Abstract
Background and aims
Intensification of savannah land-use is predicted to negatively influence soil biodiversity and functioning such as litter decomposition by detritivores. Loss of macrodetritivores, particularly termites, may be problematic in drier savannahs due to the capacity of macrodetritivores to sustain litter decomposition. Here we investigate how human land-use and spatiotemporal rainfall influence the contribution of macrodetritivores to plant litter decomposition.
Methods
We measured decomposition using globally standardized litter: labile green and recalcitrant rooibos tea litter. The contribution of macrodetritivores to litter decomposition was determined through exclusion using meshed litterbags. Litter decomposition was determined in agricultural land, pastureland and wildlife protected areas during both wet and dry seasons and in mesic and wet rainfall regions across the borders of the Serengeti National Park, Tanzania.
Results
Macrodetritivores consumed recalcitrant rooibos and mainly avoided labile green tea litter. On average macrodetritivores enhanced recalcitrant litter decomposition by 22%, but litter mass loss varied across land-uses, typically being higher on agricultural and pastureland compared to wildlife protected areas, and was sustained during periods of water scarcity. However, we observed instances of higher decomposition of recalcitrant litter by macrodetritivores in wildlife protected areas. In contrast, litter decomposition by microbes and microdetritivores was more constrained by seasonal and regional water availability with a minor influence of land-use.
Conclusion
We found that moderate human-modification of savannahs is compatible with macrodetritivore litter decomposition. As savannahs become more intensely used by humans, raising ecological awareness among agropastoralist is required to ensure continued contribution of macrodetritivores to litter decomposition.
Similar content being viewed by others
Introduction
An increasing proportion of tropical ecosystems is modified by humans with a growing land area converted from tropical forests and wooded savannahs to agricultural land and pastureland (FAO 2015; Osborne et al. 2018). More intense use of tropical ecosystems is generally predicted to have a negative influence on soil biodiversity, abundance of soil organisms and their functional contribution to plant litter decomposition and nutrient cycling (Ayuke et al. 2011; Beare et al. 1997; Black and Okwakol 1997; Giller et al. 1997; Osborne et al. 2018). However, this is not always the case in tropical ecosystems. Detritivores continue to remove plant litter in human managed landscapes, for example under regulated burning or livestock grazing (Davies et al. 2013; Smith et al. 2019). Quantifying the contribution of detritivores to plant litter decomposition remains a key knowledge gap in tropical ecosystems increasingly modified by humans.
Macrodetritivores have a body length between 2 and 20 mm distinguishing them from the smaller microdetritivores and the larger megadetritivores (Abe et al. 2000; Gessner et al. 2010). Macrodetritivores encompass a diversity of litter- and soil-dwelling invertebrates that consume plant litter, dung and soil organic matter, e.g. termites, millipedes and woodlice. The diversity of detritivores generally declines following human land-use disturbances, such as clearing tree cover (Dangerfield 1990; Kamau et al. 2017), mechanical soil tillage (Black and Okwakol 1997; Eggleton et al. 2002; Okwakol 2000) and application of chemical insecticides and pesticides (Beare et al. 1997; Jouquet et al. 2011). However, the abundance of various macrodetritivore species has been found to increase following forest clearance, cultivation and livestock intensification compared to unmodified tropical ecosystems (Ayuke et al. 2011; Black and Okwakol 1997; Decaëns et al. 2004; Eggleton et al. 2002; Hagan et al. 2017; Kamau et al. 2017; Okwakol 2000; Wood et al. 1980). One of the most important macrodetritivore taxa in tropical ecosystems are termites that forage above and below the soil surface (Abe et al. 2000; Jouquet et al. 2011; Sileshi et al. 2010). Termite species that feed on wood, grass and plant litter occur at higher abundances in tropical ecosystems moderately modified humans compared to undisturbed or protected tropical ecosystems (Ayuke et al. 2011; Black and Okwakol 1997; Eggleton et al. 2002; Okwakol 2000; Wood et al. 1980). Building on previous studies, we expect moderate human modification, such as pasturelands with limited vegetation structural change, to exhibit elevated plant litter decomposition by macrodetritivores compared to unmodified ecosystems. On the other hand, cleared and intensely disturbed land-uses, such as agricultural land, would exhibit reduced contribution of macrodetritivores to litter decomposition.
Tropical savannah ecosystems are defined by distinct wet and dry seasons based on the quantity, frequency and duration of rainfall. During the wet season, plant litter decomposes more rapidly than during the dry season (Davies et al. 2013; Freymann et al. 2010; Ngatia et al. 2014). Rainfall can vary spatially and regional differences in litter decomposition may be more nuanced following pulses of rainfall in a given season (Anaya et al. 2012; Joly et al. 2019). Yet, significant litter decomposition can also occur during the dry seasons in tropical savannahs with infrequent or limited rainfall (Freymann et al. 2010; Ngatia et al. 2014; Veldhuis et al. 2017). Plant litter can be removed by fire, consumed by detritivores and degraded by ultraviolet radiation (Austin 2011; Cornwell et al. 2009). Indeed, a growing body of work shows that macrodetritivores, particularly termites, can sustain plant litter decomposition during periods with lower water availability in contrast to free-living soil microbes (Ashton et al. 2019; Jouquet et al. 2011; Veldhuis et al. 2017). The majority of previous research has been undertaken in wildlife protected tropical ecosystems with low human disturbance. Cultivated savannahs would be expected to have a reduced tree and shrub canopy cover and a reduced structural heterogeneity, leading to higher rates of soil water evaporation and substantially drier conditions. This, in turn, could further constrain decomposition by microbes and negatively influence macrodetritivores that would seek moist refuges and minimise foraging due to higher risk of desiccation (Cornelius and Osbrink 2010; Joly et al. 2019; Woon et al. 2019). Additionally, dry season foraging is not a universal trait to all litter feeding macrodetritivore species, and several termite and isopod species are seemingly dormant during the dry season months resulting in little or no litter foraging (Davies et al. 2015; Davies et al. 2013; Leitner et al. 2018). As such, there remains ambiguity over the contribution of macrodetritivores to litter decomposition across temporally and spatially variable rainfall in tropical savannah ecosystems.
We hypothesize that (i) moderate human modification of tropical savannahs leads to an increase in litter decomposition by macrodetritivores and that (ii) on these land-uses the contribution of macrodetritivores to litter decomposition is greater in drier regions and during the dry season. In contrast, intense human land-use reduces the contribution of macrodetritivores to litter decomposition across spatially and temporally variable rainfall. To test these hypotheses, we conducted a litterbag experiment replicated across wildlife protected areas, pasturelands and agricultural lands spanning low to high human modification in mesic and wet rainfall regions across the borders of Serengeti National Park, Tanzania. We used a modified version of the Tea Bag Index (Keuskamp et al. 2013), decomposing standardized labile green and recalcitrant rooibos tea litter in the wet and dry season. Meshed litterbags were used to determine the contribution of macrodetritivores >0.3 mm body width, targeting the exclusion of termites (Smith et al. 2019; Teo et al. 2020). Other standardized litter types, e.g. straw and wooden blocks, are routinely employed to investigate macrodetritivore decomposition in tropical ecosystems (Griffiths et al. 2019; Veldhuis et al. 2017). While the standardized Tea Bag Index has been globally adopted to investigate microbial litter decomposition, its application to investigate decomposition by macrodetritivores remains understudied, particularly in tropical ecosystems (Teo et al. 2020).
