Abstract
Aims
Nickel hyperaccumulator plants require highly evolved mechanisms to avoid cellular-level toxicity to cope with the high prevailing concentrations of nickel in their seeds and seedlings. This study aimed to investigate tissue-level distribution and redistribution of Ni and other physiologically relevant elements during the germination of hyperaccumulator plants.
Methods
Berkheya coddii Roessler (Asteraceae) and Odontarrhena muralis sensu latu (Waldst. & Kit.) Endl. (O. chalcidica), formerly Alyssum murale (Waldst. & Kit.) (Brassicaceae), were germinated for several days at room temperature before using synchrotron micro X-ray Fluorescence Spectroscopy (µXRF) to obtain high-sensitivity and high-resolution elemental images of live/hydrated plants at various stages of seed germination and seedling growth.
Results
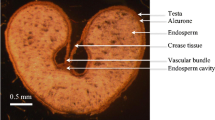
The results show that fruits and seeds of both species were highly enriched in nickel. In Berkheya coddii, nickel was located in the cotyledons, the micropylar area, the seed coat, and the point of attachment of the pappus with the cypsela body. In Odontarrhena muralis seeds, nickel occurred in the cotyledons and hypocotyl. The emergence of true leaves initiates nickel and calcium redistribution within seedlings in both species.
Conclusions
The diversity in physiological responses to nickel in Berkheya coddii and Odontarrhena muralis does not only occur at the mature stage, but is inherent to both species as seed elemental storage and tolerance mechanisms during seedling development differ.








Similar content being viewed by others
References
Afshar N, De Jonge M, Paterson D, Howard D, Starritt A (2017) Optimised multi-dimensional image scanning with RASCAN. ANSTO, Australian Synchrotron
Anderson TR, Howes AW, Slatter K, Dutton MF (1997) Studies on the nickel hyperaccumulator Berkheya coddii. In: Jaffré T, Reeves RD, Becquer T (eds) The ecology of ultramafic and metalliferous areas. Centre ORSTOM de Noumea, New Caledonia, pp 261–266
Bani A, Pavlova D, Echevarria G, Mullaj A, Reeves RD, Morel JL, Sulçe S (2010) Nickel hyperaccumulation by the species of Alyssum and Thlaspi (Brassicaceae) from the ultramafic soils of the Balkans. Bot Serb 34:3–14. https://doi.org/10.1080/15226514.2013.862204
Bani A, Echevarria G, Sulçe S, Morel JL (2015) Improving the agronomy of Alyssum murale for extensive phytomining: A five-year field study. Int J Phytorem 17(2):117–127
Barbaroux R, Meunier N, Mercier G, Taillard V, Morel JL, Simonnot MO, Blais JF (2009) Chemical leaching of nickel from the seeds of the metal hyperaccumulator plant Alyssum murale. Hydrometallurgy 100:10–14. https://doi.org/10.1016/j.hydromet.2009.09.005
Bewley JD, Black M (1994) Seeds. Springer, Boston
Bhatia NP, Orlic I, Siegele R, Ashwath N, Baker AJM, Walsh KB (2003) Elemental mapping using PIXE shows the main pathway of nickel movement is principally symplastic within the fruit of the hyperaccumulator Stackhousia tryonii. New Phytol 160:479–488. https://doi.org/10.1046/j.1469-8137.2003.00912.x
Bhatia NP, Walsh KB, Orilc I, Siegele R, Ashwath N, Baker AJM (2004) Studies on spatial distribution of nickel in leaves and stems of the metal hyperaccumulator Stackhousia tryonii Bailey using nuclear microprobe (micro-PIXE) and EDXS techniques. Funct Plant Biol 31:1061–1074. https://doi.org/10.1071/FP03192
Broadhurst CL, Chaney RL, Angle JS, Erbe EF, Maugel TK (2004a) Nickel localization and response to increasing Ni soil levels in leaves of the Ni hyperaccumulator Alyssum murale. Plant Soil 265:225–242. https://doi.org/10.1007/s11104-005-0974-8
