Abstract
Aims
Oilseed rape (Brassica napus L.) has high nitrogen (N) requirement for its optimal growth and yield. However, little is known about the transcriptomic reprogramming of rapeseed in response to N deficiency.
Methods
Two rapeseed genotypes with contrasting N use efficiency (NUE) were used to perform whole-transcriptome sequencing on the shoots and roots supplied with or without N in a time-course experiment.
Results
A total of 24,435 differentially expressed genes (DEGs) were identified. Among them, 8811 genes were differentially expressed between two N treatments and between the two genotypes. Coexpression networks reveal central N-deficiency-response modules with diverse expression patterns between the two genotypes. Major expression hubs involved in phytohormone signaling were identified and showed strong association with DEGs in each module. Genes involved in N uptake and assimilation, carbohydrate catabolism, amino acid metabolism, and fatty acid biosynthesis were enriched between the two genotypes under N deprivation. In addition, photosynthesis and carbon assimilation declined under prolonged N stress, but starch biosynthesis was enhanced by N deprivation.
Conclusions
Our results revealed N-responsive genes and gene networks and expanded the knowledge of regulatory hubs in N-deprived rapeseed. The detailed gene information might assist the genetic improvement of NUE in B. napus.









Similar content being viewed by others
References
Bouchet AS, Laperche A, Bissuel-Belaygue C, Snowdon RJ, Nesi N, Stahl A (2016) Nitrogen use effciency in rapeseed. A review. Agron Sustain Dev 36:38–58
Chalhoub B, Denoeud F, Liu S, Parkin IA, Tang H, Wang X et al (2014) Early allopolyploid evolution in the post-neolithic Brassica napus oilseed genome. Science 345:950–953
Choe S, Tanaka A, Noguchi T, Fujioka S, Takatsuto S, Ross AS, Tax FE, Yoshida S, Feldmann KA (2000) Lesions in the sterol delta reductase gene of Arabidopsis cause dwarfism due to a block in brassinosteroid biosynthesis. Plant J 21:431–443
Colebrook EH, Thomas SG, Phillips AL, Hedden P (2014) The role of gibberellin signalling in plant responses to abiotic stress. J Exp Biol 217:67–75
Conley DJ, Paerl HW, Howarth RW, Boesch DF, Seitzinger SP, Havens KE, Lancelot C, Likens GE (2009) Controlling eutrophication: nitrogen and phosphorus. Science 323:1014–1015
Curci PL, Aiese Cigliano R, Zuluaga DL, Janni M, Sanseverino W, Sonnante G (2017) Transcriptomic response of durum wheat to nitrogen starvation. Sci Rep 7:1176–1190
De Groot CC, Van Den Boogaard R, Marcelis LF, Harbinson J, Lambers H (2003) Contrasting effects of N and P deprivation on the regulation of photosynthesis in tomato plants in relation to feedback limitation. J Exp Bot 54:1957–1967
Falcon S, Gentleman R (2007) Using GOstats to test gene lists for GO term association. Bioinformatics 23:257–258
Garcia-Luis A, Oliveira MEM, Bordón Y, Siqueira DL, Tominaga S, Guardiola JL (2002) Dry matter accumulation in citrus fruit is not limited by transport capacity of the pedicel. Ann Bot 90:755–764
Gaude N, Bréhélin C, Tischendorf G, Kessler F, Dörmann P (2007) Nitrogen deficiency in Arabidopsis affects galactolipid composition and gene expression and results in accumulation of fatty acid phytyl esters. Plant J 49:729–739
Han M, Okamoto M, Beatty PH, Rothstein SJ, Good AG (2015) The genetics of nitrogen use efficiency in crop plants. Annu Rev Genet 49:269–289
