.org/10.1186/s40168-017-0241-2
Bolyen E, Rideout JR, Dillon MR et al (2019) Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat Biotechnol 37:852–857. https://doi.org/10.1038/s41587-019-0209-9
Bouskill NJ, Lim HC, Borglin S et al (2013) Pre-exposure to drought increases the resistance of tropical forest soil bacterial communities to extended drought. ISME J 7:384–394. https://doi.org/10.1038/ismej.2012.113
Callahan BJ, McMurdie PJ, Rosen MJ et al (2016) DADA2: High-resolution sample inference from Illumina amplicon data. Nat Methods 13:581–583. https://doi.org/10.1038/nmeth.3869
Cho SM, Kang BR, Kim YC (2013) Transcriptome analysis of induced systemic drought tolerance elicited by Pseudomonas chlororaphis O6 in Arabidopsis thaliana. Plant Pathol J 29:209–220. https://doi.org/10.5423/PPJ.SI.07.2012.0103
Chukwuneme CF, Babalola OO, Kutu FR, Ojuederie OB (2020) Characterization of actinomycetes isolates for plant growth promoting traits and their effects on drought tolerance in maize. J Plant Interact 15:93–105. https://doi.org/10.1080/17429145.2020.1752833
Comas LH, Becker SR, Cruz VMV et al (2013) Root traits contributing to plant productivity under drought. Front Plant Sci 4:1–16. https://doi.org/10.3389/fpls.2013.00442
Core Team R (2020) R: a language and environment for statistical computing. R Found Stat Comput
Czarnes S, Hallett P, Bengough A, Young I (2000) Root-and microbial-derived mucilages affect soil structure and water transport. Eur J Soil Sci 51:435–443
De Vries FT, Griffiths RI, Knight CG et al (2020) Harnessing rhizosphere microbiomes for drought-resilient crop production. Science 368:270–274. https://doi.org/10.1126/science.aaz5192
Doty S, Oakley B, Xin G et al (2009) Diazotrophic endophytes of native black cottonwood and willow. Symbiosis 47:23–33
Estendorfer J, Stempfhuber B, Haury P et al (2017) The influence of land use intensity on the plant-associated microbiome of Dactylis glomerata L. Front Plant Sci 8:1–10. https://doi.org/10.3389/fpls.2017.00930
Fan P, Chen D, He Y et al (2016) Alleviating salt stress in tomato seedlings using Arthrobacter and Bacillus megaterium isolated from the rhizosphere of wild plants grown on saline–alkaline lands. Int J Phytoremediation 18:1113–1121. https://doi.org/10.1080/15226514.2016.1183583
Fuchslueger L, Bahn M, Fritz K et al (2014) Experimental drought reduces the transfer of recently fixed plant carbon to soil microbes and alters the bacterial community composition in a mountain meadow. New Phytol 201:916–927. https://doi.org/10.1111/nph.12569
Giard-Laliberté C, Azarbad H, Tremblay J et al (2019) A water stress-adapted inoculum affects rhizosphere fungi, but not bacteria nor wheat. FEMS Microbiol Ecol 95:1–10. https://doi.org/10.1093/femsec/fiz080
Govindasamy V, George P, Kumar M et al (2020) Multi-trait PGP rhizobacterial endophytes alleviate drought stress in a senescent genotype of sorghum [Sorghum bicolor (L.) Moench]. 3 Biotech 10:1–14. https://doi.org/10.1007/s13205-019-2001-4
Gutiérrez-Luna FM, López-Bucio J, Altamirano-Hernández J et al (2010) Plant growth-promoting rhizobacteria modulate root-system architecture in Arabidopsis thaliana through volatile organic compound emission. Symbiosis 51:75–83. https://doi.org/10.1007/s13199-010-0066-2
Han B, Fu L, Zhang D et al (2016) Interspecies and intraspecies analysis of trehalose contents and the biosynthesis pathway gene family reveals crucial roles of trehalose in osmotic-stress tolerance in cassava. Int J Mol Sci 17:1–18. https://doi.org/10.3390/ijms17071077
Hartmann M, Brunner I, Hagedorn F et al (2017) A decade of irrigation transforms the soil microbiome of a semi-arid pine forest. Mol Ecol 26:1190–1206. https://doi.org/10.1111/mec.13995
