Abstract
Aims
To identify the rhizobia nodulating Astragalus sinicus in the acid soils at Xinyang, China and to estimate the effects of fertilization strategies on the rhizobial community.
Methods
Soil samples were collected from treatments submitted to different fertilization strategies (chemical fertilizers, A. sinicus green manure, various input levels). Nodules obtained from the A. sinicus trapping plants grown in the soil samples were used for rhizobial isolation. The isolates were grouped into PCR-RFLP genotypes based on analyses of 16S rRNA gene and IGS, and representatives for each genotype were further subjected to phylogenetic analyses of housekeeping genes (16S rRNA gene, atpD, glnII, and rpoB), as well as symbiotic genes (nodC and nifH). Soil physicochemical characteristics were also estimated and redundancy analysis was performed to examine the relations between soil features and distribution of rhizobial genotypes across different fertilization treatments.
Results
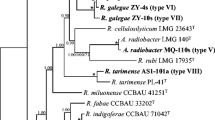
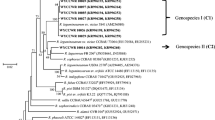
A total of 257 rhizobial isolates were obtained and discriminated into 12 IGS types in a single 16S rRNA type by RFLP analyses. Further 16S rRNA gene sequence-based phylogeny and multilocus sequence analysis (MLSA) distinguished a minor group (10 strains, 4 IGS types) affiliated to Mesorhizobium huakuii and a main group (247 strains, 8 IGS types) to Mesorhizobium jarvisii, for which a novel symbiovar, sv. astragali, was described. The long-term input of A. sinicus green manure increased rhizobial diversity and modified rhizobial community structures, via the increase of organic matter and decrease of soil pH, available phosphate, available potassium, and total salt.
Conclusions
M. jarvisii sv. nov. astragali was the predominant microsymbiont of A. sinicus in the tested acidic soils. The long-term input of green manure modified the soil features, enhancing the genetic diversity of rhizobia associated with A. sinicus and alternating the rhizobial community structure.



Similar content being viewed by others
References
Braak C, Smilauer P (2002) CANOCO reference manual and CanoDraw for Windows user’s guide: sofware for canonical community ordination (version 4.5). Section on Permutation Methods Microcomputer Power, Ithaca, Newyork
Chen DM, Yuan L, Liu YR, Ji JH, Hou HQ (2017) Long-term application of manures plus chemical fertilizers sustained high rice yield and improved soil chemical and bacterial properties. Eur J Agron 90:34–42. https://doi.org/10.1016/j.eja.2017.07.007
Chen WC, Wang KR, Xie XL (2009) Effects on distributions of carbon and nitrogen in a reddish paddy soil under long-term different fertilization treatments. Chin J Soil Sci
Chen WX, Li GS, QI YL, Wang ET, Yuan HL, Li JL (1991) Rhizobium huakuii sp. nov. isolated from the root nodules of Astragalus sinicus. Int J Syst Bacteriol 41:275–280
Duodu S, Carlsson G, Huss-Danell K, Svenning MM (2007) Large genotypic variation but small variation in N2 fixation among rhizobia nodulating red clover in soils of northern Scandinavia. J Appl Microbiol 102:1625–1635
Duodu S, Nsiah EK, Bhuvaneswari TV, Svenning MM (2006) Genetic diversity of a natural population of Rhizobium leguminosarum biovar trifolii analysed from field nodules and by a plant infection technique. Soil Biol Biochem 38:1162–1165. https://doi.org/10.1016/j.soilbio.2005.07.015
Fang Y, Yan ZL, Chen JC, Wang F, Wang MK, Lin XJ (2015) Effect of chemical fertilization and green manure on the abundance and community structure of ammonia oxidizers in a paddy soil. Chil J Agric Res 75:488–496. https://doi.org/10.4067/s0718-58392015000500015
