Abstract
Aims
Here, we assess the differential impact of drought on root carbon metabolism in the widely cultivated alfalfa (Medicago sativa, Ms) and the model legume Medicago truncatula (Mt). Understanding how carbon allocation is regulated under drought stress conditions is a central issue to improving alfalfa productivity under future climate change scenarios.
Methods
Alfalfa and Medicago truncatula were compared under water deficit conditions. Root carbon metabolism of the taproot and fibrous roots was analysed. M. truncatula drought tolerance variability was compared to that of alfalfa using six accessions of the Medicago Hapmap project. The prominent taproot is much less developed in M. truncatula than in alfalfa with the former exhibiting an extensive fibrous root system.
Results
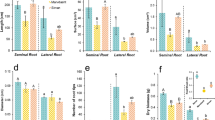
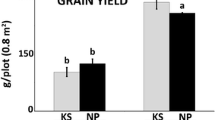
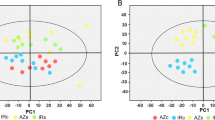
In both examined Medicago species the taproot contained the major pools of soluble protein, sucrose and pinitol, whereas the major pools of hexoses and carbon metabolism enzymes appeared to be in the fibrous roots. Under water-deficit conditions, the response of M. sativa strongly differed from that of M. truncatula at the root level.
Conclusions
Water deficit conditions differentially modulate the root carbon metabolism of M. sativa and M. truncatula. Mt maintained a more active carbon metabolism in the fibRs, as sucrose, myo-inositol and pinitol accumulated to cope with the water deficit (WD). Conversely, the root system of Ms did not accumulate cyclitols and carbon metabolism was more severely affected under water deficit conditions. This differentially exerted control may determine the drought response of these two close relatives.







Similar content being viewed by others
Abbreviations
- C:
-
control
- DW:
-
dry weight
- G6PDH:
-
glucose-6-phosphate dehydrogenase
- fibRs:
-
fibrous roots
- FW:
-
fresh weight
- g s :
-
stomatal conductance
- INV:
-
alkaline invertase
- MD:
-
moderate deficit
- Ms :
-
Medicago sativa
- Mt :
-
Medicago truncatula
- SuSy:
-
sucrose synthase
- T:
-
transpiration
- tapRs:
-
taproots
- WC:
-
water content
- WD:
-
water deficit
References
Al-Suod H, Ligor M, Rațiu IA et al (2017) A window on cyclitols: characterization and analytics of inositols. Phytochem Lett 20:507–519. https://doi.org/10.1016/j.phytol.2016.12.009
Araújo SS, Beebe S, Crespi M, Delbreil B, González EM, Gruber V, Lejeune-Henaut I, Link W, Monteros MJ, Prats E, Rao I, Vadez V, Patto MCV (2015) Abiotic stress responses in legumes: strategies used to cope with environmental challenges. CRC Crit Rev Plant Sci 34:237–280. https://doi.org/10.1080/07352689.2014.898450
Aubert G, Morin J, Jacquin F, Loridon K, Quillet MC, Petit A, Rameau C, Lejeune-Hénaut I, Huguet T, Burstin J (2006) Functional mapping in pea, as an aid to the candidate gene selection and for investigating synteny with the model legume Medicago truncatula. Theor Appl Genet 112:1024–1041. https://doi.org/10.1007/s00122-005-0205-y
Barker DG, Bianchl S, Blondon F et al (1990) Medicago truncatula, a model plant for studying the molecular genetics of the rhizobium-legume symbiosis. Plant Mol Biol Rep 8:40–49. https://doi.org/10.1007/BF02668879
Barratt DHP, Derbyshire P, Findlay K, Pike M, Wellner N, Lunn J, Feil R, Simpson C, Maule AJ, Smith AM (2009) Normal growth of Arabidopsis requires cytosolic invertase but not sucrose synthase. PNAS 106:13124–13129. https://doi.org/10.1073/pnas.0900689106