Broadhurst CL, Chaney RL, Angle JS, Maugel TK, Erbe EF, Murphy CA (2004b) Simultaneous hyperaccumulation of nickel, manganese, and calcium in Alyssum leaf trichomes. Environ Sci Technol 38:5797–5802. https://doi.org/10.1021/es0493796
Broadhurst CL, Tappero RV, Maugel TK, Erbe EF, Sparks DL, Chaney RL (2009) Interaction of nickel and manganese in accumulation and localization in leaves of the Ni hyperaccumulators Alyssum murale and Alyssum corsicum. Plant Soil 314:35–48. https://doi.org/10.1007/s11104-008-9703-4
Brooks RR, Morrison RS, Reeves RD, Dudley TR, Akman Y (1979) Hyperaccumulation of nickel by Alyssum linnaeus (Cruciferae). P Roy Soc B-Biol Sci 203:387–403. https://doi.org/10.1098/rspb.1979.0005
Budka D, Mesjasz-Przybyłowicz J, Tylko G, Przybyłowicz WJ (2005) Freeze-substitution methods for Ni localization and quantitative analysis in Berkheya coddii leaves by means of PIXE. Nucl Instrum Methods Phys Res B 231:338–344. https://doi.org/10.1016/j.nimb.2005.01.080
Cecchi L, Bettarini I, Colzi I, Coppi A, Echevarria G, Pazzagli L, Bani A, Gonnelli C, Selvi A (2018) The genus Odontarrhena (Brassicaceae) in Albania: Taxonomy and Nickel accumulation in a critical group of metallophytes from a major serpentine hot-spot. Phytotaxa 351 (1):1
Centofanti T, Tappero RV, Davis AP, Chaney RL (2011) Chelator-buffered nutrient solution is ineffective in extracting Ni from seeds of Alyssum. Int J Phytoremediat 13:434–440. https://doi.org/10.1080/15226514.2010.483264
Deng THB, van der Ent A, Tang YT, Sterckeman T, Echevarria G, Morel JL, Qiu RL (2018) Nickel hyperaccumulation mechanisms: a review on the current state of knowledge. Plant Soil 423:1–11. https://doi.org/10.1007/s11104-017-3539-8
Dudley TR (1964) Synopsis of the genus Alyssum. J Arnold Arboretum 4:358–373
Estrade N, Cloquet C, Echevarria G, Sterckeman T, Deng T-H-B, Tang Y-T, Morel JL (2015) Weathering and vegetation controls on nickel isotope fractionation in surface ultramafic environments (Albania). Earth Planet Sci Lett 423:24–25 https://doi.org/10.1016/j.epsl.2015.04.018
Gei V, Erskine PD, Harris HH, Echevarria G, Mesjasz-Przybyłowicz J, Barnabas AD, Przybyłowicz WJ, Kopittke PM, van der Ent A (2018) New tools for discovery of hyperaccumulator plant species and understanding their ecophysiology. In: van der Ent A, Echevarria G, Baker AJM, Morel JL (eds) Agromining: extracting unconventional resources from plants. Mineral Resource Reviews series. Springer Nature, Cham, pp 117–133. https://doi.org/10.1007/978-3-319-61899-9_7
Groeber S, Przybyłowicz W, Echevarria G, Montargès-Pelletier E, Barnabas A, Mesjasz-Przybyłowicz J (2015) Fate of nickel and calcium in seedlings of the hyperaccumulator Berkheya coddii during germination. Biol Plantarum 59:560–569. https://doi.org/10.1007/s10535-015-0527-9
Jaffré T, Brooks RR, Lee J, Reeves RD (1976) Sebertia acuminata: a hyperaccumulator of nickel from New Caledonia. Science 193:579–580. https://doi.org/10.1126/science.193.4253.579
Jaffré T, Pillon Y, Thomine S, Merlot S (2013) The metal hyperaccumulators from New Caledonia can broaden our understanding of nickel accumulation in plants. Front Plant Sci 4:279. https://doi.org/10.3389/fpls.2013.00279
Jones MWM, Kopittke PM, Casey L, Reinhardt J, Blamey FPC, van der Ent A (2019) Assessing radiation dose limits for X-ray fluorescence microscopy analysis of plant specimens. Ann Bot 125:599–610. https://doi.org/10.1093/aob/mcz195