Hofmann N (2008) Abscisic acid-mediated suppression of systemic acquired resistance signaling. Plant Cell 20:200612. https://doi.org/10.1105/tpc.108.200612
Igiehon NO, Babalola OO, Aremu BR (2019) Genomic insights into plant growth promoting rhizobia capable of enhancing soybean germination under drought stress. BMC Microbiol 19:1–22. https://doi.org/10.1186/s12866-019-1536-1
Jochum MD, McWilliams KL, Pierson EA, Jo YK (2019) Host-mediated microbiome engineering (HMME) of drought tolerance in the wheat rhizosphere. PLoS One 14:1–15. https://doi.org/10.1371/journal.pone.0225933
Kaisermann A, de Vries FT, Griffiths RI, Bardgett RD (2017) Legacy effects of drought on plant–soil feedbacks and plant–plant interactions. New Phytol 215:1413–1424. https://doi.org/10.1111/nph.14661
Kannenberg SA, Phillips RP (2017) Soil microbial communities buffer physiological responses to drought stress in three hardwood species. Oecologia 183:631–641. https://doi.org/10.1007/s00442-016-3783-2
Khan MA, Ullah I, Waqas M et al (2019) Halo-tolerant rhizospheric Arthrobacter woluwensis AK1 mitigates salt stress and induces physio-hormonal changes and expression of GmST1 and GmLAX3 in soybean. Symbiosis 77:9–21. https://doi.org/10.1007/s13199-018-0562-3
Kozich JJ, Westcott SL, Baxter NT et al (2013) Development of a dual-index sequencing strategy and curation pipeline for analyzing amplicon sequence data on the Miseq Illumina sequencing platform. Appl Environ Microbiol 79:5112–5120. https://doi.org/10.1128/AEM.01043-13
Kuzyakov Y, Razavi BS (2019) Rhizosphere size and shape : Temporal dynamics and spatial stationarity. Soil Biol Biochem 135:343–360. https://doi.org/10.1016/j.soilbio.2019.05.011
Lafi FF, Ramirez-Prado JS, Alam I et al (2017) Draft Genome Sequence of Plant Growth-Promoting Micrococcus luteus Strain K39 Isolated from Cyperus conglomeratus in Saudi Arabia. Genome Announc 5:e01520-e1616
Lafi FF, Ramirez-Prado JS, Alam I et al (2016) Draft genome sequence of Halomonas elongata strain K4, an endophytic growth-promoting bacterium enhancing salinity tolerance in planta. Genome Announc 4:1–2. https://doi.org/10.1128/genomeA.01214-16
Lata R, Chowdhury S, Gond SK, White JF (2018) Induction of abiotic stress tolerance in plants by endophytic microbes. Lett Appl Microbiol 66:268–276. https://doi.org/10.1111/lam.12855
Ledger T, Rojas S, Timmermann T et al (2016) Volatile-mediated effects predominate in Paraburkholderia phytofirmans growth promotion and salt stress tolerance of Arabidopsis thaliana. Front Microbiol 7:1–18. https://doi.org/10.3389/fmicb.2016.01838
Lenth R, Singmann HJL (2018) Emmeans: Estimated marginal means, aka least-squares means
Lim JH, Kim SD (2013) Induction of drought stress resistance by multi-functional PGPR Bacillus licheniformis K11 in pepper. Plant Pathol J 29:201–208. https://doi.org/10.5423/PPJ.SI.02.2013.0021
Liu F, Xing S, Ma H et al (2013) Cytokinin-producing, plant growth-promoting rhizobacteria that confer resistance to drought stress in Platycladus orientalis container seedlings. Appl Microbiol Biotechnol 97:9155–9164. https://doi.org/10.1007/s00253-013-5193-2
López-Mondéjar R, Zühlke D, Becher D et al (2016) Cellulose and hemicellulose decomposition by forest soil bacteria proceeds by the action of structurally variable enzymatic systems. Sci Rep 6:1–12. https://doi.org/10.1038/srep25279
Love MI, Huber W, Anders S (2014) Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol 15:1–21. https://doi.org/10.1186/s13059-014-0550-8
Lozupone C, Lladser ME, Knights D et al (2011) UniFrac: An effective distance metric for microbial community comparison. ISME J 5:169–172. https://doi.org/10.1038/ismej.2010.133
Lu T, Ke M, Lavoie M et al (2018) Rhizosphere microorganisms can influence the timing of plant flowering. Microbiome 6:1–12. https://doi.org/10.1186/s40168-018-0615-0