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Castañeda V, de la Peña M, Azcárate L, Aranjuelo I, Gonzalez EM (2019) Functional analysis of the taproot and fibrous roots of Medicago truncatula: sucrose and proline catabolism primary response to water deficit. Agric Water Manag 216:473–483. https://doi.org/10.1016/j.agwat.2018.07.018
Chaves MM, Flexas J, Pinheiro C (2009) Photosynthesis under drought and salt stress: regulation mechanisms from whole plant to cell. Ann Bot 103:551–560. https://doi.org/10.1093/aob/mcn125
Chaves MM, Maroco JP, Pereira JS (2003) Understanding plant responses to drought — from genes to the whole plant. Funct Plant Biol 30:239–264. https://doi.org/10.1071/fp02076
Chimungu JG, Brown KM, Lynch JP (2014) Reduced root cortical cell file number improves drought tolerance in maize. Plant Physiol 166:1943–1955. https://doi.org/10.1104/pp.114.249037
Ciereszko I, Kleczkowski LA (2002) Glucose and mannose regulate the expression of a major sucrose synthase gene in Arabidopsis via hexokinase-dependent mechanisms. Plant Physiol Biochem 40:907–911. https://doi.org/10.1016/S0981-9428(02)01452-3
Dai A (2011) Drought under global warming: a review. Wiley Interdiscip Rev Clim Chang 2:45–65. https://doi.org/10.1002/wcc.81
Dejardin A, Rochat C, Maugenest S, Boutin J-P (1997) Purification, characterization and physiological role of sucrose synthase in the pea seed coat (Pisum sativum L.). Planta 201:128–137. https://doi.org/10.1007/bf01007697
Erice G, Irigoyen JJ, Sánchez-Díaz M, Avice JC, Ourry A (2007) Effect of drought, elevated CO2 and temperature on accumulation of N and vegetative storage proteins (VSP) in taproot of nodulated alfalfa before and after cutting. Plant Sci 172:903–912. https://doi.org/10.1016/j.plantsci.2006.12.013
Evans HJ (1981) Symbiotic nitrogen fixation in legume nodules. In TC Moore, ed, Research Experiences in Plant Physiology. Springer-Verlag, New York, pp 294–310
Foley JA, Ramankutty N, Brauman KA et al (2011) Solutions for a cultivated planet. https://doi.org/10.1038/nature10452
Frechilla S, Gonzalez EM, Royuela M et al (2000) Source of nitrogen nutrition (nitrogen fixation or nitrate assimilation) is a major factor involved in pea response to moderate water stress. J Plant Physiol 157:609–617. https://doi.org/10.1016/S0176-1617(00)80003-6
Gálvez L, González EM, Arrese-Igor C (2005) Evidence for carbon flux shortage and strong carbon/nitrogen interactions in pea nodules at early stages of water stress. J Exp Bot 56:2551–2561. https://doi.org/10.1093/jxb/eri249
Gargallo-Garriga A, Sardans J, Pérez-Trujillo M, Rivas-Ubach A, Oravec M, Vecerova K, Urban O, Jentsch A, Kreyling J, Beierkuhnlein C, Parella T, Peñuelas J (2014) Opposite metabolic responses of shoots and roots to drought. Sci Rep 4:1–7. https://doi.org/10.1038/srep06829
Gibon Y, Blaesing OE, Hannemann J, Carillo P, Höhne M, Hendriks JHM, Palacios N, Cross J, Selbig J, Stitt M (2004) A robot-based platform to measure multiple enzyme activities in Arabidopsis using a set of cycling assays: comparison of changes of enzyme activities and transcript levels during diurnal cycles and in prolonged darkness. Plant Cell 16:3304–3325. https://doi.org/10.1105/tpc.104.025973
Gonzalez EM, Aparicio-Tejo PM, Gordon AJ, Minchin FR, Royuela M, Arrese-Igor C (1998) Water-deficit effects on carbon and nitrogen metabolism of pea nodules. J Exp Bot 49:1705–1714. https://doi.org/10.1093/jxb/49.327.1705
Haigler C (2001) Carbon partitioning to cellulose synthesis. Plant Mol Biol 47:29–51. https://doi.org/10.1023/A:1010615027986