Gao SJ, Cao WD, Gao JS, Huang J, Bai JS, Zeng NH, Chang DN, Shimizu K (2017) Effects of long-term application of different green manures on ferric iron reduction in a red paddy soil in Southern China. J Integr Agric 16:959–966. https://doi.org/10.1016/s2095-3119(16)61509-5
Guo XW, Zhang XX, Zhang ZM, Li FD (1999) Characterization of Astragalus sinicus rhizobia by restriction fragment length polymorphism analysis of chromosomal and nodulation genes regions. Curr Microbiol 39:358–0364
Han L, Wang E, Han T, Liu J, Sui X, Chen W, Chen W (2009) Unique community structure and biogeography of soybean rhizobia in the saline-alkaline soils of Xinjiang, China. Plant Soil 324:291–305
Harrison S, Young JPW, Jones DG (1987) Rhizobium population genetics: effect of clover variety and inoculum dilution on the genetic diversity sampled from natural populations. Plant Soil 103:147–150
Ji ZJ, Yan H, Cui QG, Wang ET, Chen WX, Chen WF (2015) Genetic divergence and gene flow among Mesorhizobium strains nodulating the shrub legume Caragana. Syst Appl Microbiol 38:176–183. https://doi.org/10.1016/j.syapm.2015.02.007
Leps J, Smilauer P (2003) Multivariate analysis of ecological data using CANOCO. Cambridge University Press
Lin DH, Gu RS (1998) Chinese milk vetch. Fujian Science and Technology Publishing House
Martens M, Dawyndt P, Coopman R, Gillis M, De Vos P, Willems A (2008) Advantages of multilocus sequence analysis for taxonomic studies: a case study using 10 housekeeping genes in the genus Ensifer (including former Sinorhizobium). Int J Syst Evol Microbiol 58:200–214
Martinez-Hidalgo P, Ramirez-Bahena MH, Flores-Felix JD, Rivas R, Igual JM, Mateos PF, Martinez-Molina E, Leon-Barrios M, Peix A, Velazquez E (2015) Revision of the taxonomic status of type strains of Mesorhizobium loti and reclassification of strain USDA 3471T as the type strain of Mesorhizobium erdmanii sp. nov. and ATCC 33669T as the type strain of Mesorhizobium jarvisii sp. nov. Int J Syst Evol Microbiol 65:1703–1708. https://doi.org/10.1099/ijs.0.000164
Murooka Y, Xu Y, Sanada K, Araki M, Morinaga T, Yokota A (1993) Formation of root nodules by Rhizobium huakuii biovar renge, bv. nov. on Astragalus sinicus cv. Japan. J Ferment Bioeng 76:38–44
Nuswantara S, Fujie M, Yamada T, Malek W, Inaba M, Kaneko Y, Murooka Y (1999) Phylogenetic position of Mesorhizobium huakuii subsp. rengei, a symbiont of Astragalus sinicus cv. Japan. J Biosci Bioeng 87:49–55
Odair A, Glaciela K, Mariangela H (2006) Sampling effects on the assessment of genetic diversity of rhizobia associated with soybean and common bean. Soil Biol Biochem 38:1298–1307
Sullivan JT, Eardly BD, van Berkum P, Ronson CW (1996) Four unnamed species of nonsymbiotic rhizobia isolated from the rhizosphere of Lotus corniculatus. Appl Environ Microbiol 62:2818–2825
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729. https://doi.org/10.1093/molbev/msr121
Tan Z, Xu X, Wang E, Gao J, Martinez-Romero E, Chen W (1997) Phylogenetic and genetic relationships of Mesorhizobium tianshanense and related rhizobia. Int J Syst Evol Microbiol 47:874
Tena W, Wolde-Meskel E, Degefu T, Walley F (2017) Genetic and phenotypic diversity of rhizobia nodulating chickpea (Cicer arietinum L.) in soils from southern and central Ethiopia. Can J Microbiol 63:690–707. https://doi.org/10.1139/cjm-2016-0776
Terefework Z, Kaijalainen S, Lindstrom K (2001) AFLP fingerprinting as a tool to study the genetic diversity of Rhizobium galegae isolated from Galega orientalis and Galega officinalis. J Biotechnol 91:169–180
Van Cauwenberghe J, Lemaire B, Stefan A, Efrose R, Michiels J, Honnay O (2016) Symbiont abundance is more important than pre-infection partner choice in a Rhizobium-legume mutualism. Syst Appl Microbiol 39:345–349
