Abstract
Purpose
This work is a quantitative evaluation of current hypotheses of iron acquisition by higher plants.
Methods
The outputs of three root uptake models were compared to the iron uptake of a single plant species grown in three different soils. Iron hydroxide dissolution kinetics by organic ligands were formalised in the reactive transport modelling, together with ligand and ferric complex degradation. A global sensitivity analysis was performed.
Results
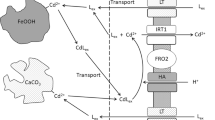
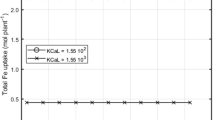
Iron uptake by Noccaea caerulescens does not appear to differ from that of other higher plants. The solubility of soil iron hydroxides alone cannot satisfy the plant iron requirements, even in an acidified rhizosphere. For this reason, all plants growing in aerated soils should suffer extreme iron deficiencies, not only those in alkaline soils. The dissociation rate of an Fe3+ complex (for instance with an aminopolycarboxilate ligand) in solution near the root cell membrane is not sufficient to provide the metal needed by the plant. In contrast, the adsorption of the ferric complex on the root cell membrane, before Fe3+ reduction and the absorption of Fe2+ appears to be a very efficient process. This is also the case for iron acquisition through the root secretion of an iron organic ligand dissolving soil iron hydroxides, the rate of ligand excretion being a key parameter.
Conclusions
Mechanistic modelling supports the hypothesis of the reduction at the root cell membrane of a ferric complex resulting of iron hydroxide dissolution by a soil- or root-borne organic ligand.







Similar content being viewed by others
References
Abadía J, Vázquez S, Rellán-Álvarez R, El-Jendoubi H, Abadía A, Álvarez-Fernández A, López-Millán AF (2011) Towards a knowledge-based correction of iron chlorosis. Plant Physiol Biochem 49:471–482. https://doi.org/10.1016/j.plaphy.2011.01.026
Anderson WB (1982) Diagnosis and correction of iron deficiency in field crops - an overview. J Plant Nutr 5:785–795
Astolfi S, Pii Y, Mimmo T, Lucini L, Miras-Moreno MB, Coppa E, Violino S, Celletti S, Cesco S (2020) Single and combined Fe and S deficiency differentially modulate root exudate composition in tomato: a double strategy for Fe acquisition? Int J Mol Sci 21:4038
Barber SA (1995) Soil nutrient bioavailability. A mechanistic approach, John Wiley & Sons, New York
Baxter I, Hermans C, Lahner B, Yakubova E, Tikhonova M, Verbruggen N, D-y C, Salt DE (2012) Biodiversity of mineral nutrient and trace element accumulation in Arabidopsis thaliana. PLoS One 7:e35121. https://doi.org/10.1371/journal.pone.0035121
Blossfeld S, Perriguey J, Sterckeman T, Morel JL, Lösch R (2010) Rhizosphere pH dynamics in trace-metal-contaminated soils, monitored with planar pH optodes. Plant Soil 330:173–184
Bucheli-Witschel M, Egli T (2001) Environmental fate and microbial degradation of aminopolycarboxylic acids. FEMS Microbiol Rev 25:69–106
Chaney RL, Brown JC, Tiffin LO (1972) Obligatory reduction of ferric chelates in iron uptake by soybeans. Plant Physiol 50:208–213. https://doi.org/10.1104/pp.50.2.208
Clemens S, Weber M (2016) The essential role of coumarin secretion for Fe acquisition from alkaline soil. Plant Signaling & Behavior 11:e1114197. https://doi.org/10.1080/15592324.2015.1114197
Cline GR, Powell PE, Szaniszlo PJ, Reid CPP (1982) Comparison of the abilities of hydroxamic, synthetic, and other natural organic acids to chelate iron and other ions in nutrient solution. Soil Sci Soc Am J 46:1158–1164. https://doi.org/10.2136/sssaj1982.03615995004600060008x