Hsiao TC, Acevedo E (1974) Plant responses to water deficits, water use efficiency, and drought resistance. Ag Meteorogical 14:59–84
Huang Z, Liu Y, Cui Z, Fang Y, He H, Liu BR, Wu GL (2018) Soil water storage deficit of alfalfa (Medicago sativa) grasslands along ages in arid area (China). F Crop Res 221:1–6. https://doi.org/10.1016/j.fcr.2018.02.013
Humphries AW, Auricht GC (2001) Breeding lucerne for Australia’s southern dryland cropping environments. Aust J Agric Res 52:153–169. https://doi.org/10.1071/AR99171
Kang Y, Han Y, Torres-Jerez I, Wang M, Tang Y, Monteros M, Udvardi M (2011) System responses to long-term drought and re-watering of two contrasting alfalfa varieties. Plant J 68:871–889. https://doi.org/10.1111/j.1365-313X.2011.04738.x
Kang Y, Sakiroglu M, Krom N, Stanton-Geddes J, Wang M, Lee YC, Young ND, Udvardi M (2015) Genome-wide association of drought-related and biomass traits with HapMap SNPs in Medicago truncatula. Plant Cell Environ 38:1997–2011. https://doi.org/10.1111/pce.12520
Kirova E, Tzvetkova N, Vaseva I, Ignatov G (2008) Photosynthetic responses of nitrate-fed and nitrogen-fixing soybeans to progressive water stress. J Plant Nutr 31:445–458. https://doi.org/10.1080/01904160801894988
Koch KE (1996) Carbohydrate-modulated gene expression in plants. https://doi.org/10.1146/annurev.arplant.47.1.509
Lahuta LB, Ciak M, Rybiński W, Bocianowski J, Börner A (2018) Diversity of the composition and content of soluble carbohydrates in seeds of the genus Vicia (Leguminosae). Genet Resour Crop Evol 65:541–554. https://doi.org/10.1007/s10722-017-0552-y
Lipiec J, Doussan C, Nosalewicz A (2013) Effect of drought and heat stresses on plant growth and yield: a review. 2015–2020. https://doi.org/10.2478/intag-2013-0017
Lobell D, Burke M, Tebaldi C, et al (2008) Prioritizing climate change adaptation needs for food security in 2030. 319:607–611. https://doi.org/10.1126/science.1151194
Lodeiro AR, González P, Hernández A et al (2000) Comparison of drought tolerance in nitrogen-fixing and inorganic nitrogen-grown common beans. Plant Sci 154:31–41. https://doi.org/10.1016/S0168-9452(99)00246-0
Loewus FA, Murthy PPN (2000) Myo-inositol metabolism in plants. Plant Sci 150:1–19. https://doi.org/10.1016/S0168-9452(99)00150-8
Lynch JP, Brown KM (2012) New roots for agriculture: exploiting the root phenome. Philos Trans R Soc B Biol Sci 367:1598–1604. https://doi.org/10.1098/rstb.2011.0243
Michaud R (1988) World distribution and historical development. In: Hanson A (ed) Alfalfa and alfalfa improvement, vol. 29. American society of agronomy. Madison, Wisconsin, pp. 259–291
Morell M, Copeland L (1984) Enzymes of sucrose breakdown in soybean nodules. Plant Physiol 74:1030–1034. https://doi.org/10.1104/pp.74.4.1030
Naya L, Ladrera R, Ramos J, González EM, Arrese-Igor C, Minchin FR, Becana M (2007) The response of carbon metabolism and antioxidant defenses of alfalfa nodules to drought stress and to the subsequent recovery of plants. Plant Physiol 144:1104–1114. https://doi.org/10.1104/pp.107.099648
Paez-Garcia A, Motes CM, Scheible WR, Chen R, Blancaflor E, Monteros M (2015) Root traits and phenotyping strategies for plant improvement. Plants 4:334–355. https://doi.org/10.3390/plants4020334
Phan HTT, Ellwood SR, Hane JK, Ford R, Materne M, Oliver RP (2007) Extensive macrosynteny between Medicago truncatula and Lens culinaris ssp. culinaris. Theor Appl Genet 114:549–558. https://doi.org/10.1007/s00122-006-0455-3