Kachenko AG, Bhatia NP, Siegele R, Walsh KB, Singh B (2009) Nickel, Zn and Cd localization in seeds of metal hyperaccumulators using µ-PIXE spectroscopy. Nucl Instrum Meth B 267:2176–2180. https://doi.org/10.1016/j.nimb.2009.03.059
Kirkham R, Dunn PA, Kucziewski A, Siddons DP, Dodanwela R, Moorhead G, Ryan CG, De Geronimo G, Beuttenmuller R, Pinelli D, Pfeffer M, Davey P, Jensen M, Paterson D, de Jonge MD, Kusel M, McKinlay J (2010) The Maia spectroscopy detector system: engineering for integrated pulse capture, low-latency scanning and real-time processing. AIP Conf Proc 1234:240–243. https://doi.org/10.1063/1.3463181
Kitajima K, Fenner M (2000) Ecology of seedling regeneration. In: Fenner M (ed) Seeds: the ecology of regeneration in plant communities. CABI Publishing, Wallingford, pp 331–359
Kopittke PM, Punshon T, Paterson DJ, Tappero RV, Wang P, Blamey FPC, van der Ent A, Lombi E (2018) Synchrotron-based X-ray fluorescence microscopy as a technique for imaging of elements in plants. Plant Physiol 178:507–523. https://doi.org/10.1104/pp.18.00759
Kukier U, Peters CA, Chaney RL, Angle JS, Roseberg RJ (2004) The effect of pH on metal accumulation in two Alyssum species. J Environ Qual 33:2090–2102. https://doi.org/10.2134/jeq2004.2090
Kupper H, Lombi E, Zhao FJ, Wieshammer G, McGrath SP (2001) Cellular compartmentation of nickel in the hyperaccumulators Alyssum lesbiacum, Alyssum bertolonii and Thlaspi goesingense. J Exp Bot 52:2291–2300. https://doi.org/10.1093/jexbot/52.365.2291
Lombi E, De Jonge MD, Donner E, Ryan CG, Paterson D (2011) Trends in hard X-ray fluorescence mapping: environmental applications in the age of fast detectors. Anal Bioanal Chem 400:1637–1644. https://doi.org/10.1007/s00216-011-4829-2
McNear DH, Kupper JV (2014) Mechanisms of trichome-specific Mn accumulation and toxicity in the Ni hyperaccumulator Alyssum murale. Plant Soil 377:407–422. https://doi.org/10.1007/s11104-013-2003-7
Merlot S, Sanchez G, de la Torre V, Hanikenne M (2018) Physiology and molecular biology of trace element hyperaccumulation. In: van der Ent A, Echevarria G, Baker AJM, Morel JM (eds) Agromining: Farming for Metals. Springer Nature, Cham, pp 93–116. https://doi.org/10.1007/978-3-319-61899-9_6
Mesjasz-Przybyłowicz J, Przybyłowicz WJ (2002) Micro-PIXE in plant sciences: present status and perspectives. Nucl Instrum Meth B 189:470–481. https://doi.org/10.1016/S0168-583X(01)01127-2
Mesjasz-Przybyłowicz J, Przybyłowicz WJ (2003) Nickel distribution in Berkheya coddii leaves by Micro-PIXE and SEM-EDS. Proc Microsc Soc South Afr 33:68. ISSN/ISBN: 0250-0418/0-620-31180-0
Mesjasz-Przybyłowicz J, Przybyłowicz WJ (2011) PIXE and metal hyperaccumulation: from soil to plants and insects. X-ray Spectrom 40:181–185. https://doi.org/10.1002/xrs.1304
Mesjasz-Przybyłowicz J, Przybyłowicz WJ, Pineda CA (1998) Elemental distribution in young leaves of the Ni hyperaccumulator – Berkheya coddii. Proc Microsc Soc South Afr 28:57. ISSN/ISBN: 0250-0418/0-620-23355-9
Mesjasz-Przybyłowicz J, Przybyłowicz WJ, Pineda CA (2001) Nuclear microprobe studies of elemental distribution in apical leaves of the Ni hyperaccumulator Berkheya coddii. South Afr J Sci 97:591–593
Mesjasz-Przybyłowicz J, Nakonieczny M, Migula P, Augustyniak M, Tarnawska M, Reimold UW, Koeberl C, Przybyłowicz W, Głowacka E (2004) Uptake of cadmium, lead nickel and zinc from soil and water solutions by the nickel hyperaccumulator Berkheya coddii. Acta Biol Cracov Bot 46:75–85