Vauterin LaV P (1992) Computer-aided objective comparison of electrophoresis patterns for grouping and identification of microorganisms. Eur Microbiol 1:37–41
Vincent JM (1970) A manual for the practical study of rootnodule bacteria. International Biological Programme, Blackwell Scientific, Oxford
Vinuesa P, Silva C, Lorite MJ, Izaguirre-Mayoral ML, Bedmar EJ, Martinez-Romero E (2005a) Molecular systematics of rhizobia based on maximum likelihood and Bayesian phylogenies inferred from rrs, atpD, recA and nifH sequences, and their use in the classification of Sesbania microsymbionts from Venezuelan wetlands. Syst Appl Microbiol 28:702–716. https://doi.org/10.1016/j.syapm.2005.05.007
Vinuesa P, Silva C, Werner D, Martínez-Romero E (2005b) Population genetics and phylogenetic inference in bacterial molecular systematics: the roles of migration and recombination in Bradyrhizobium species cohesion and delineation. Mol Phylogenet Evol 34:29–54
Wang ET, Van Berkum P, Sui XH, Beyene D, Chen WX, Martínez-Romero E (1999) Diversity of rhizobia associated with Amorpha fruticosa isolated from Chinese soils and description of Mesorhizobium amorphae sp. nov. Int J Syst Bacteriol 49:51–65
Yan J, Han XZ, Ji ZJ, Li Y, Wang ET, Xie ZH, Chen WF (2014) Abundance and diversity of soybean-nodulating rhizobia in black soil are impacted by land use and crop management. Appl Environ Microbiol 80:5394–5402
Zhang JJ, Lou K, Jin X, Mao PH, Wang ET, Tian CF, Sui XH, Chen WF, Chen WX (2012) Distinctive Mesorhizobium populations associated with Cicer arietinum L. in alkaline soils of Xinjiang, China. Plant Soil 353:123–134. https://doi.org/10.1007/s11104-011-1014-5
Zhang JJ, Yang X, Guo C, de Lajudie P, Singh RP, Wang ET, Chen WF (2017) Mesorhizobium muleiense and Mesorhizobium gsp. nov. are symbionts of Cicer arietinum L. in alkaline soils of Gansu, Northwest China. Plant Soil 410:103–112. https://doi.org/10.1007/s11104-016-2987-x
Zhang JJ, Yu T, Lou K, Mao PH, Wang ET, Chen WF, Chen WX (2014) Genotypic alteration and competitive nodulation of Mesorhizobium muleiense against exotic chickpea rhizobia in alkaline soils. Syst Appl Microbiol 37:520–524. https://doi.org/10.1016/j.syapm.2014.07.004
Zhang XX, Turner SL, Guo XW, Yang HJ, Debelle F, Yang GP, Denarie J, Young JP, Li FD (2000) The common nodulation genes of Astragalus sinicus rhizobia are conserved despite chromosomal diversity. Appl Environ Microbiol 66:2988–2995
Zhang YM, Li Y, Chen WF, Wang ET, Tian CF, Li QQ, Zhang YZ, Sui XH, Chen WX (2011) Biodiversity and biogeography of rhizobia associated with soybean plants grown in the north China plain. Appl Environ Microbiol 77:6331–6342. https://doi.org/10.1128/aem.00542-11
Zheng WT, Li Y, Wang R, Sui XH, Zhang XX, Zhang JJ, Wang ET, Chen WX (2013) Mesorhizobium qingshengii sp nov., isolated from effective nodules of Astragalus sinicus. Int J Syst Evol Microbiol 63:2002–2007. https://doi.org/10.1099/ijs.0.044362-0
Funding
This work was financed by the China Agriculture Research System—Green Manure (Project No. CARS-22), the National Natural Science Foundation of China (Project No. 31400008 and 31571778), National Human and Social Security Ministry of Science and Technology for Overseas Returnees (Project No. 21104000005). ETW is financially supported by grants SIP20171259 authorized by Instituto Politécnico Nacional, Mexico.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Responsible Editor: Katharina Pawlowski.
Rights and permissions
About this article
Cite this article
Zhang, J., Shang, Y., Wang, E. et al. Mesorhizobium jarvisii sv. astragali as predominant microsymbiont for Astragalus sinicus L. in acidic soils, Xinyang, China. Plant Soil 433, 201–212 (2018). https://doi.org/10.1007/s11104-018-3830-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-018-3830-3