Pignocchi C, Ivakov A, Feil R, Trick M, Pike M, Wang TL, Lunn JE, Smith AM (2020) Restriction of cytosolic sucrose hydrolysis profoundly alters development, metabolism, and gene expression in Arabidopsis roots. J Exp Bot 72:1850–1863. https://doi.org/10.1093/jxb/eraa581
Quan W, Liu X, Wang H, Chan Z (2016) Comparative physiological and transcriptional analyses of two contrasting drought tolerant alfalfa varieties. Front Plant Sci 6:1–16. https://doi.org/10.3389/fpls.2015.01256
Radovic J, Sokolovic D, Markovic J (2009) Alfalfa-most important perennial forage legume in animal husbandry. Biotechnol Anim Husb 25:465–475. https://doi.org/10.2298/bah0906465r
Rieger M, Litvin P (1999) Root system hydraulic conductivity in species with contrasting root anatomy. J Exp Bot 50:201–209. https://doi.org/10.1093/jxb/50.331.201
Rothstein SJ (2007) Returning to our roots: Making plant biology research relevant to future challenges in agriculture 19:2695–2699. https://doi.org/10.1105/tpc.107.053074
Ruan Y, Jin Y, Yang Y et al (2010) Sugar input , metabolism , and signaling mediated by invertase: Roles in development , yield potential , and response to drought and heat. Mol Plant 3:942–955. https://doi.org/10.1093/mp/ssq044
Ruan YL (2014) Sucrose metabolism: gateway to diverse carbon use and sugar signaling. Annu Rev Plant Biol 65:33–67. https://doi.org/10.1146/annurev-arplant-050213-040251
Scholander PF, Bradstreet ED, Hemmingsen EA, Hammel HT (1965) Sap pressure in vascular plants: negative hydrostatic pressure can be measured in plants. Science 148:339–346. https://doi.org/10.1126/science.148.3668.339
Sinclair TR (2011) Challenges in breeding for yield increase for drought. Trends Plant Sci 16:289–293. https://doi.org/10.1016/j.tplants.2011.02.008
Smith AE, Phillips DV (1982) Influence of sequential prolonged periods of dark and light on pinitol concentration in clover and soybean tissue. Physiol Plant 54:31–33. https://doi.org/10.1111/j.1399-3054.1982.tb00572.x
Soba D, Zhou B, Arrese-Igor C, Munné-Bosch S, Aranjuelo I (2019) Physiological, hormonal and metabolic responses of two alfalfa cultivars with contrasting responses to drought. Int J Mol Sci 20. https://doi.org/10.3390/ijms20205099
Stanton-geddes J, Paape T, Epstein B, et al (2013) Candidate genes and genetic architecture of symbiotic and agronomic traits revealed by whole-genome , Sequence-Based Association Genetics in Medicago truncatula 8:1–9. https://doi.org/10.1371/journal.pone.0065688
Sturm A (1999) Invertases: primary structures, functions and roles in plant development and sucrose partitioning. Plant Physiol 121:1–7. https://doi.org/10.1104/pp.121.1.1
Taiz L, Zeiger E (2010) Plant physiology. Sinauer Associates
Tian H, De Smet I, Ding Z (2014) Shaping a root system: regulating lateral versus primary root growth. Trends Plant Sci 19:426–431. https://doi.org/10.1016/j.tplants.2014.01.007
Valluru R, Van den Ende W (2011) Myo-inositol and beyond - emerging networks under stress. Plant Sci 181:387–400. https://doi.org/10.1016/j.plantsci.2011.07.009
Welham T, Pike J, Horst I, Flemetakis E, Katinakis P, Kaneko T, Sato S, Tabata S, Perry J, Parniske M, Wang TL (2009) A cytosolic invertase is required for normal growth and cell development in the model legume, Lotus japonicus. J Exp Bot 60:3353–3365. https://doi.org/10.1093/jxb/erp169
Winter H, Huber SC (2000) Regulation of sucrose metabolism in higher plants: localization and regulation of activity of key enzymes. Crit Rev Biochem Mol Biol 35:253–289. https://doi.org/10.1080/10409230008984165