Lunn JE, Delorge I, Figueroa CM et al (2014) Trehalose metabolism in plants. Plant J 79:544–567. https://doi.org/10.1111/tpj.12509
Marasco R, Rolli E, Ettoumi B et al (2012) A drought resistance-promoting microbiome is selected by root system under desert farming. PLoS One. https://doi.org/10.1371/journal.pone.0048479
Martiny JBH, Martiny AC, Weihe C et al (2017) Microbial legacies alter decomposition in response to simulated global change. ISME J 11:490–499. https://doi.org/10.1038/ismej.2016.122
Mayak S, Tirosh T, Glick BR (2004) Plant growth-promoting bacteria that confer resistance to water stress in tomatoes and peppers. Plant Sci 166:525–530. https://doi.org/10.1016/j.plantsci.2003.10.025
Mayrberger JM (2011) Studies of genera Cytophaga-Flavobacterium in context of the soil carbon cycle. Michigan State University
McMurdie PJ, Holmes S (2013) Phyloseq: an R package for reproducible interactive analysis and graphics of microbiome census data. PLoS One. https://doi.org/10.1371/journal.pone.0061217
Mendes R, Garbeva P, Raaijmakers JM (2013) The rhizosphere microbiome: Significance of plant beneficial, plant pathogenic, and human pathogenic microorganisms. FEMS Microbiol Rev 37:634–663. https://doi.org/10.1111/1574-6976.12028
Naylor D, Coleman-Derr D (2018) Drought stress and root-associated bacterial communities. Front Plant Sci 8:1–16. https://doi.org/10.3389/fpls.2017.02223
Nehl DB, Allen SJ, Brown JF (1997) Deleterious rhizosphere bacteria: An integrating perspective. Appl Soil Ecol 5:1–20. https://doi.org/10.1016/S0929-1393(96)00124-2
Ober ES, Setter TL (1992) Water deficit induces abscisic acid accumulation in endosperm of maize viviparous mutants. Plant Physiol 98:353–356. https://doi.org/10.1104/pp.98.1.353
Oksanen J, Blanchet F, Kindt R et al (2015) Vegan: community ecology package. R Packag
Olson MS, Levsen N, Soolanayakanahally RY et al (2013) The adaptive potential of Populus balsamifera L. to phenology requirements in a warmer global climate. Mol Ecol 22:1214–1230. https://doi.org/10.1111/mec.12067
Panke-Buisse K, Poole AC, Goodrich JK et al (2015) Selection on soil microbiomes reveals reproducible impacts on plant function. ISME J 9:980–989. https://doi.org/10.1038/ismej.2014.196
Pirttilä AM, Joensuu P, Pospiech H et al (2004) Bud endophytes of Scots pine produce adenine derivatives and other compounds that affect morphology and mitigate browning of callus cultures. Physiol Plant 121:305–312. https://doi.org/10.1111/j.0031-9317.2004.00330.x
Preece C, Verbruggen E, Liu L et al (2019) Effects of past and current drought on the composition and diversity of soil microbial communities. Soil Biol Biochem 131:28–39. https://doi.org/10.1016/j.soilbio.2018.12.022
Qin S, Feng W-W, Zhang Y-J et al (2018) Diversity of bacterial microbiota of coastal halophyte Limonium sinense and amelioration of salinity stress damage. Appl Environ Microbiol 84:1–19
Quast C, Pruesse E, Yilmaz P et al (2013) The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res 41:590–596. https://doi.org/10.1093/nar/gks1219
Rodríguez-Salazar J, Suárez R, Caballero-Mellado J, Iturriaga G (2009) Trehalose accumulation in Azospirillum brasilense improves drought tolerance and biomass in maize plants. FEMS Microbiol Lett 296:52–59. https://doi.org/10.1111/j.1574-6968.2009.01614.x
Rolli E, Marasco R, Vigani G et al (2015) Improved plant resistance to drought is promoted by the root-associated microbiome as a water stress-dependent trait. Environ Microbiol 17:316–331. https://doi.org/10.1111/1462-2920.12439
Rubin RL, van Groenigen KJ, Hungate BA (2017) Plant growth promoting rhizobacteria are more effective under drought: a meta-analysis. Plant Soil 416:309–323. https://doi.org/10.1007/s11104-017-3199-8