Material and methods
Study area and site selection
We conducted a plant litter decomposition study inside and outside the Serengeti National Park, Tanzania (latitude 2°40′ to 2°50′S and longitude 34°00′ to 34°90′E) during peak wet and dry seasons in 2017. We focused our study in two rainfall regions, a mesic region in the south-east and a wet region in the south-west of the Serengeti ecosystem (Table 1). Annual rainfall in these regions varies from 700 mm in the mesic east to 1300 mm in the wetter west (2015–2017 (Huffman 2017)). Rainfall varies seasonally with the majority of rainfall, up to 800 mm, occurring during the wet season between November and May. The dry season between June and October receives around 150 mm of rainfall, although the dry season is drier and longer in the mesic region compared to the wet region (Norton-Griffiths et al. 1975). Soil temperatures across the system are more uniform with small fluctuations around 29 °C in the upper 10 cm of the soil profile across the seasons (McNally et al. 2017). The elevation of our mesic and wet regions ranges between 1200 and 1670 m. Underlying soil types are primarily calcareous and stony leptosols with pockets of clay-rich and organic vertisols in the mesic region and organic planosols, sandy aeronsols and vertisols in the wet region (ISRIC 2018) (Table 1). Two-thirds of the Serengeti National Park is open wooded savannah, dominated by leguminous trees with nitrogen fixing symbionts such as Vachellia spp. and non-leguminous trees such as Commiphora spp. that are interspersed amongst a C4 grass dominated herbaceous layer.
Knowledge of the distribution and abundance of macrodetritivore species remains limited for the Serengeti ecosystem. There have been a handful of studies on macrodetritivores in the Serengeti mainly investigating litter and detritus decomposition by termite species (i.e. Macrotermes sp. and Odontotermes sp.) or on the diets of insectivorous mammals, for example aardwolf consumption of Trinervitermes spp. termites (de Visser et al. 2015; Freymann et al. 2008; Freymann et al. 2007; Freymann et al. 2010; Kruuk and Sands 1972; Smith et al. 2019). Inside wildlife protected areas, the majority of wild herbivores (including elephants, buffalos, impalas and various species of antelope) occur at low densities, with the exception of migratory wildebeest and zebra (Hopcraft et al. 2015; Sinclair et al. 2007). Within wildlife protected areas, landscape-scale spatial patterns of litter removal by macrodetritivores overlap with wild herbivore movements (de Visser et al. 2015; Freymann et al. 2010). Wildlife protected areas are regularly managed through burning, but fire management is presumed to have limited impact on rates of litter removal by macrodetritivores (Davies et al. 2013). The dominant land-use outside wildlife protected areas is agropastoral, comprising mosaics of small-scale agricultural holdings intermixed with livestock pastures (Veldhuis et al. 2019) (Table 1). Annual aboveground grass biomass production can be similar on pasturelands (470 g m−2 yr−1) and wildlife protected areas (515 g m−2 yr−1); however, livestock consume on average 70% of aboveground biomass in pasturelands compared to 40% by wildlife in protected areas (Arneberg et al. unpublished data). Intense livestock rearing removes stubble from agricultural land and reduces fuel loads on pasturelands and results in infrequent or no fires on agropastoral land (Veldhuis et al. 2019).
Within the mesic and wet rainfall regions, sites were selected across three human land-uses: agricultural land, pastureland and wildlife protected areas. Within each rainfall region all land-uses were within 10 km of each other to minimize spatiotemporal variation in rainfall between land-uses. Termites were found in the soil at all sites suggesting presence of termite-driven litter decomposition processes. All sites had Macrotermes and/or Odontotermes termite mounds, although we did not quantify the termite activity within mounds (Table 1). Agricultural sites were primarily used for growing maize (Zea mays) with minimal intercropping, but occasionally with beans and vegetables. Agricultural land was managed by hand hoeing with limited use of tractors and without pesticides or manmade fertilizers. The influence of illegal livestock grazing on sites inside wildlife protected areas was minimised by selecting sites a minimum of 9 km into wildlife protected areas in the mesic region, and in close proximity to a ranger post in the wet region.
We selected four replicate sites within each land-use (agricultural land, pastureland and wildlife protected area), each comprising an area of approximately 50 m2. All sites were a minimum of 500 m apart from one another, except for the agricultural sites. For these, we selected two agricultural fields in each rainfall region which were managed by the landowner of the adjacent pastural site. Each agricultural field was divided in two, thus creating paired agricultural sites on each agricultural field approximately 100 m apart. In total we had 24 sites (2 rainfall regions × 3 land-uses × 4 replicate sites).
Experimental design
To investigate the influence of season, rainfall region and land-use on plant litter decomposition we buried litterbags during both the wet season (from late January to early March 2017) and the dry season (from July to September 2017). Within each site, plots for litterbag burial were selected following a random cardinal direction and number of paces from the centre of the site. All plots were a minimum of 1 m apart and 2 m away from the nearest termite mound or tree canopy edge, with 100 m as the furthest distance from mound/tree. For the wet season there were eight plots per site and for the dry season seven plots per site.
We used a modified version of the globally standardized Tea Bag Index (Keuskamp et al. 2013). The Tea Bag Index uses two types of tea litter with distinct qualities: (1) green tea (Camellia sinesis; EAN no.: 8722700055525 Lipton® Tea) with high cellulose content (46.8% carbon (C), 4.1% nitrogen (N) and 11.5 C:N ratio, particle size ~6 mm2) and expected fast decomposition, hereafter referred to as ‘labile litter’; and (2) rooibos tea (Aspalanthus linearis; EAN no.: 722700188438 Lipton® Tea) with high lignin content (48% C, 1.2% N and 39.2 C:N ratio, particle size ~3 mm2) and expected slow decomposition, hereafter referred to as ‘recalcitrant litter’ (Keuskamp et al. 2013). The Tea Bag Index has based the lability and recalcitrance of tea litter types on rates of decomposition by soil microbes, while litter preferences of detritivores may differ from microbes. Several macrodetritivores, e.g. millipedes, termites and woodlice, prefer recalcitrant litter types (Hättenschwiler and Gasser 2005; Peguero et al. 2019; Sitters et al. 2014). For example, termite species ferment litter within the gut through a mutualistic relationship with microorganisms (i.e. bacteria, archaea and fungi) (Abe et al. 2000). Due to this fermentation process, termites avoid substrates high in readily digestible sugars (Abe et al. 2000), yet other macrodetritivore species can differ in litter preferences (Hättenschwiler and Gasser 2005).
Litterbag material was made of woven nylon with a mesh size of 0.25 mm, allowing access to soil microbes, microdetritivores and roots, but not macrodetritivores. However, prior work in the Serengeti found that termites eat through nylon litterbags to access plant litter creating holes ~0.55 mm2 in size (Smith et al. 2019). To quantify the contributions of macrodetritivores to litter decomposition, we used a stainless steel metal mesh treatment with an aperture size of 0.3 mm designed to exclude the head-width of the smallest foraging termites (Smith et al. 2019; Teo et al. 2020). Our litterbag approach targeted termites, but could equally apply to other savannah macrodetritivores with strong mandibles such as beetle larvae, millipedes and woodlice. Within each 20 × 20 cm plot we buried four litterbags in a factorial design using two pairs of labile and recalcitrant tea litter, one pair open to macrodetritivores (hereafter ‘accessible to macrodetritivores’) and the other pair excluding macrodetritivores with metal mesh (hereafter ‘excluding macrodetritivores’) (sensu (Griffiths et al. 2019); Fig. 1). In total 1488 litterbags were buried as part of the main experiment.