Cline GR, Powell PE, Szaniszlo PJ, Reid CPP (1983) Comparison of the abilities of hydroxamic and other natural organic acids to chelate iron and other ions in soil. Soil Sci 136:145–157
Custos J-M, Moyne C, Treillon T, Sterckeman T (2014) Contribution of cd-EDTA complexes to cadmium uptake by maize: a modelling approach. Plant Soil 374:497–512
Custos J-M, Moyne C, Sterckeman T (2020) How root nutrient uptake affects rhizosphere pH: a modelling study. Geoderma 369:114314. https://doi.org/10.1016/j.geoderma.2020.114314
Faivre R, Ioos B, Mahévas S, Makowski D, Monod H (eds) (2013) Analyse de sensibilité et exploration de modèles. Editions Quae, Versailles, p 324
Furrer G, Stumm W (1986) The coordination chemistry of weathering: I. dissolution kinetics of δ-Al2O3 and BeO. Geochim Cosmochim Acta 50:1847–1860. https://doi.org/10.1016/0016-7037(86)90243-7
George E, Horst WJ, Neumann E (2012) Adaptation of plants to adverse chemical conditions. In: Marschner P (ed) Marschner’s mineral nutrition of higher plants. Third edn. Elsevier, Amsterdam
Gonneau C, Genevois N, Frérot H, Sirguey C, Sterckeman T (2014) Variation of trace metal accumulation, major nutrient uptake and growth parameters and their correlations in 22 populations of Noccaea caerulescens. Plant Soil 384:271–287. https://doi.org/10.1007/s11104-014-2208-4
Guerinot ML, Yi Y (1994) Iron: nutritious, noxious, and not readily available. Plant Physiol 104:815–820. https://doi.org/10.1104/pp.104.3.815
Holmén BA, Tejedor-Tejedor MI, Casey WH (1997) Hydroxamate complexes in solution and at the goethite−water Interface: a cylindrical internal reflection Fourier transform infrared spectroscopy study. Langmuir 13:2197–2206. https://doi.org/10.1021/la960944v
Hudson RJM, Covault DT, Morel FMM (1992) Investigations of iron coordination and redox reactions in seawater using 59Fe radiometry and ion-pair solvent extraction of amphiphilic iron complexes. Mar Chem 38:209–235. https://doi.org/10.1016/0304-4203(92)90035-9
Kannan S (1971) Kinetics of iron absorption by excised rice roots. Planta 96:262–270. https://doi.org/10.1007/bf00387445
Kirkby E (2012) Introduction, definition and classification of nutrients. In: Marshner P (ed) Marschner’s mineral nutrition of higher plants. Third edn. Elsevier, London
Kraemer SM (2004) Iron oxide dissolution and solubility in the presence of siderophores. Aquat Sci 66:3–18
Lide DR (ed) (2010) CRC Handbook of Chemistry and Physics, 90th Edition (Internet Version 2010). CRC press/Taylor and Francis, Boca Raton, FL, p 2758
Lin Z, Schneider A, Nguyen C, Sterckeman T (2014) Can ligand addition to soil enhance cd phytoextraction? A mechanistic model study. Environ Sci Pollut Res 21:12811–12826. https://doi.org/10.1007/s11356-014-3218-8
Lin Z, Schneider A, Sterckeman T, Nguyen C (2016) Ranking of mechanisms governing the phytoavailability of cadmium in agricultural soils using a mechanistic model. Plant Soil 399:89–107. https://doi.org/10.1007/s11104-015-2663-6
Lindsay WL, Schwab AP (1982) The chemistry of iron in soils and its availability to plants. J Plant Nutr 5:821–840. https://doi.org/10.1080/01904168209363012
Lucena JJ (2003) Fe chelates for remediation of Fe chlorosis in strategy I plants. J Plant Nutr 26:1969–1984. https://doi.org/10.1081/pln-120024257
Marschner H, Römheld V (1994) Strategies of plants for acquisition of iron. Plant Soil 165:261–274