Hawkesford M, Horst W, Kichey T, Lamber H, Schjoerring J, Møller IS, White P (2012) Chapter 6- functions of macronutrients. In: Marschner’s mineral nutrition of higher plants, 3rd edn. Academic Press, London, pp 158–165
Hu B, Wang W, Ou S, Tang J, Li H, Che R, Zhang Z, Chai X, Wang H, Wang Y, Liang C, Liu L, Piao Z, Deng Q, Deng K, Xu C, Liang Y, Zhang L, Li L, Chu C (2015) Variation in NRT1.1B contributes to nitrate-use divergence between rice subspecies. Nat Genet 47:834–838
Jin JP, Tian F, Yang DC, Meng YQ, Kong L, Luo J, Gao G (2017) Plant TFDB 4.0: toward a central hub for transcription factors and regulatory interactions in plants. Nucleic Acids Res 45:1040–1045
Kiba T, Krapp A (2016) Plant nitrogen acquisition under low availability: regulation of uptake and root architecture. Plant Cell Physiol 57:707–714
Kiba T, Kudo T, Kojima M, Sakakibara H (2011) Hormonal control of nitrogen acquisition: roles of auxin, abscisic acid, and cytokinin. J Exp Bot 62:1399–1409
Kim D, Langmead B, Salzberg SL (2015) HISAT: a fast-spliced aligner with low memory requirements. Nat Methods 12:357–360
Kinsella RJ, Kähäri A, Haider S, Zamora J, Proctor G, Spudich G, Almeida-King J, Staines D, Derwent P, Kerhornou A, Kersey P, Flicek P (2011) Ensembl BioMarts: a hub for data retrieval across taxonomic space. Database (Oxford) bar030
Krapp A, Berthomé R, Orsel M, Mercey-Boutet S, Yu A, Castaings L, Elftieh S, Major H, Renou JP, Daniel-Vedele F (2011) Arabidopsis roots and shoots show distinct temporal adaptation patterns toward nitrogen starvation. Plant Physiol 157:1255–1282
Krouk G (2016) Hormones and nitrate: a two-way connection. Plant Mol Biol 91:599–606
Langfelder P, Horvath S (2008) WGCNA: an R package for weighted correlation network analysis. BMC Bioinformatics 9:559–572
Li Q, Ding GD, Yang YM, White PJ, Ye XS, Cai HM, Lu JW, Shi L, Xu FS (2020) Comparative genome and transcriptome analysis unravel key factors of nitrogen use efficiency in Brassica napus L. Plant Cell Environ 43:712–731
Liscum E, Reed J (2002) Genetics of Aux/IAA and ARF action in plant growth and development. Plant Mol Biol 49:387–400
Liu Q, Ren T, Zhang Y, Li X, Cong R, White PJ, Lu J (2019) Yield loss of oilseed rape (Brassica napus L.) under nitrogen deficiency is associated with under-regulation of plant population density. Eur J Agron 103:80–89
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCT method. Methods 25:402–408
Love MI, Huber W, Anders S (2014) Moderated estimation of fold change and dispersion for RNA Seq data with DESeq2. Genome Biol 15:550–571
Martin M (2011) Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnetjournal, North America 2011:17
Moriya Y, Itoh M, Okuda S, Yoshizawa AC, Kanehisa M (2007) KAAS: an automatic genome annotation and pathway reconstruction server. Nucleic Acids Res 35:182–185
Muchow RC (1998) Nitrogen utilization efficiency in maize and grain sorghum. Field Crop Res 56:209–216
Nunes-Nesi A, Fernie AR, Stitt M (2010) Metabolic and signaling aspects underpinning the regulation of plant C nitrogen interactions. Mol Plant 3:973–996
Pertea M, Pertea GM, Antonescu CM, Chang TC, Mendell JT, Salzberg SL (2015) StringTie enables improved reconstruction of a transcriptome from RNA_Seq reads. Nat Biotechnol 33:290–295
Pertea M, Kim D, Pertea GM, Leek JT, Salzberg SL (2016) Transcript-level expression analysis of RNA_seq experiments with HISAT, StringTie and Ballgown. Nat Protoc 11:1650–1667
Qin L, Walk TC, Han P, Chen L, Zhang S, Li Y, Hu X, Xie L, Yang Y, Liu J, Lu X, Yu C, Tian J, Shaff JE, Kochian LV, Liao X, Liao H (2019) Adaption of root to nitrogen deficiency revealed by 3D quantification and proteomic analysis. Plant Physiol 179:329–347