Tea litterbag decomposition locations and experimental set-up. Tea litterbags was buried in agricultural lands (A), pasturelands (P), inside wildlife protected areas (W) and a common garden (C) in the centre of the Serengeti National Park (SNP), Tanzania. Agricultural and pasture site are in close promixity to one another. The common garden consists of blocks with transplanted soil from each respective land-use (A, P, W) from each site and region in the main experiement and a plot with local common garden soil (C). The regional gradient in rainfall is shown from wetter west (blue symbols) to drier east (yellow symbols)
Following the Tea Bag Index protocol for warm climates (Keuskamp et al. 2013), one litterbag was placed in each corner of the excavated plot at a soil depth of 8 cm and incubated for approximately 2 months (52 days in the wet season and 69 days in the dry season). The majority of leaf litter decomposes on the soil-surface in tropical ecosystems. Burial of litter avoids decomposition by superterranean detritivore species, fire and UV degradation (Austin 2011; Cornwell et al. 2009; Davies et al. 2013). Nevertheless, we opted to follow the Tea Bag Index methodology and assumed observed decomposition processes were likely to be similar to those acting on root litter decomposition in savannahs (Smith et al. 2019). Upon collection, litterbags were taken out of the metal mesh, brushed clean of any adhering soil, plant roots and termites, then placed in paper bags and air-dried (30–40 °C) within 4 days of collection.
Common garden experiment
To further disentangle climatic effects of season and rainfall region and edaphic effects of land-use in the main experiment, an additional common garden experiment was established. The common garden was located near to the Serengeti Wildlife Research station in Seronera in the central part of the Serengeti National Park (Fig. 1). This involved decomposing litter in soil transplanted from the different rainfall regions and land-uses to a single location. Transplanting soil allowed us to examine the direct climate response of litter decomposition by microbes and microdetritivores, controlling for soil properties, and the indirect response of macrodetritivores as soil fauna originating from the common garden could enter transplanted soil. From the common garden, we expected that if rainfall was the main driver of litter mass loss, decomposition by microbes and microdetritivores or macrodetritivores would be similar in the main experiment and common garden when rainfall was similar between the two experiments. Here, litter mass loss from the common garden plotted against litter mass from the main experiment would follow a one-to-one line. Conversely, if there was less rainfall in the main experiment than common garden, or vice versa, then litter mass loss would deviate from a one-to-one line. If soil properties, transplanted microbial community or the local macrodetritivore community were the main drivers of litter mass loss, we would expect similarities or deviations from the one-to-one line to be site-specific or relate to land-use rather than variation in rainfall between the two experiments. The common garden site comprised a total of 50 m2 with four experiment blocks (Fig. 1). Each block was approximately 2 m2 and located a minimum of 5 m apart from one another. Within each block we established seven plots, the same area and size as the main experiment plots though excavated slightly deeper to ensure that the litterbags decomposed in transplanted soil, thus totalling 28 plots.
In the common garden experiment, 24 out of 28 plots were randomly assigned to be filled with soil from sites in the main experiment. Approximately 25 l of fresh soil were collected down to a depth of 20 cm at each site in the main experiment. Excavated soil was transported in loosely sealed plastic buckets to the common garden within 5 days. The remaining four common garden plots, one plot per block, were re-filled with local soil to serve as controls (Fig. 1). In each plot, four litterbags – a combination of labile and recalcitrant tea litter, with and without metal mesh to exclude macrodetritivores – were buried in soil immediately after creating the plot. Litterbags were incubated for 2 months and collected following the protocols outlined above. The common garden experiment was repeated for both the wet and dry season, removing old soil and re-collecting fresh soil for each season. In total, 224 litterbags were buried in the common garden experiment.
Soil moisture and rainfall
At the start of incubation and at litterbag collection, spot measurements of soil moisture were taken using hand-held probes in every plot. Soil moisture was measured via electrical conductivity (±0.1%) at a depth of 5.5 cm (ML3, Delta-T, Cambridge, U.K.). All measurements were taken between 7:15 h and 18:30 h during daylight hours. One permanent logger was established in one site per land-use within each rainfall region, to measure soil moisture throughout the experiment. Soil moisture was measured via electrical conductivity using a Decagon Device Em5b Analog data logger and GS1 water content sensor with a probe length of 5 cm (± 0.03 m−3 m−3 equivalent to ±3% volumetric water content in mineral soils). To reduce the visibility of data loggers due to risk of theft, loggers were buried next to the base of trees. Extended buried cables were used so that the probe was placed outside of the tree canopy and between 1.5 and 2 m from the tree trunk. Loggers were regularly checked and replaced. During the litterbag incubation period we only had missing data for soil moisture for agricultural land in the mesic region and pastureland in the wet region during the dry season due to repeated logger theft. To obtain a comparable zero measure across loggers, all soil moisture logger readings were adjusted for differences in soil water holding capacity by subtracting the lowest recorded value from all values for each logger soil type.
Rainfall for the wet and dry season incubation periods were obtained from satellite-based daily rainfall estimates from NASA’s Goddard Earth Sciences Data and Information Services Centre (Huffman 2017), based upon half-hourly measurements of cloud cover retrieved using multi-satellite microwave data at 10 × 10 km resolution. Previous work at the study sites showed significant positive correlation between these remote satellite based estimates of rainfall and soil moisture content (Smith et al. 2020). From daily satellite rainfall estimates we calculated cumulative rainfall for each seasonal incubation period averaged at the site-scale.
Measurements
Litter decomposition was calculated as ash-corrected percentage mass loss for the duration of each incubation period. Prior to burial, all litterbags were weighed (±0.001 g) with tea litter weights calculated by deducting the standard weight of nylon mesh, cord and label −0.25 g (Keuskamp et al. 2013). After the decomposition experiment, litterbags were oven-dried at 60 °C for 48 h and re-weighed. Litter was then extracted from the litterbag and weighed separately. Due to termite and other macrodetritivore intrusions into litterbags, remaining plant litter needed to be corrected for the weight of soil debris. Decomposed litter and debris inside litterbags was homogenized by pestle and mortar. Subsamples of homogenized litter were burned in a furnace at 550 °C for 4 h to determine Loss of Ignition (LOI). The remaining inorganic mineral ash was used to correct for amount of soil in the litterbags. Litter from 10 undecomposed labile and recalcitrant litterbags were also combusted via LOI to estimate undecomposed litter ash content. Litter mass differences of ash-corrected undecomposed and decomposed litter were used to calculate ash-corrected percentage litter mass loss.