Morrey DR, Balkwill K, Balkwill MJ (1989) Studies on serpentine flora: preliminary analyses of soils and vegetation associated with serpentinite rock formations in the south-eastern Transvaal. S Afr J Bot 55:171–177. https://doi.org/10.1016/S0254-6299(16)31203-0
Morrison RS, Brooks RR, Reeves RD (1980) Nickel uptake by Alyssum species. Plant Sci Lett 17:451–457. https://doi.org/10.1016/0304-4211(80)90132-7
Nkrumah PN, Echevarria G, Erskine PD, Chaney RL, Sumail S, van der Ent A (2019a) Effect of nickel concentration and soil pH on metal accumulation and growth in tropical agromining ‘metal crops’. Plant Soil 443:27–39. https://doi.org/10.1007/s11104-019-04200-z
Nkrumah PN, Echevarria G, Erskine PD, Chaney RL, Sumail S, van der Ent A (2019b) Soil amendments affecting nickel uptake and growth performance of tropical ‘metal crops’ used for agromining. J Geochem Explor 203:78–86. https://doi.org/10.1016/j.gexplo.2019.03.009
Paterson D, de Jonge MD, Howard DL, Lewis W, McKinlay J, Starritt A, Kusel M, Ryan CG, Kirkham R, Moorhead G, Siddons DP (2011) The X-ray flurorescence microscopy beamline at the Australian Synchroton. AIP Conf Proc 1365:219–222. https://doi.org/10.1063/1.3625343
Przybyłowicz WJ, Pineda CA, Prozesky VM, Mesjasz-Przybyłowicz J (1995) Investigation of Ni hyperaccumulation by true elemental imaging. Nucl Instrum Meth B 104:176–181. https://doi.org/10.1016/0168-583X(95)00445-9
Przybyłowicz WJ, Mesjasz-Przybyłowicz J, Prozesky VM, Pineda CA (1997) Botanical applications in nuclear microscopy. Nucl Instrum Meth B 130:335–345. https://doi.org/10.1016/S0168-583X(97)00224-3
Psaras GK, Manetas Y (2001) Nickel localization in seeds of the metal hyperaccumulator Thlaspi pindicum Hausskn. Ann Bot 88:513–516. https://doi.org/10.1006/anbo.2001.1470
Reeves RD (2003) Tropical hyperaccumulators of metals and their potential for phytoextraction. Plant Soil 249:57–65. https://doi.org/10.1023/A:1022572517197
Reeves RD, Baker AJM, Jaffré T, Erskine PD, Echevarria G, van der Ent A (2017) A global database for hyperaccumulator plants of metal and metalloid trace elements. New Phytol 218:407–411. https://doi.org/10.1111/nph.14907
Ryan CG (2000) Quantitative trace element imaging using PIXE and the nuclear microprobe. Int J Imag Syst Tech 11:219–230. https://doi.org/10.1002/ima.1007
Ryan CG, Jamieson DN (1993) Dynamic analysis: on-line quantitative PIXE microanalysis and its use in overlap-resolved elemental mapping. Nucl Instrum Meth B 77:203–214. https://doi.org/10.1016/0168-583X(93)95545-G
Ryan C, Cousens D, Sie S, Griffin W (1990) Quantitative analysis of PIXE spectra in geoscience applications. Nucl Instrum Meth B 49:271–276. https://doi.org/10.1016/0168-583X(90)90259-W
Ryan CG, Etschmann BE, Vogt S, Maser J, Harland CL, van Achterbergh E, Legnini D (2005) Nuclear microprobe-synchrotron synergy: towards integrated quantitative real-time elemental imaging using PIXE and SXRF. Nucl Instrum Meth B 231:183–188. https://doi.org/10.1016/j.nimb.2005.01.054
Ryan CG, Kirkham R, Hough RM, Moorhead G, Siddons DP, De Jonge MD, Paterson DJ, De Geronimo G, Howard DL, Cleverley JS (2010) Elemental X-ray imaging using the Maia detector array: The benefits and challenges of large solid-angle. Nucl Instrum Meth A 619:37–43. https://doi.org/10.1016/j.nima.2009.11.035
Sarret G, Smits EAHP, Michel HC, Isaure MP, Zhao FJ, Tappero R (2013) Chapter one - Use of synchrotron-based techniques to elucidate metal uptake and metabolism in plants. Adv Agron 119:1–82. https://doi.org/10.1016/b978-0-12-407247-3.00001-9