Mengel K (1994) Iron availability in plant tissues-iron chlorosis on calcareous soils. Plant Soil 165:275–283. https://doi.org/10.1007/bf00008070
Mimmo T, Del Buono D, Terzano R, Tomasi N, Vigani G, Crecchio C, Pinton R, Zocchi G, Cesco S (2014) Rhizospheric organic compounds in the soil–microorganism–plant system: their role in iron availability. Eur J Soil Sci 65:629–642. https://doi.org/10.1111/ejss.12158
Mullins GL, Sommers LE, Barber SA (1986) Modelling the plant uptake of cadmium and zinc from soils treated with sewage sludge. Soil Sci Soc Am J 50:1245–1250
Orera I, Rodríguez-Castrillón JA, Moldovan M, García-Alonso JI, Abadía A, Abadía J, Álvarez-Fernández A (2010) Using a dual-stable isotope tracer method to study the uptake, xylem transport and distribution of Fe and its chelating agent from stereoisomers of an Fe(iii)-chelate used as fertilizer in Fe-deficient strategy I plants. Metallomics 2:646–657. https://doi.org/10.1039/c0mt00018c
Pagès L, Xie J, Serra V (2013) Potential and actual root growth variations in root systems: modeling them with a two-step stochastic approach. Plant Soil 373:723–735. https://doi.org/10.1007/s11104-013-1820-z
Robinson NJ, Procter CM, Connolly EL, Guerinot ML (1999) A ferric-chelate reductase for iron uptake from soils. Nature 397:694–697. https://doi.org/10.1038/17800
Römheld V (1987) Different strategies for iron acquisition in higher plants. Physiol Plant 70:231–234. https://doi.org/10.1111/j.1399-3054.1987.tb06137.x
Römheld V, Marschner H (1983) Mechanism of iron uptake by peanut plants. I. Fe(III) reduction, chelate splitting, and release of phenolics. Plant Physiol 71:949–954. https://doi.org/10.1104/pp.71.4.949
Römheld V, Marschner H (1986) Evidence for a specific uptake system for iron phytosiderophores in roots of grasses. Plant Physiol 80:175–180. https://doi.org/10.1104/pp.80.1.175
Saltelli A, Tarantola S, Campolongo F, Ratto M (2004) Sensitivity analysis in practice. A guide to assessing scientific models. John Wiley & Sons, Chichester
Satroutdinov AD, Dedyukhina EG, Chistyakova TI, Witschel M, Minkevich IG, Eroshin VK, Egli T (2000) Degradation of metal-EDTA complexes by resting cells of the bacterial strain DSM 9103. Environ Sci Technol 34:1715–1720
Schenkeveld WDC, Reichwein AM, Temminghoff EJM, van Riemsdijk WH (2014) Considerations on the shuttle mechanism of FeEDDHA chelates at the soil-root interface in case of Fe deficiency. Plant Soil 379:373–387. https://doi.org/10.1007/s11104-014-2057-1
Schwertmann U (1991) Solubility and dissolution of iron oxides. Plant Soil 130:1–25
Sposito G (1989) The chemistry of soils. Oxford University Press, Oxford
Sterckeman T, Perriguey J, Caël M, Schwartz C, Morel JL (2004) Applying a mechanistic model to cadmium uptake by Zea mays and Thlaspi caerulescens: consequences for the assessment of the soil quantity and capacity factors. Plant Soil 262:289–302
Sterckeman T, Cazes Y, Gonneau C, Sirguey C (2017) Phenotyping 60 populations of Noccaea caerulescens provides a broader knowledge of variation in traits of interest for phytoextraction. Plant Soil 418:523–540
Sterckeman T, Cazes Y, Sirguey C (2019) Breeding the hyperaccumulator Noccaea caerulescens for trace metal phytoextraction: first results of a pure-line selection. Int J Phytorem 21:448–455. https://doi.org/10.1080/15226514.2018.1537250
Swoboda-Colberg NG, Drever JI (1993) Mineral dissolution rates in plot-scale field and laboratory experiments. Chem Geol 105:51–69
Thomine S, Vert G (2013) Iron transport in plants: better be safe than sorry. Curr Opin Plant Biol 16:322–327. https://doi.org/10.1016/j.pbi.2013.01.003