Quan X, Zeng J, Chen G, Zhang G (2019) Transcriptomic analysis reveals adaptive strategies to chronic low nitrogen in Tibetan wild barley. BMC Plant Biol 19:68–82
Ranocha P, Dima O, Nagy R, Felten J, Corratgé-Faillie C, Novák O, Morreel K, Lacombe B, Martinez Y, Pfrunder S, Jin X, Renou JP, Thibaud JB, Ljung K, Fischer U, Martinoia E, Boerjan W, Goffner D (2013) Arabidopsis WAT1 is a vacuolar auxin transport facilitator required for auxin homoeostasis. Nat Commun 4:2625–2634
Robertson GP, Vitousek PM (2009) Nitrogen in agriculture: balancing the cost of an essential resource. Annu Rev Environ Resour 34:97–125
Ruan YL (2014) Sucrose metabolism: gateway to diverse C use and sugar signaling. Annu Rev Plant Biol 65:33–67
Santopolo S, Boccaccini A, Lorrai R, Ruta V, Capauto D, Minutello E, Serino G, Costantino P, Vittorioso P (2015) DOF AFFECTING GERMINATION 2 is a positive regulator of light-mediated seed germination and is repressed by DOF AFFECTING GERMINATION 1. BMC Plant Biol 15:453–464
Sebilo M, Mayer B, Nicolardot B, Pinay G, Mariotti A (2013) Long-term fate of nitrate fertilizer in agricultural soils. Proc Natl Acad Sci U S A 110:18185–18189
Shannon P, Markiel A, Ozier O, Baliga NS, Wang JT, Ramage D, Amin N, Schwikowski B, Ideker T (2003) Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res 13:2498–2504
Shen T, Xiong Q, Zhong L, Shi X, Cao C, He H, Chen X (2019) Analysis of main metabolisms during nitrogen deficiency and compensation in rice. Acta Physiol Plant 41:68–82
Sinha SK, Sevanthi VAM, Chaudhary S, Tyagi P, Venkadesan S, Rani M, Mandal PK (2018) Transcriptome analysis of two rice varieties contrasting for nitrogen use efficiency under chronic N starvation reveals differences in chloroplast and starch metabolism-related genes. Genes 9:206–228
Smith AM (2012) Starch in the Arabidopsis plant. Starch-Starke 64:421–434
Stahl A, Friedt W, Wittkop B, Snowdon RJ (2016) Complementary diversity for nitrogen uptake and utilisation efficiency reveals broad potential for increased sustainability of oilseed rape production. Plant Soil 400:245–262
Stahl A, Vollrath P, Samans B, Frisch M, Wittkop B, Snowdon RJ (2019) Effect of breeding on nitrogen use efficiency-associated traits in oilseed rape. J Exp Bot 70:1969–1986
Terasaka K, Blakeslee JJ, Titapiwatanakun B, Peer WA, Bandyopadhyay A, Makam SN, Lee OR, Richards EL, Murphy AS, Sato F, Yazaki K (2005) PGP4, an ATP binding cassette P-glycoprotein, catalyzes auxin transport in Arabidopsis thaliana root. Plant Cell 17:2922–2939
Thimm O, Bläsing O, Gibon Y, Nagel A, Meyer S, Krüger P, Selbig J, Müller LA, Rhee SY, Stitt M (2004) MAPMAN: a user-driven tool to display genomics data sets onto diagrams of metabolic pathways and other biological processes. Plant J 37:914–939
Thompson JE, Froese CD, Madey E, Smith MD, Hong Y (1998) Lipid metabolism during plant senescence. Prog Lipid Res 37:119–141
Uno Y, Furihata T, Abe H, Yoshida R, Shinozaki K, Yamaguchi-Shinozaki K (2000) Arabidopsis basic leucine zipper transcription factors involved in an abscisic acid-dependent signal transduction pathway under drought and high-salinity conditions. Proc Natl Acad Sci U S A 97:11632–11637
Vidal EA, Alvarez JM, Araus V, Riveras E, Brooks M, Krouk G, Ruffel S, Lejay L, Crawford N, Coruzzi GM, Gutiérrez RA (2020) Nitrate 2020: thirty years from transport to signaling networks. Plant Cell 32:2094–2119
