Abstract
Aims
Our aim was to explore the way that root system type affects mycorrhizal growth response of plants.
Methods
An extensive meta-analysis with 943 peer-review publications was conducted to test the difference in mycorrhizal responses between taproot plants and plants with a fibrous root system.
Results
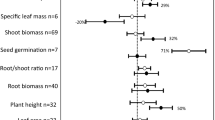
We found that taproot plants showed greater growth response (biomass, P and N uptake) to colonization by arbuscular mycorrhizal fungi (AMF) than do plant species with fibrous root systems. This response pattern was dependent on stress types, AMF identity and species richness, and particularly the type of stress (abiotic vs. biotic). Taproot plants respond more to AMF than plants with a fibrous root system; but no difference was shown under biotic stress. The interaction effect seen for AMF and biotic stress was significantly higher for plants with fibrous root system, but was not significant between taproot plants and abiotic stress. Difference in biomass response was only found for Glomeraceae and Gigasporaceae between the two types of plants, while difference was found in P uptake response for Glomeraceae and Claroideoglomeraceae. However, plants with fibrous root system showed higher growth response than taproot plants under nematode stress.
Conclusions
Taproot plants might be more dependent on mycorrhiza than plants with fibrous root system. This indicates that environmental conditions can modify the relative abundance of taproot plants and plants with fibrous root system through mycorrhizal functioning, which will regulate plant community dynamics and processes.




Similar content being viewed by others
References
Atkinson D, Berta G, Hooker JE (1994) Impact of mycorrhizal colonisation on root architecture, root longevity and the formation of growth regulators. Impact of arbuscular mycorrhizas on sustainable agriculture and natural ecosystems. Springer, pp. 89–99.
Azcón R, Rubio R, Barea JM (1991) Selective interactions between different species of mycorrhizal fungi and rhizobium-meliloti strains, and their effects on growth, N2-fixation (N15) and nutrition of Medicago sativa L. New Phytol 117:399–404
Azcón-Aguilar C, Barea JM (1997) Arbuscular mycorrhizas and biological control of soil-born plant pathogens- An overview of the mechanisms involved. Mycorrhiza 6:457–464
Barto EK, Rillig MC (2010) Does herbivory really suppress mycorrhiza? A meta-analysis. J Ecol 98:745–753
Bates T, Lynch J (2001) Root hairs confer a competitive advantage under low phosphorus availability. Plant Soil 236:243–250
Baylis GTS (1970) Root hairs and phycomycetous mycorrhizas in phosphorus- deficient soil. Plant Soil 33:713–716
Berta G, Fusconi A, Trotta A, Scannerini S (1990) Morphogenetic modifications induced by the mycorrhizal fungus Glomus strain E3 in the root system of Allium porrum L. New Phytol 114:207–215
Berta G, Fusconi A, Trotta A (1993) VA mycorrhizal infection and the morphology and function of root systems. Environ Exp Bot 33:159–173
Berta G, Trotta A, Fusconi A, Hooker JE, Munro M, Atkinson D, Gianinazzi S (1995) Arbuscular mycorrhizal induced changes to plant growth and root system morphology in Prunus cerasifera. Tree Physiol 15:281–293
Borowicz VA (2001) Do arbuscular mycorrhizal fungi alter plant-pathogen relations? Ecology 82:3057–3068
Cavagnaro TR, Smith FA, Smith SE, Jakobsen I (2005) Functional diversity in arbuscular mycorrhizas, exploitation of soil patches with different phosphate enrichment differs among fungal species. Plant Cell Environ 28:642–650
Grace EJ, Cotsaftis O, Tester M, Smith FA, Smith SE (2009) Arbuscular mycorrhizal inhibition of growth in barley cannot be attributed to extent of colonization, fungal phosphorus uptake or effects on expression of plant phosphate transporter genes. New Phytol 181:938–949
Gurevitch J, Hedges LV (1999) Statistical issues in ecological meta-analyses. Ecology 80:1142–1149
Hasanuzzaman M, Nahar K, Alam MM, Fujita M (2014) Modulation of antioxidant machinery and the methylglyoxal detoxification system in selenium- supplemented brassica napus seedlings confers tolerance to high temperature stress. Biol Trace Elem Res 161:297–307
Hedges LV, Olkin I (1985) Statistical methods for meta-Analysis. Accedemic press, New York
Heijne B, Vandam D, Heil GW, Bobbink R (1996) Acidification effects on vesicular-arbuscular mycorrhizal (VAM) infection, growth and nutrient uptake of established heathland herb species. Plant Soil 179:197–206
Hetrick BAD, Kitt DG, Wilson GT (1988) Mycorrhizal dependence and growth habit of warm-season and cool-season tallgrass prairie plants. Can J Bot 66:1376–1380
Hetrick B, Wilson GWT, Todd TC (1992) Relationships of mycorrhizal symbiosis, rooting strategy, and phenology among tallgrass prairie forbs. Can J Bot 70:1521–1528
Hoeksema JD, Chaudhary VB, Gehring CA, Johnson NC, Karst J, Koide RT, Pringle A, Zabinski C, Bever JD, Moore JC, Wilson GWT, Klironomos JN, Umbanhowar J (2010) A meta-analysis of context-dependency in plant response to inoculation with mycorrhizal fungi. Ecol Lett 13:394–407