Soil texture, carbon and nitrogen concentrations were determined from soil samples collected for the common garden experiment (sampling outlined above). Soil was sieved to 2 mm to remove stones and homogenised using pestle and mortar. Soil texture was determined using wet season samples only, following the pipette method (Gee and Bauder 1986). In brief, deionized water and hydrogen peroxide were added to 10 g of soil, which was heated until the organic material was fully oxidized. Water was added to each sample, rather than hydrochloric acid due to high pH, and the resultant solution went through a sedimentation analysis by repeatedly removing solution and heating to determine percentage of clay, silt and sand by weighing the precipitate. Soil carbon and nitrogen concentrations were determined for both wet and dry season soil samples. Soil subsamples of 16–22 mg were analysed for carbon and nitrogen concentrations by dry-combustion using an automated elemental analyser (vario MICRO cube, Langenselbold, Germany).
Statistical analyses
Tea litter from 1604 litterbags (out of 1712 buried) were recovered from the main decomposition experiment and the common garden experiment across both seasons. The effects of land-use, macrodetritivore exclusion, season and rainfall region on litter mass loss were analysed separately for labile and recalcitrant litter types using generalized linear mixed models. Percentage litter mass loss was fitted using a Beta distribution transforming mass loss to values between 0 and 1, thus ensuring model predictions were bounded between 0 and 100% after back-transformation. In our full models, fixed effect terms included: land-use (agricultural, pastureland and wildlife protected area), macrodetritivore exclusion (litter accessible to or excluding macrodetritivores), season (wet and dry) and rainfall region (wet and mesic) along with two and three-way interactions. Inclusion of rainfall region as a fixed term captured part of the spatial design of the experiment, and the remaining spatial structure was incorporated into random components, namely burial plot nested within replicate site. Soil moisture spot measurements were analysed separately using a generalized linear mixed model fitted with a Gaussian distribution and the same model structure outlined above with the omission of macrodetritivore exclusion, but with the inclusion of soil sand content and soil carbon-to-nitrogen ratio.
The common garden experiment was analysed by subtracting the mass loss in the main decomposition experiment from the common garden experiment. Mass loss in the main experiment was averaged at the site-scale because each site in the main experiment corresponded to a single plot in the common garden experiment. Difference in mass loss between the experiments were analysed using a linear-mixed model fitted with a Gaussian distribution. In the model season, rainfall region, land-use, and macrodetritivore exclusion were fixed effect factors without any interactions due to lower number of data points at the site-scale. Nevertheless, site was retained as random factor to account for paired litterbags accessible to and excluding macrodetritivores.
Final models were simplified following Akaike’s Information Criterion (AIC), removing terms from the full model to improve the model likelihood and lower AIC value. Fixed variables were retained if significant in Likelihood Ratio Tests. For the final model, significance of each term was assessed contrasting models using Maximum Likelihood with and without fixed factors to generate P-values (Bolker et al. 2009; Zuur et al. 2009). Significant differences within terms and interactions were obtained through multiple contrasts as a function of least square means. All analyses were carried out in R version 3.5.3 (R Foundation for Statistical Computing, 2019) with GLMM and LMM models tested using the ‘glmmTMB’ and ‘lmer’ functions in lme4 (Bates et al. 2015) and glmmTMB (Brooks et al. 2017) packages and model contrasts using the emmeans package (Lenth 2016).
Results
Litter decomposition by macrodetritivores across land-uses and seasons
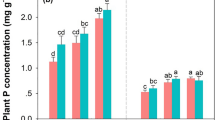
Macrodetritivore accessibility significantly increased litter mass loss of recalcitrant (rooibos tea) litter, but macrodetritivores seemingly did not affect mass loss of labile (green tea) litter (Fig. 2; Table 2). Recalcitrant litter accessible to macrodetritivores had significantly higher mass loss on agricultural and pasturelands than wildlife protected areas (Table 2, i.e. a significant land-use × macrodetritivore exclusion interaction). Contrasting litter accessible and inaccessible to macrodetritivores, macrodetritivores enhanced recalcitrant litter decomposition on average by 6% in agricultural and pastural land compared to wildlife protected areas across seasons and rainfall regions. However, there were instances when the contribution from macrodetritivores to recalcitrant litter decomposition was greater in wildlife protected areas than agriculture and pasturelands depending on the season and rainfall region (see results below). Land-use alone did not significantly influence recalcitrant litter decomposition (Table 2). Across all land-uses, recalcitrant litter mass loss was 21.6% higher for litter accessible to macrodetritivores compared to excluding macrodetritivores, an increase from 23.7 to 45.3% mass loss, respectively (Fig. 2).
Mass loss of labile green and rooibos recalcitrant tea leaf litter across seasons (wet and dry), rainfall regions (mesic and wet) and land-uses (agriculture, pasture and wildlife protected areas) around the Serengeti ecosystem. Labile (circle symbol) and recalcitrant (square symbol) tea litter are shown either as “excluding macrodetritivores” when metal mesh was used (filled symbols) or “accessible to macrodetritivores” without metal mesh (open symbols) across different land-uses: agriculture (brown), pastures (orange) and wildlife protected areas (green). Error bars are shown as ±1 standard deviation
During the dry season, the contribution of macrodetritivores to recalcitrant litter decomposition was consistently greater in agricultural and pasturelands than in wildlife protected areas (Fig. 2a–b). The contribution of macrodetritivores to recalcitrant litter decomposition was also higher overall in the wet compared to the mesic region (Fig. 2a–b). During the dry season, soil moisture loggers recorded elevated soil water content in the wet region (Fig. 3), which corroborated with satellite estimates of 150 mm rainfall. Yet, there was no detectable rainfall in the mesic region with satellite estimates of only 8 mm rainfall (Fig. 3). Macrodetritivores increased recalcitrant litter decomposition in non-transplanted local control soil in the common garden experiment during the dry season when the common garden received an estimated 172 mm rainfall (Fig. 4a). Despite negligible rainfall during the dry season in the mesic region, recalcitrant litter decomposition accessible to macrodetritivores exceeded labile litter decomposition by 15, 8 and 3% in pastures, agricultural land and wildlife protected areas, respectively (Fig. 2a).
Temporal and regional patterns in soil moisture measured using permanent soil moisture loggers (grey circles) and spot measurements using a hand-held probe (black filled squares). Logger readings are for one site per land-use and rainfall region (a–g). Spot measurements are averages from multiple litterbag decomposition points (eight per site) measured during the daytime at the start and end of wet season (grey box) and dry season incubation period (white box). Multiple moisture loggers on the agricultural land in mesic rainfall region and pastureland in wet region were stolen resulting in missing data during the litterbag incubation period. Loggers in all wildlife protected areas were disturbed by animals leading to some intermittent readings. Error bars for spot measurements are shown as ±1 standard deviation
Relationship between mass loss of labile green and recalcitrant rooibos tea litter incubated across seasons, rainfall regions and land-uses in the main experiment compared to the common garden in central Serengeti. Each data point represents the mean mass loss for a given land-use across four sites in the main experiment (x-axis) compared to the mean mass loss in four plots across four replicated blocks of transplanted land-use soils placed in the common garden (y-axis). Labile (circle symbol) and recalcitrant (square symbol) tea litter decomposition is shown either as “excluding macrodetritivores” using metal mesh (filled symbols) or “accessible to macrodetritivores” without metal mesh (open symbols) across different land-uses: agriculture (brown), pastures (orange), wildlife protected areas (green) and local common garden soil (grey). Estimated rainfall during the litterbag incubation periods are stated for each season, rainfall region and the common garden. The solid black line depicts the one-to-one line and error bars are shown as ±1 standard deviation
During the wet season and in the mesic region, recalcitrant litter decomposition by macrodetritivores in wildlife protected areas was higher than in pasture and agricultural land with 17 and 25% greater mass loss on average, respectively (Fig. 2c). This upregulation in the contribution of macrodetritivore to litter decomposition in wildlife protected areas in the mesic region during the wet season resulted in multiple combinations of significant three-way interactions with land-use, macrodetritivore exclusion, season and rainfall region (Table 2).