Wang G, Ding G, Li L, Cai H, Ye X, Zou J, Xu F (2014) Identification and characterization of improved nitrogen efficiency in interspecific hybridized new-type Brassica napus. Ann Bot (London) 114:549–559
Wang GL, Ding GD, Xu FS, Cai HM, Zou J, Ye XS (2015) Genotype differences in photosynthetic characteristics and nitrogen efficiency of new-type oilseed rape responding to low nitrogen stress. J Agric Sci 153:1030–1043
Xu G, Fan X, Miller AJ (2012) Plant nitrogen assimilation and use efficiency. Annu Rev Plant Biol 63:153–182
Xuan W, Beeckman T, Xu G (2017) Plant nitrogen nutrition: sensing and signaling. Curr Opin Plant Biol 39:57–65
Yuan L, Loqué D, Kojima S, Rauch S, Ishiyama K, Inoue E, Takahashi H, Von WN (2007) The organization of high-affinity ammonium uptake in Arabidopsis root depends on the spatial arrangement and biochemical properties of AMT1-type transporters. Plant Cell 19:2636–2652
Zhang ZH, Zhou T, Liao Q, Yao JY, Liang GH, Song HX, Guan CY, Hua YP (2018) Integrated physiologic, genomic and transcriptomic strategies involving the adaptation of allotetraploid rapeseed to nitrogen limitation. BMC Plant Biol 18:322–340
Zuluaga DL, Sonnante G (2019) The use of nitrogen and its regulation in cereals: structural genes, transcription factors, and the role of miRNAs. Plants 8:294–309
Acknowledgments
This work was supported by the Grants from the National Key Research and Development Program of China (2018YFD0200900) and the Fundamental Research Funds for the Central Universities of China (2662019PY013). We thank Professor Jianwei Lu (Huazhong Agricultural University, China) for his critical advice in experimental design.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Ad C. Borstlap.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
Number of differentially expressed genes (DEGs) between D4–15 and D2–1 across the four time points after nitrogen (N) starvation. A, Number of DEGs in root after N starvation. B, Number of DEGs in shoot after N starvation. DEGs specifically modulated in D2–1 and D4–15 are shown in red and blue color, respectively. Grey color indicates the shared DEGs in both genotypes. X-axis indicates the time after N starvation. DEGs were identified using DESeq2 with the threshold: adjusted P value <0.05 and |log2 (fold change)| > 1. (DOCX 2303 kb)
ESM 2
Comparison between the log2 fold change of relative expression obtained from RNA-Seq data (X-axis) and qRT-PCR (Y-axis) under N-starvation (0 mM N) condition compared to control (6 mM N) condition. (DOCX 136 kb)
ESM 3
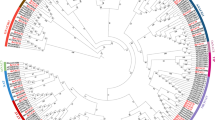
Hierarchical cluster trees and co-expression modules identified by WGCNA in roots (A) and shoots (B) of the two Brassica napus genotypes based on the DEGs. Each branch in the dendrogram constitutes a module and each leaf in the branch represents one gene. Different modules were labeled with different colors in order to be distinguished from each other and the grey part contains genes not belonging to any modules. (DOCX 384 kb)
ESM 4
(DOCX 16 kb)
ESM 5
(DOCX 34 kb)
ESM 6
(XLSX 299 kb)
ESM 7
(DOCX 49 kb)
ESM 8
(DOCX 38 kb)
ESM 9
(DOCX 46 kb)
ESM 10
(DOCX 29 kb)
ESM 11
(DOCX 73 kb)
Rights and permissions
About this article
Cite this article
Yang, N., Li, S., Wang, S. et al. Dynamic transcriptome analysis indicates extensive and discrepant transcriptomic reprogramming of two rapeseed genotypes with contrasting NUE in response to nitrogen deficiency. Plant Soil 456, 369–390 (2020). https://doi.org/10.1007/s11104-020-04720-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-020-04720-z