Siddons DP, Kirkham R, Ryan CG, De Geronimo G, Dragone A, Kuczewski AJ, Li ZY, Carini GA, Pinelli D, Beuttenmuller R, Elliott D, Pfeffer M, Tyson TA, Moorhead GF, Dunn PA (2014) Maia X-ray Microprobe Detector Array System. J Phys Conf Ser 499:012001–012010. https://doi.org/10.1088/1742-6596/499/1/012001
van der Ent A, Baker AJM, Reeves RD, Pollard AJ, Schat H (2013) Hyperaccumulators of metal and metalloid trace elements: facts and fiction. Plant Soil 362:319–334. https://doi.org/10.1007/s11104-012-1287-3
van der Ent A, Przybyłowicz WJ, de Jonge MD, Harris HH, Ryan CG, Tylko G, Paterson DJ, Barnabas AD, Kopittke PM, Mesjasz-Przybyłowicz J (2018) X-ray elemental mapping techniques for elucidating the ecophysiology of hyperaccumulator plants. New Phytol 218:432–452. https://doi.org/10.1111/nph.14810
van der Pas L, Ingle R (2019) Towards an understanding of the molecular basis of nickel hyperaccumulation in plants. Plants 8:11. https://doi.org/10.3390/plants8010011
Verbruggen N, Hermans C, Schat H (2009) Molecular mechanisms of metal hyperaccumulation in plants. New Phytol 181:759–776. https://doi.org/10.1111/j.1469-8137.2008.02748.x
Vogel-Mikuš K, Pongrac P, Kump P, Necêmer M, Simĉiĉ J, Peliconb P (2007) Localisation and quantification of elements within seeds of Cd/Zn hyperaccumulator Thlaspi praecox by micro-PIXE. Environ Pollut 147:50–59. https://doi.org/10.1016/j.envpol.2006.08.026
Weigend M, Mustafa A, Ensikat HJ (2018) Calcium phosphate in plant trichomes: the overlooked biomineral. Planta 247:277–285. https://doi.org/10.1007/s00425-017-2826-1
Zongo C, Fogliani B, Rabier J, Laffont-Schwob I, Saintpierre D, Bouraïma-Madjèbi S (2008) Nickel and calcium localisation in fruit and seed of the metal hyperaccumulator Geissois pruinosa versus the non-hyperaccumulator G. montana. Presented at the 6th International Conference on Serpentine Ecology at the College of the Atlantic, Bar Harbor, Maine (June 16–23, 2008)
Acknowledgements
This research was undertaken on the X-Ray Fluorescence Microscopy beamline of the Australian Synchrotron (part of ANSTO), Victoria, Australia. This work was supported by the Multi-modal Australian ScienceS Imaging and Visualisation Environment (MASSIVE). We thank Rachel Mak (University of Sydney) for help with the fitting of the µXRF data. The Centre for Microscopy and Microanalysis of The University of Queensland is acknowledged for support with the SEM-EDS analysis. A.L.D. Paul is the recipient of a UQ Graduate School Scholarship (UQGSS) from The University of Queensland. W.J. Przybyłowicz and J. Mesjasz-Przybyłowicz are recipients of the South African National Research Foundation incentive grants No 114693 and 114694, respectively.
Author information
Authors and Affiliations
Contributions
A.vdE., P.D.E., W.P., J.M.P. and H.H. conducted the synchrotron X-ray Fluorescence Spectroscopy (µXRF) experiment. A.vdE. conducted the SEM imaging and bulk elemental analysis. A.L.D.P., A.vdE., J.M.P., W.P., G.E., P.D.E., and H.H. wrote the manuscript.
Corresponding author
Additional information
Responsible Editor: Juan Barcelo
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 1230 kb)
Rights and permissions
About this article
Cite this article
Paul, A.L.D., Harris, H.H., Erskine, P.D. et al. Synchrotron µXRF imaging of live seedlings of Berkheya coddii and Odontarrhena muralis during germination and seedling growth. Plant Soil 453, 487–501 (2020). https://doi.org/10.1007/s11104-020-04591-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-020-04591-4