Season and macrodetritivore accessibility were the most important factors resulting in recalcitrant litter decomposition in the main experiment to deviate from decomposition observed in the common garden experiment, i.e. the deviation from the one-to-one line (Fig. 4; Table 3). In the wet season, when all sites received similar quantities of rainfall, decomposition of recalcitrant litter accessible to macrodetritivores differed between the main experiment and common garden (Fig. 4b, d). We found no effect of land-use, determined by transplanting soil, on recalcitrant litter decomposition in the common garden (Table 3). Furthermore, rainfall region did not significantly explain differences between the main and common garden experiment due to sustained decomposition by macrodetritivores in the dry season in both rainfall regions and the common garden (Fig. 4a, c; Table 3).
Litter decomposition without macrodetritivores across land-uses depends on precipitation
Decomposition of labile and recalcitrant litter by microbes and microdetritivores, excluding macrodetritivores, did not significantly differ between wildlife protected areas, agricultural and pasturelands (Table 2; Fig. 2). However, significant land-use differences in labile litter mass loss occurred during the dry season (Table 2). In the wet region, labile litter mass loss was significantly higher in wildlife protected areas than in agricultural and pasturelands (Fig. 2b), while in the mesic region, agricultural lands had significantly higher litter mass loss compared to pasturelands and wildlife protected areas (Fig. 2c). These land-use patterns in decomposition of labile litter by microbes and microdetritivore followed patterns of soil moisture. In the wet region, permanent soil moisture loggers estimated wildlife protected areas to be on average 27% wetter than agricultural lands, although we lacked data for a comparison to pasturelands (Fig. 3b, d, f; Table 2). In the mesic region, soil moisture spot measurements at two agricultural sites, characterized as vertisols (Table 1), were wetter than neighbouring sites and other land-uses during the dry season. These agricultural sites on vertisols had on average 13% soil moisture compared to 4% in the other agricultural sites, 5% in pastures and 10% in wildlife protected areas (Fig. 3a, c, e). Unfortunately, we lacked soil moisture logger readings for agricultural land in the mesic region during the dry season (see methods).
Season and rainfall region, but not land-use or macrodetritivore exclusion, exerted the strongest influence on labile litter decomposition in the common garden experiment (Fig. 4). During the wet season, labile litter (and recalcitrant litter excluding macrodetritivores) mass loss in the main experiment was similar to the common garden experiment, i.e. following the one-to-one line (Fig. 4b, d; Table 3). This similarity corresponds with the uniform amount of rainfall across the mesic region, wet region and common garden during the wet season (Fig. 3). In the main experiment, there were no significant land-use or rainfall region differences in decomposition of labile litter by microbes and microdetritivores during the wet season (Fig. 2c, d; Table 2). Meanwhile, significantly drier conditions in the mesic region, and wetter conditions in the common garden during the dry season, reduced decomposition by microbes and microdetritivores in the main experiment compared to the common garden experiment (Fig. 4a; Table 3).
Land-use impact on soil moisture
Land-use exerted a weaker influence on soil moisture compared to season or rainfall region, with no statistically significant influence on soil moisture spot measurements (Table 2). All land-uses were wetter during the wet season compared to the dry season. However, during the wet season, soil moisture across rainfall regions was similar in pasturelands, but agricultural land and wildlife protected areas were wetter in the mesic compared to the wet region (Fig. 3; Table 2, i.e. land-use × season × rainfall region). Spot measurements of moisture were negatively related to sand content, i.e. greater water infiltration rates, but not significantly related to soil C:N ratio (Table 2).
Discussion
Our study demonstrates that recalcitrant litter decomposition by macrodetritivores can be sustained in human-modified tropical savannahs. Contrary to our hypotheses, the contribution of macrodetritivores to litter decomposition was not diminished by agricultural cultivation of savannahs compared pasturelands or wildlife protected areas. Moreover, on both agricultural and pastoral land-uses in drier regions, macrodetritivore communities were able to sustain recalcitrant litter decomposition during periods of water scarcity. Continued macrodetritivore foraging during the dry season has been observed in other tropical ecosystems (Ashton et al. 2019; Jouquet et al. 2011; Veldhuis et al. 2017), but is not exhibited by all savannah macrodetritivore species (Davies et al. 2015; Davies et al. 2013; Leitner et al. 2018). In contrast, litter decomposition by microbes and microdetritivores in our study was constrained by regional and seasonal water availability across all land-use types, as also seen in other tropical ecosystems (Anaya et al. 2012; Becker and Kuzyakov 2018; Ngatia et al. 2014). In spite of a lower sensitivity to variable water availability, litter decomposition by macrodetritivores exhibited strong seasonal differences across land-uses and regions suggesting alternative factors regulating the temporal dynamics of macrodetritivore foraging. Currently macrodetritivores continue to contribute to litter decomposition outside of the borders of the Serengeti, yet the longer-term resilience of this ecosystem function requires further research on the composition, abundance and ecology of the macrodetritivore communities.
Across the Serengeti, recalcitrant litter decomposition by macrodetritivores was on average higher on agropastoral land-uses, yet there were periods of elevated decomposition by macrodetritivores in wildlife protected areas. We are unable to determine to what extent differences in macrodetritivore species composition and abundance between land-uses underlie differences in decomposition. Nevertheless, our results show the contribution of macrodetritivores to litter decomposition was fairly consistent on agricultural and pasture land and more seasonally variable in wildlife protected areas – notably upregulated in the mesic region during the wet season and common garden during the dry season (Figs. 2c and 4b, c). Both these periods coincided with high wild herbivore abundances. During the wet season (January–April) the wildebeest migration concentrates in wildlife protected areas of the mesic region (Boone et al. 2006; Hopcraft et al. 2015) and during the dry season (July–October) wildebeest pass central Serengeti (Boone et al. 2006). As investigated by other studies in wildlife protected savannahs, the extent of macrodetritivore foraging may fluctuate seasonal depending on resource availability derived from wild herbivores (Buitenwerf et al. 2011; Freymann et al. 2010; Sitters et al. 2014). Likewise, higher decomposition by macrodetritivores on agropastoral land might reflect larger amounts of litter and detritus generated from crop residues, dropped or senesced plant material from livestock grazing and livestock dung (Dangerfield 1990; Hagan et al. 2017; Sileshi et al. 2010; Wood et al. 1980). This said, resource availability is one of several possible reasons for differences in macrodetritivore litter consumption between land-uses, other reasons could include species-specific foraging behaviours, competition between detritivore species, regulation of macrodetritivore abundances by predators (i.e. ants, aardvarks, aardwolves etc.) amongst others (Abe et al. 2000; Hagan et al. 2017; Jouquet et al. 2011; Kruuk and Sands 1972; Sileshi et al. 2010).