Sandhya V, Shrivastava M, Ali SZ, Prasad SSK, V, (2017) Endophytes from maize with plant growth promotion and biocontrol activity under drought stress. Russ Agric Sci 43:22–34. https://doi.org/10.3103/s1068367417010165
Santos-Medellín C, Edwards J, Liechty Z et al (2017) Drought stress results in a compartment-specific restructuring of the rice root-associated microbiomes. MBio 8:1–15. https://doi.org/10.1128/mBio.00764-17
Savage JA, Cavender-Bares JM (2011) Contrasting drought survival strategies of sympatric willows (genus: Salix): Consequences for coexistence and habitat specialization. Tree Physiol 31:604–614. https://doi.org/10.1093/treephys/tpr056
Seaton FM, Jones DL, Creer S et al (2019) Science of the Total Environment Plant and soil communities are associated with the response of soil water repellency to environmental stress. Sci Total Environ 687:929–938. https://doi.org/10.1016/j.scitotenv.2019.06.052
Shade A, Hogan CS, Klimowicz AK et al (2012) Culturing captures members of the soil rare biosphere. Environ Microbiol 14:2247–2252. https://doi.org/10.1111/j.1462-2920.2012.02817.x
Shcherbakova E, Andronov E, Shcherbakov A et al (2016) Bacterial endophytes from seeds of conifers with potential role in biocontrol: microbiome analysis. In: Preceedings of the Meeting Biocontrol and Microbial Ecology pp 192–199
Siddikee MA, Chauhan PS, Anandham R et al (2010) Isolation, characterization, and use for plant growth promotion under salt stress, of ACC deaminase-producing halotolerant bacteria derived from coastal soil. J Microbiol Biotechnol 20:1577–1584. https://doi.org/10.4014/jmb.1007.07011
Stoof CR, Richards BK, Woodbury PB et al (2015) Untapped Potential: Opportunities and challenges for sustainable bioenergy production from marginal lands in the northeast USA. Bioenergy Res 8:482–501. https://doi.org/10.1007/s12155-014-9515-8
Sziderics AH, Rasche F, Trognitz F et al (2007) Bacterial endophytes contribute to abiotic stress adaptation in pepper plants (Capsicum annuum L.). Can J Microbiol 53:1195–1202. https://doi.org/10.1139/W07-082
Tardif S, Yergeau É, Tremblay J et al (2016) The willow microbiome is influenced by soil petroleum-hydrocarbon concentration with plant compartment-specific effects. Front Microbiol 7:1–14. https://doi.org/10.3389/fmicb.2016.01363
Timmusk S, Abd El-Daim IA, Copolovici L et al (2014) Drought-tolerance of wheat improved by rhizosphere bacteria from harsh environments: Enhanced biomass production and reduced emissions of stress volatiles. PLoS One. https://doi.org/10.1371/journal.pone.0096086
Ulrich K, Ulrich A, Ewald D (2008) Diversity of endophytic bacterial communities in poplar grown under field conditions. FEMS Microbiol Ecol 63:169–180. https://doi.org/10.1111/j.1574-6941.2007.00419.x
Vílchez JI, García-Fontana C, Román-Naranjo D et al (2016) Plant drought tolerance enhancement by trehalose production of desiccation-tolerant microorganisms. Front Microbiol 7:1–11. https://doi.org/10.3389/fmicb.2016.01577
Wang W (2018) Genomics of genotype-by-environment interactions in shrub willow (Salix spp.): Insect herbivory and soil microbiomes. The Pennsylvania State University
Wickham H (2009) Elegant graphics for data analysis. Media 35:211. https://doi.org/10.1007/978-0-387-98141-3
Wilhelm RC, Munoz-Ucros J, Weikl F et al (2021a) The effects of root mixing on the response of beech and spruce forest soil microbiomes to reduced precipitation. bioRxiv
Wilhelm RC, Pepe-Ranney C, Weisenhorn P et al (2021b) Competitive exclusion and metabolic dependency among microorganisms structure the cellulose economy of an agricultural soil. Mbio 12:1–19. https://doi.org/10.1128/mBio.03099-20