Tiedje JM (1975) Microbial degradation of ethylenediaminetetraacetate in soils and sediments. Appl Environ Microbiol 30:327–329
Tiffin LO, Brown JC (1961) Selective absorption of iron from iron chelates by soybean plants. Plant Physiol 36:710–714. https://doi.org/10.1104/pp.36.5.710
Tinker PB, Nye PH (2000) Solute movement in the rhizosphere. Oxford University Press, Oxford
Torquato S (2002) Random heterogenous materials: microstructure and macroscopic properties. Springer, New-York
Tsai HH, Schmidt W (2017a) Mobilization of Iron by plant-borne coumarins. Trends in Plant Science 22: 538-548. https://doi.org/10.1016/j.tplants.2017.03.008
Tsai HH, Schmidt W (2017b) One way. Or another? Iron uptake in plants. New Phytol 214:500–505. https://doi.org/10.1111/nph.14477
Vose PB (1982) Iron nutrition in plants: a world overview. J Plant Nutr 5:233–249. https://doi.org/10.1080/01904168209362954
Wen J, Stacey SP, McLaughlin MJ, Kirby JK (2009) Biodegradation of rhamnolipid, EDTA and citric acid in cadmium and zinc contaminated soils. Soil Biol Biochem 41:2214–2221. https://doi.org/10.1016/j.soilbio.2009.08.006
Weng L, Temminghoff EJM, Van Riemsdijk WH (2001) Contribution of individual sorbents to the control of heavy metal activity in sandy soil. Environ Sci Technol 35:4436–4443. https://doi.org/10.1021/es010085j
White PJ (2012) Ion uptake mechanisms of individual cells and roots: short-distance transport. In: Marschner P (ed) Marschner’s mineral nutrition of higher plants. Third edn. Elsevier, Amsterdam
Whiting SN, Broadley MR, White PJ (2003) Applying a solute transfer model to phytoextraction: zinc acquisition by Thlaspi caerulescens. Plant Soil 249:45–56
Xue H, Sigg L, Kari FG (1995) Speciation of EDTA in natural waters: exchange kinetics of Fe-EDTA in river water. Environ Sci Technol 29:59–68. https://doi.org/10.1021/es00001a007
Zinder B, Furrer G, Stumm W (1986) The coordination chemistry of weathering: II. Dissolution of Fe(III) oxides. Geochim Cosmochim Acta 50:1861–1869. https://doi.org/10.1016/0016-7037(86)90244-9
Zuo Y, Zhang F (2011) Soil and crop management strategies to prevent iron deficiency in crops. Plant Soil 339:83–95. https://doi.org/10.1007/s11104-010-0566-0
Acknowledgements
The authors are grateful to Dr. Sébastien Thomine for his careful reading of the manuscript.
Availability of data and material
Experimental data are given in Supplementary Material.
Code availability
MATLAB® scripts are available on demand.
Funding
The experimental part of this work was supported by the French National Research Agency (SimTraces project, ANR 2011 CESA 008 01).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible Editor: Miroslav Nikolic
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
Values of the model parameters; Range of variation of the parameters used in the global sensitivity analysis; Main results of the experiment based on the cultivation of Noccaea caerulescens; Detailed results of the simulations of Fe uptake using the various models; Mini-reviews about the rates of organic ligand excretion by roots, the sorption and the degradation of the ligand and of the Fe complex. (PDF 483 kb)
Rights and permissions
About this article
Cite this article
Sterckeman, T., Moyne, C. & Le, T.D. A modelling study to evaluate the mechanisms of root iron uptake by Noccaea caerulescens. Plant Soil 463, 125–144 (2021). https://doi.org/10.1007/s11104-021-04873-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-021-04873-5