Macrodetritivores in drier regions sustained plant litter decomposition during periods of water scarcity in the Serengeti. However, litter decomposition by macrodetritivore was highest in regions that received rainfall during the dry season rather than in the mesic region without rainfall. In South Africa, Veldhuis et al. (2017) found that litter removal by large fungus growing termites versus other macrodetritivores had forage optima between 40 and 60 mm and 80–100 mm of rainfall per month, respectively, and declined above or below these thresholds. We found a similar intermediary rainfall optima for recalcitrant litter decomposition by macrodetritivores in the wet region and common garden during the dry season to be around 68–76 mm of rainfall per month. However, macrodetritivores may be responding to the lower frequency of rainfall in the dry season rather than the lower quantity of rainfall. In a mesocosm manipulation, Joly et al. (2019) showed that litter decomposition by woodlice increased with less frequent rainfall events when controlling for rainfall quantity. The exoskeleton of several macrodetritivores reduces sensitivity to variations in soil moisture, yet there is likely to be a moisture threshold whereby macrodetritivore foraging is constrained by the risk of desiccation and mortality (Joly et al. 2019; Woon et al. 2019). This moisture threshold for macrodetritivore foraging can be prolonged by soil properties that retain water between rainfall events (Cornelius and Osbrink 2010), as seen in vertisols on agricultural land in our mesic region (Fig. 2a, c). Macrodetritivores may be less sensitive to fluctuations in soil water than soil microbes and microdetritivores, but it is important to note that higher litter decomposition by macrodetritivores occurs when water is available.
One of the most striking results from our litter decomposition experiments was that macrodetritivore consumed only recalcitrant litter and avoided labile litter. In this study, we applied the Tea Bag Index that is founded on the principle that distinct litter qualities differ in rates of decomposition by soil microbes. Labile green tea (Camellia sinensis) has a low C:N ratio and high cellulose content and is predicted to decompose quickly, whereas recalcitrant rooibos tea (Aspalanthus linearis) is the opposite, with a high C:N ratio and lignin content (Keuskamp et al. 2013). In the Serengeti, by excluding macrodetritivores, litter mass loss was greater for labile than recalcitrant litter. When accessible to macrodetritivores recalcitrant litter mass loss could equal or exceed labile litter mass loss. A number of species of millipedes, termites and woodlice have been shown to exhibit a preference for recalcitrant or nutrient poor litter qualities over labile litter (Hättenschwiler and Gasser 2005; Peguero et al. 2019; Sitters et al. 2014). As mentioned earlier, termites avoid readily digestible litter – high sugar or labile fractions – due to the fermentation process in termite guts (Abe et al. 2000). Similar results to our study have been found in recent work in South East Asian tropical rainforests testing the exclusion of termites using the Tea Bag Index with and without additional metal mesh. Teo et al. (2020) found both green and rooibos tea had similar detection rates by termites evident from holes in nylon mesh bags; however, only recalcitrant rooibos tea litter had significantly higher mass loss when accessible to termites compared to when excluding termites. Macrodetritivore affinity for recalcitrant litter represents a major challenge utilising decomposition indices using the Tea Bag Index: decomposition rates (K-rates) cannot reliably be calculated due to variable consumption of recalcitrant litter, and, although stabilisation (S factor) of labile fraction into recalcitrant fraction calculation is based on labile litter only, its underlying meaning is questionable as it ignores the contribution of macrodetritivores to litter stabilisation and mineralisation in the soil. For ecosystems where macrodetritivores can easily enter litterbags, K-rates and S factors derived from the Tea Bag Index should be interpreted with caution unless accounting for macrodetiritvore damage or excluding macrodetritivores from litterbags (Teo et al. 2020). Learning from the Tea Bag Index, integrating litter quality into the conceptual framework and design of future global or biome-wide soil fauna decomposition initiatives would provide valuable insights into the generality of macrodetritivore litter preferences.
The contribution of macrodetritivores and soil microorganisms to litter decomposition has been proposed to be synergistic, i.e. macrodetritivores amplify decomposition by microbes (Liu et al. 2015). In the tropical savannahs of Serengeti using tea litter, we found decomposition by macrodetritivores versus microbes and microdetritivore to have divergent effects depending on human land-use, water availability and litter type. These conclusions are supported by other studies showing that macrodetritivores continue to regulate litter decomposition across spatiotemporal variable rainfall (Ashton et al. 2019; Jouquet et al. 2011; Veldhuis et al. 2017) and that macrodetritivores can be selective foragers (Acanakwo et al. 2019; Smith et al. 2019). Here we show that moderate human intervention is compatible with macrodetritivore litter decomposition in an African savannah. The lack of intense mechanical tillage and use of pesticides may allow macrodetritivores to persist on human dominated land-uses bordering the Serengeti. Raising ecological awareness is required to ensure that the contribution of macrodetritivores to litter decomposition is not lost following the predicted development and intensification of agropastoral land-use practices outside the Serengeti and other wildlife protected savannahs.
References
Abe T, Bignell DE, Higashi M (2000) Termites: evolution, sociality, symbioses. Springer, Ecology
Acanakwo EF, Sheil D, Moe SR (2019) Wood decomposition is more rapid on than off termite mounds in an African savanna. Ecosphere 10:e02554. https://doi.org/10.1002/ecs2.2554
Anaya CA, Jaramillo VJ, Martínez-Yrízar A, García-Oliva F (2012) Large rainfall pulses control litter decomposition in a tropical dry forest: evidence from an 8-year study. Ecosystems 15:652–663. https://doi.org/10.1007/s10021-012-9537-z
Ashton LA, Griffiths HM, Parr CL, Evans TA, Didham RK, Hasan F, Teh YA, Tin HS, Vairappan CS, Eggleton P (2019) Termites mitigate the effects of drought in tropical rainforest. Science 363:174–177. https://doi.org/10.1126/science.aau9565
Austin AT (2011) Has water limited our imagination for aridland biogeochemistry? Trends Ecol Evol 26:229–235. https://doi.org/10.1016/j.tree.2011.02.003