Wilhelm RC, Singh R, Eltis LD, Mohn WW (2019) Bacterial contributions to delignification and lignocellulose degradation in forest soils with metagenomic and quantitative stable isotope probing. ISME J 13:413–429. https://doi.org/10.1038/s41396-018-0279-6
Wipf HML, Bùi TN, Coleman-Derr D (2021) Distinguishing between the impacts of heat and drought stress on the root microbiome of Sorghum bicolor. Phytobiomes J 5:166–176. https://doi.org/10.1094/PBIOMES-07-20-0052-R
Wubs ERJ, Bezemer TM (2016) Effects of spatial plant-soil feedback heterogeneity on plant performance in monocultures. J Ecol 104:364–376. https://doi.org/10.1111/1365-2745.12521
Yasuda M, Ishikawa A, Jikumaru Y et al (2008) Antagonistic interaction between systemic acquired resistance and the abscisic acid-mediated abiotic stress response in Arabidopsis. Plant Cell 20:1678–1692. https://doi.org/10.1105/tpc.107.054296
Yergeau E, Bell TH, Champagne J et al (2015) Transplanting soil microbiomes leads to lasting effects on willow growth, but not on the rhizosphere microbiome. Front Microbiol 6:1–14. https://doi.org/10.3389/fmicb.2015.01436
Yogendra SG, U. SS, A. KS, (2015) Bacterial mediated amelioration of drought stress in drought tolerant and susceptible cultivars of rice (Oryza sativa L.). African J Biotechnol 14:764–773. https://doi.org/10.5897/ajb2015.14405
Zevenhuizen LPTM (1992) Levels of trehalose and glycogen in Arthrobacter globiformis under conditions of nutrient starvation and osmotic stress. Antonie Van Leeuwenhoek 61:61–68. https://doi.org/10.1007/BF00572124
Zhang L, Zhang W, Li Q et al (2020) Deciphering the Root Endosphere Microbiome of the Desert Plant Alhagi sparsifolia for Drought Resistance-Promoting Bacteria. Appl Environmantal Microbiol 86:e02863-e2919
Zhang W, Yang K, Lyu Z, Zhu J (2019) Microbial groups and their functions control the decomposition of coniferous litter: A comparison with broadleaved tree litters. Soil Biol Biochem 133:196–207. https://doi.org/10.1016/j.soilbio.2019.03.009
Zheng W, Zeng S, Bais H, Lamanna JM (2018) Plant growth-promoting rhizobacteria (PGPR) reduce evaporation and increase soil water retention. Water Resour Res 54:3673–3687. https://doi.org/10.1029/2018WR022656
Zwetsloot MJ, Muñoz Ucros J, Wickings K et al (2020) Prevalent root-derived phenolics drive shifts in microbial community composition and prime decomposition in forest soil. Soil Biol Biochem 145:530–541. https://doi.org/10.1016/j.soilbio.2020.107797
Acknowledgements
We would like to thank Larry Smart and his research group for providing plant material, growing advice and access to plantings; Emily Detrick and the Cornell Botanic Gardens for allowing us to collect soil; Hannah Swegarden and Zachary Stansell for collecting and transporting plant materials and soil, and Tim Setter for ABA assays.
Funding
Funding was provided by the Sustainable Biodiversity Fund from the Atkinson Center for a Sustainable Future and the Schmittau-Novak small grants program the School of Integrative Plant Science to JMU.
Author information
Authors and Affiliations
Contributions
JMU, RCW, DHB and TLB conceived and designed experiments. JMU performed the experiments. RCW and JMU analyzed the data and wrote the manuscript with contributions from all authors.
Corresponding author
Ethics declarations
Consent for publication
All authors consent to the publication of this work in Plant and Soil.
Conflicts of interest/Competing interests
The authors report no conflict of interest.
Additional information
Responsible Editor: Ricardo Aroca
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Munoz-Ucros, J., Wilhelm, R.C., Buckley, D.H. et al. Drought legacy in rhizosphere bacterial communities alters subsequent plant performance. Plant Soil 471, 443–461 (2022). https://doi.org/10.1007/s11104-021-05227-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-021-05227-x