Xiang L, Li Y, Rolland F, van den Ende W (2011) Neutral invertase, hexokinase and mitochondrial ROS homeostasis: emerging links between sugar metabolism, sugar signaling and ascorbate synthesis. Plant Signal Behav 6:1567–1573. https://doi.org/10.4161/psb.6.10.17036
Ye H, Roorkiwal M, Valliyodan B, Zhou L, Chen P, Varshney RK, Nguyen HT (2018) Genetic diversity of root system architecture in response to drought stress in grain legumes. J Exp Bot 69:3267–3277. https://doi.org/10.1093/jxb/ery082
Yoder JB, Stanton-Geddes J, Zhou P, Briskine R, Young ND, Tiffin P (2014) Genomic signature of adaptation to climate in Medicago truncatula. Genetics 196:1263–1275. https://doi.org/10.1534/genetics.113.159319
Young ND, Udvardi M (2009) Translating Medicago truncatula genomics to crop legumes. Curr Opin Plant Biol 12:193–201. https://doi.org/10.1016/J.PBI.2008.11.005
Zhang C, Shi S, Liu Z, Yang F, Yin G (2019) Drought tolerance in alfalfa (Medicago sativa L.) varieties is associated with enhanced antioxidative protection and declined lipid peroxidation. J Plant Physiol 232:226–240. https://doi.org/10.1016/J.JPLPH.2018.10.023
Zhang JY, Cruz De Carvalho MH, Torres-Jerez I et al (2014) Global reprogramming of transcription and metabolism in Medicago truncatula during progressive drought and after rewatering. Plant Cell Environ 37:2553–2576. https://doi.org/10.1111/pce.12328
Zhang T, Yu L-X, Zheng P et al (2015) Identification of loci associated with drought resistance traits in heterozygous autotetraploid alfalfa (Medicago sativa L.) using genome-wide association studies with genotyping by sequencing. PLoS one 10:e0138931. https://doi.org/10.1371/journal.pone.0138931
Zrenner R, Salanoubat M, Willmitzer L, Sonnewald U (1995) Evidence of the crucial role of sucrose synthase for sink strength using transgenic potato plants (Solanum tuberosum L.). Plant J 7:97–107. https://doi.org/10.1046/j.1365-313x.1995.07010097.x
Acknowledgements
Andres Echeverria was supported by a predoctoral fellowship from the Government of Navarra. We would like to thank Dr. Gustavo Garijo for technical assistance and the seed bank of the Medicago truncatula Hapmap Project consortium for supplying seeds.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible Editor: Ian Dodd.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Fig S1
(a) Percentage of transpiration (T) and (b) stomatal conductance (gs) of WD-treated plants, expressed as a percentage of the control levels in Medicago sativa (Ms) and Medicago truncatula HM307 (Mt. HM307) during the first 4 days of WD treatment which lasted for 7 days. Bars represent the mean ± SE (n = 4). The transpiration of control plants on day 1 was 97.16 ± 3.68 and 102.42 ± 5.60 g of water per g shoot DW for Ms and Mt. HM307, respectively. The stomatal conductance of control plants on day 1 was 0.34 ± 0.03 and 0.32 ± 0.02 nmol m−2 s−1 for Ms and Mt. HM307, respectively. An asterisk indicates significant differences between the control and water deficit treatments (Student’s t test, P ≤ 0.05). Different letters indicate significant differences between varieties and days 1–4 into the water deficit treatment according to Duncan’s test (P ≤ 0.05) (PDF 11 kb)
Supplementary Table 1
(DOCX 125 kb)
Rights and permissions
About this article
Cite this article
Echeverria, A., Gonzalez, E.M. Root system of Medicago sativa and Medicago truncatula: drought effects on carbon metabolism. Plant Soil 463, 249–263 (2021). https://doi.org/10.1007/s11104-021-04912-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-021-04912-1