Ayuke FO, Pulleman MM, Vanlauwe B, de Goede RGM, Six J, Csuzdi C, Brussaard L (2011) Agricultural management affects earthworm and termite diversity across humid to semi-arid tropical zones. Agric Ecosyst Environ 140:148–154. https://doi.org/10.1016/j.agee.2010.11.021
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–51. https://doi.org/10.18637/jss.v067.i01
Beare MH, Reddy MV, Tian G, Srivastava SC (1997) Agricultural intensification, soil biodiversity and agroecosystem function in the tropics: the role of decomposer biota. Appl Soil Ecol 6:87–108. https://doi.org/10.1016/S0929-1393(96)00150-3
Becker JN, Kuzyakov Y (2018) Teatime on Mount Kilimanjaro: assessing climate and land-use effects on litter decomposition and stabilization using the tea bag index. Land Degrad Dev 29:2321–2329. https://doi.org/10.1002/ldr.2982
Black HIJ, Okwakol MJN (1997) Agricultural intensification, soil biodiversity and agroecosystem function in the tropics: the role of termites. Appl Soil Ecol 6:37–53. https://doi.org/10.1016/S0929-1393(96)00153-9
Bolker BM, Brooks ME, Clark CJ, Geange SW, Poulsen JR, Stevens MH, White JS (2009) Generalized linear mixed models: a practical guide for ecology and evolution. Trends Ecol Evol 24:127–135. https://doi.org/10.1016/j.tree.2008.10.008
Boone RB, Thirgood SJ, Hopcraft JGC (2006) Serengeti wildebeest migratory patterns modeled from rainfall and new vegetation growth. Ecology 87:1987–1994. https://doi.org/10.1890/0012-9658(2006)87[1987:SWMPMF]2.0.CO;2
Brooks ME, Kristensen K, van Benthem KJ, Magnusson A, Berg CW, Nielsen A, Skaug HJ, Mächler M, Bolker BM (2017) glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. The R J 9:378–400
Buitenwerf R, Stevens N, Gosling CM, Anderson TM, Olff H (2011) Interactions between large herbivores and litter removal by termites across a rainfall gradient in a south African savanna. J Trop Ecol 27:375–382. https://doi.org/10.1017/s0266467411000125
Cornelius ML, Osbrink WLA (2010) Effect of soil type and moisture availability on the foraging behavior of the formosan subterranean termite (Isoptera: Rhinotermitidae). J Econ Entomol 103:799–807. https://doi.org/10.1603/ec09250
Cornwell WK, Cornelissen JHC, Allison SD, Bauhus J, Eggleton P, Preston CM, Scarff F, Weedon JT, Wirth C, Zanne AE (2009) Plant traits and wood fates across the globe: rotted, burned, or consumed? Glob Chang Biol 15:2431–2449. https://doi.org/10.1111/j.1365-2486.2009.01916.x
Dangerfield J (1990) Abundance, biomass and diversity of soil macrofauna in savanna woodland and associated managed habitats. Pedobiologia 34:141–150
Davies AB, Eggleton P, van Rensburg BJ, Parr CL (2015) Seasonal activity patterns of African savanna termites vary across a rainfall gradient. Insect Soc 62:157–165. https://doi.org/10.1007/s00040-014-0386-y
Davies AB, van Rensburg BJ, Eggleton P, Parr CL (2013) Interactive effects of fire, rainfall, and litter quality on decomposition in savannas: frequent fire leads to contrasting effects. Ecosystems 16:866–880. https://doi.org/10.1007/s10021-013-9657-0
de Visser SN, Freymann BP, Foster RF, Nkwabi AK, Metzger KL, Harvey AW, Sinclair AR (2015) Invertebrates of the Serengeti: disturbance effects on arthropod diversity and abundance. In: Sinclair AR, Metzger KL, Mduma SA, Fryxell JM (eds) Serengeti IV: sustaining biodiversity in a coupled human-natural system. The University of Chicago Press, Chicago, IL
Decaëns T, Jiménez JJ, Barros E, Chauvel A, Blanchart E, Fragoso C, Lavelle P (2004) Soil macrofaunal communities in permanent pastures derived from tropical forest or savanna. Agric Ecosyst Environ 103:301–312. https://doi.org/10.1016/j.agee.2003.12.005
Eggleton P, Bignell DE, Hauser S, Dibog L, Norgrove L, Madong B (2002) Termite diversity across an anthropogenic disturbance gradient in the humid forest zone of West Africa. Agric Ecosyst Environ 90:189–202. https://doi.org/10.1016/S0167-8809(01)00206-7
FAO (2015) Global forest resources and assessment 2015. Food and Agircuture Organizatin of the United Nations, Rome, Italy
Freymann BP, Buitenwerf R, Desouza O, Olff H (2008) The importance of termites (Isoptera) for the recycling of herbivore dung in tropical ecosystems: a review. Eur J Entomol 105:165–173. https://doi.org/10.14411/eje.2008.025
Freymann BP, de Visser SN, Mayemba EP, Olff H (2007) Termites of the genus Odontotermes are optionally keratophagous. Ecotropica 13:143–147
Freymann BP, de Visser SN, Olff H (2010) Spatial and temporal hotspots of termite-driven decomposition in the Serengeti. Ecography 33:443–450. https://doi.org/10.1111/j.1600-0587.2009.05960.x
Gee GW, Bauder JW (1986) Particle-size analysis. In: Klute A (ed) Methods of soil analysis, part 1 physical and mineralogical methods. Soil Science Society of America, Madison, WI
Gessner MO, Swan CM, Dang CK, McKie BG, Bardgett RD, Wall DH, Hättenschwiler S (2010) Diversity meets decomposition. Trends Ecol Evol 25:372–380. https://doi.org/10.1016/j.tree.2010.01.010
Giller KE, Beare MH, Lavelle P, Izac AMN, Swift MJ (1997) Agricultural intensification, soil biodiversity and agroecosystem function. Appl Soil Ecol 6:3–16. https://doi.org/10.1016/S0929-1393(96)00149-7
Griffiths HM, Ashton LA, Evans TA, Parr CL, Eggleton P (2019) Termites can decompose more than half of deadwood in tropical rainforest. Curr Biol 29:R118–R119
Hagan JG, du Toit JCO, Cramer MD (2017) Long-term livestock grazing increases the recruitment success of epigeal termites: insights from a >75-year grazing experiment in the Karoo, South Africa. African J Range Forage Sci 34:123–132. https://doi.org/10.2989/10220119.2017.1314981
Hättenschwiler S, Gasser P (2005) Soil animals alter plant litter diversity effects on decomposition. Proc Natl Acad Sci U S A 102:1519–1524. https://doi.org/10.1073/pnas.0404977102
Hopcraft JG, Holdo RM, Mwangomo E, Mduma SA, Thirgood SJ, Borner M, Fryxell JM, Olff H, Sinclair AR (2015) Why are wildebeest the most abundant herbivore in the Serengeti ecosystem? In: Sinclair AR, Metzger KL, Mduma SA, Fryxell JM (eds) Serengeti IV: sustaining biodiversity in a coupled human-natural system. The University of Chicago Press, Chicago, IL
Huffman G (2017) GPM IMERG final precipitation L3 1 day 0.1 degree × 0.1 degree V05. In: Savtchenko AK (ed). Goddard earth sciences data and information services center (GES DISC)
ISRIC (2018) SoilGrids - Global gridded soil information. Retrieved from https://www.isric.org/explore/soilgrids
Joly F-X, Weibel AK, Coulis M, Throop HL (2019) Rainfall frequency, not quantity, controls isopod effect on litter decomposition. Soil Biol Biochem 135:154–162. https://doi.org/10.1016/j.soilbio.2019.05.003
Jouquet P, Traoré S, Choosai C, Hartmann C, Bignell D (2011) Influence of termites on ecosystem functioning, ecosystem services provided by termites. Eur J Soil Biol 47:215–222. https://doi.org/10.1016/j.ejsobi.2011.05.005
Kamau S, Barrios E, Karanja NK, Ayuke FO, Lehmann J (2017) Soil macrofauna abundance under dominant tree species increases along a soil degradation gradient. Soil Biol Biochem 112:35–46. https://doi.org/10.1016/j.soilbio.2017.04.016
Keuskamp JA, Dingemans BJJ, Lehtinen T, Sarneel JM, Hefting MM (2013) Tea bag index: a novel approach to collect uniform decomposition data across ecosystems. Methods Ecol Evol 4:1070–1075. https://doi.org/10.1111/2041-210X.12097
Kruuk H, Sands WA (1972) The aardwolf (Proteles cristatm Sparrman) 1783 as predator of termites*. Afr J Ecol 10:211–227. https://doi.org/10.1111/j.1365-2028.1972.tb00728.x
Leitner M, Davies AB, Parr CL, Eggleton P, Robertson MP (2018) Woody encroachment slows decomposition and termite activity in an African savanna. Glob Chang Biol 24:2597–2606. https://doi.org/10.1111/gcb.14118
Lenth R (2016) Least-Squares Means: The R Package lsmeans.Journal of Statistical Software, 69(1):1–33. https://doi.org/10.18637/jss.v069.i01
Liu G, Cornwell WK, Cao K, Hu Y, Van Logtestijn RSP, Yang S, Xie X, Zhang Y, Ye D, Pan X, Ye X, Huang Z, Dong M, Cornelissen JHC, Austin A (2015) Termites amplify the effects of wood traits on decomposition rates among multiple bamboo and dicot woody species. J Ecol 103:1214–1223. https://doi.org/10.1111/1365-2745.12427
McNally A, Arsenault K, Kumar S, Shukla S, Peterson P, Wang S, Funk C, Peters-Lidard CD, Verdin JP (2017) A land data assimilation system for sub-Saharan Africa food and water security applications. Sci Data 4:170012. https://doi.org/10.1038/sdata.2017.12
Ngatia LW, Reddy KR, Nair PKR, Pringle RM, Palmer TM, Turner BL (2014) Seasonal patterns in decomposition and nutrient release from east African savanna grasses grown under contrasting nutrient conditions. Agric Ecosyst Environ 188:12–19. https://doi.org/10.1016/j.agee.2014.02.004
Norton-Griffiths M, Herlocker D, Pennycuick L (1975) The patterns of rainfall in the Serengeti ecosystem, Tanzania. Afr J Ecol 13:347–374. https://doi.org/10.1111/j.1365-2028.1975.tb00144.x
Okwakol MJN (2000) Changes in termite (Isoptera) communities due to the clearance and cultivation of tropical forest in Uganda. Afr J Ecol 38:1–7. https://doi.org/10.1046/j.1365-2028.2000.00189.x
Osborne CP, Charles-Dominique T, Stevens N, Bond WJ, Midgley G, Lehmann CER (2018) Human impacts in African savannas are mediated by plant functional traits. New Phytol 220:10–24. https://doi.org/10.1111/nph.15236
Peguero G, Sardans J, Asensio D, Fernández-Martínez M, Gargallo-Garriga A, Grau O, Llusià J, Margalef O, Márquez L, Ogaya R, Urbina I, Courtois EA, Stahl C, Van Langenhove L, Verryckt LT, Richter A, Janssens IA, Peñuelas J (2019) Nutrient scarcity strengthens soil fauna control over leaf litter decomposition in tropical rainforests. Proc Royal Soc B Biol Sci 286:20191300. https://doi.org/10.1098/rspb.2019.1300
Sileshi GW, Arshad MA, Konate S, Nkunika POY (2010) Termite-induced heterogeneity in African savanna vegetation: mechanisms and patterns. J Veg Sci 21:923–937. https://doi.org/10.1111/j.1654-1103.2010.01197.x
Sinclair ARE, Mduma SAR, Hopcraft JGC, Fryxell JM, Hilborn RAY (2007) Long-term ecosystem dynamics in the Serengeti: lessons for conservation. Conserv Biol 21:580–590. https://doi.org/10.1111/j.1523-1739.2007.00699.x
Sitters J, Maechler M-J, Edwards PJ, Suter W, Olde Venterink H, Kay A (2014) Interactions between C : N : P stoichiometry and soil macrofauna control dung decomposition of savanna herbivores. Funct Ecol 28:776–786. https://doi.org/10.1111/1365-2435.12213
Smith SW, Graae BJ, Bukombe J, Hassan SN, Lyamuya RD, Jacob Mtweve P, Treydte AC, Speed JDM (2020) Savannah trees buffer herbaceous plant biomass against wild and domestic herbivores. Appl Veg Sci 23:185–196. https://doi.org/10.1111/avsc.12472
Smith SW, Speed JDM, Bukombe J, Hassan SN, Lyamuya RD, Mtweve PJ, Sundsdal A, Graae BJ (2019) Litter type and termites regulate root decomposition across contrasting savanna land-uses. Oikos 128:596–607. https://doi.org/10.1111/oik.05697
Teo A, Kristensen NP, Keuskamp JA, Evans TA, Foo M, Chisholm RA (2020) Validation and extension of the tea bag index to collect decomposition data from termite-rich ecosystems. Pedobiologia 80:150639. https://doi.org/10.1016/j.pedobi.2020.150639
Veldhuis MP, Laso FJ, Olff H, Berg MP (2017) Termites promote resistance of decomposition to spatiotemporal variability in rainfall. Ecology 98:467–477. https://doi.org/10.1002/ecy.1658
Veldhuis MP, Ritchie ME, Ogutu JO, Morrison TA, Beale CM, Estes AB, Mwakilema W, Ojwang GO, Parr CL, Probert J, Wargute PW, Hopcraft JGC, Olff H (2019) Cross-boundary human impacts compromise the Serengeti-Mara ecosystem. Science 363:1424–1428. https://doi.org/10.1126/science.aav0564
Wood TG, Johnson RA, Ohiagu CE (1980) Termite damage and crop loss studies in Nigeria — a review of termite (Isoptera) damage to maize and estimation of damage, loss in yield and termite (Microtermes) abundance at Mokwa. Trop Pest Manag 26:241–253. https://doi.org/10.1080/09670878009414406
Woon JS, Boyle MJW, Ewers RM, Chung A, Eggleton P (2019) Termite environmental tolerances are more linked to desiccation than temperature in modified tropical forests. Insect Soc 66:57–64. https://doi.org/10.1007/s00040-018-0664-1
Zuur AF, Leno E, Walker N, Saveliev AA, Smith GM (2009) Mixed effects models and extensions in ecology with R. Springer-Verlag, New York
Acknowledgements
We thank AfricanBioServices project coordinator Eivin Røskaft and director Robert Fyumagwa and staff at Serengeti Wildlife Research Centre, Tanzanian Wildlife Research Institute, Tanzanian National Parks and Tanzanian Commission for Science and Technology for support in undertaking fieldwork and all the field assistance from various ranger posts and villagers. We are indebted to Rozália Kapás for processing teabags. Comments and suggestions by Stein Moe, Kenny Helsen, Louis Hunninck and Susanna Karlsson improved earlier versions of this manuscript. This work was part of the AfricanBioServices project funded by the EU Horizon 2020 grant number 641918.
Funding
Open Access funding provided by NTNU Norwegian University of Science and Technology (incl St. Olavs Hospital - Trondheim University Hospital).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Yolima Carrillo.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sundsdal, A., Graae, B.J., Speed, J.D.M. et al. Teatime in the Serengeti: macrodetritivores sustain recalcitrant plant litter decomposition across human-modified tropical savannahs. Plant Soil 456, 241–258 (2020). https://doi.org/10.1007/s11104-020-04704-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-020-04704-z