Klironomos JN (2003) Variation in plant response to native and exotic arbuscular mycorrhizal fungi. Ecology 84:2292–2301
Koide RT (2000) Functional complementarity in the arbuscular mycorrhizal symbiosis. New Phytol 147:233–235
Koide R, Dickie I (2002) Effects of mycorrhizal fungi on plant populations. Plant Soil 244:307–317
Koricheva J, Gange AC, Jones T (2009) Effects of mycorrhizal fungi on insect herbivores: a meta-analysis. Ecology 90:2088–2097
Larkin MA, Blackshields G, Brown NP, Chenna R, McGettigan PA, McWilliam H, Valentin F, Wallace IM, Wilm A, Lopez R, Thompson JD, Gibson TJ, Higgins DG (2007) Clustal W and Clustal X version 2.0. Bioinformatics 23:2947–2948
Li H, Smith SE, Holloway RE, Zhu Y, Smith FA (2006) Arbuscular mycorrhizal fungi contribute to phosphorus uptake by wheat grown in a phosphorus-fixing soil even in the absence of positive growth responses. New Phytol 172:536–543
Maherali H (2014) Is there an association between root architecture and mycorrhizal growth response? New Phytol 204:192–200
Maherali H, Klironomos JN (2007) Influence of phylogeny on fungal community assembly and ecosystem functioning. Science 316:1746–1748
Mardukhi B, Rejali F, Daei G, Ardakani MR, Malakouti MJ, Miransari M (2011) Arbuscular mycorrhizas enhance nutrient uptake in different wheat genotypes at high salinity levels under field and greenhouse conditions. C R Biol 334:564–571
Morris WF, Hufbauer RA, Agrawal AA, Bever JD, Borowicz VA, Gilbert GS, Maron JL, Mitchell CE, Parker IM, Power AG, Torchin ME, Vázquez DP (2007) Direct and interactive effects of enemies and mutualists on plant performance: a meta-analysis. Ecology 88:1021–1029
Ohsowski BM, Zaitsoff PD, Öpik M, Hart MM (2014) Where the wild things are: looking for uncultured Glomeromycota. New Phytol 204:171–179
Öpik M, Vanatoa A, Vanatoa E, Moora M, Davison MJ, Kalwij JM, Reier Ü, Zobel M (2010) The online database MaarjAM reveals global and ecosystemic distribution patterns in arbuscular mycorrhizal fungi (Glomeromycota). New Phytol 188:223–241
Peters JL, Sutton AJ, Jones DR, Abrams KR, Rushton L (2006) Comparison of two methods to detect publication bias in meta-analysis. JAMA 295:676–680
Rosenberg MS, Adams DC, Gurevitch J (2000) MetaWin: statistical software for meta-analysis, version 2.0. Sinauer Associated, Sunderland
Schellenbaum L, Berta G, Ravolanirina F, Tisserant B, Gianinazzi S, Fitter AH (1991) Influence of endomycorrhizal infection on root morphology in a micropropagated woody plant species (Vitis vinifera L.). Ann Bot-Lond 68:135–141
Schloss PD, Westcott SL, Ryabin T, Hall JR, Hartmann M, Hollister EB, Weber CF (2009) Introducing mothur: open-source, platform-independent, community- supported software for describing and comparing microbial communities. Appl Environ Microbiol 75:7537–7541
Schweiger P, Robson AD, Barrow NJ (1995) Root hair length determines beneficial effect of a Glomus species on shoot growth of some pasture species. New Phytol 131:247–254
Sheng M, Tang M, Chen H, Yang B, Zhang F, Huang Y (2008) Influence of arbuscular mycorrhizae on photosynthesis and water status of maize plants under salt stress. Mycorrhiza 18:287–296
Sikes BA, CottenieK KJN (2009) Plant and fungal identity determines pathogen protection of plant roots by arbuscular mycorrhizas. J Ecol 97:1274–1280
Smith SE, Read DJ (2008) Mycorrhizal symbiosis. Academic press Inc, San Diego
Sullivan WM, Jiang ZC, Hull RJ (2000) Root morphology and its relationship with nitrate uptake in Kentucky bluegrass. Crop Sci 40:765–772
Torrey JG, Clarkson DT (1975) The development and function of roots. Academic, New York, p 618
van der Heijden MGA, Klironomos JN, Ursic M, Moutoglis P, Streitwolf-Engel R, Boller T, Wiemken A, Sander IR (1998) Mycorrhizal fungal diversity determines plant biodiversity, ecosystem variability and productivity. Nature 396:69–72
van der Heijden MGA, Wiemken A, Sanders IR (2003) Different arbuscular mycorrhizal fungi alter coexistence and resource distribution between co-occurring plant. New Phytol 157:569–578
van Groenigen KJ, Osenberg CW, Hungate BA (2011) Increased soil emissions of potent greenhouse gases under increased atmospheric CO2. Nature 475:214–216
Veresoglou SD, Menexes G, Rillig MC (2012) Do arbuscular mycorrhizal fungi affect the allometric partition of host plant biomass to shoots and roots? A meta-analysis of studies from 1990 to 2010. Mycorrhiza 22:227–235
Veresoglou SD, Powell JR, Davison J, Lekberg Y, Rillig MC (2014) The Leinster and Cobbold indices improve inferences about microbial diversity. Fungal Ecol 11:1–7
Vilà M, Espinar JL, Hejda M, Hulme PE, Jarošík V, Maron JL, Pergl J, Schaffner U, Sun Y, Pyšek P (2011) Ecological impacts of invasive alien plants: a meta-analysis of their effects on species, communities and ecosystems. Ecol Lett 14:702–708
Wagg C, Jansa J, Stadler M, Schmid B, van der Heijden MGA (2011) Mycorrhizal fungal identity and diversity relaxes plant-plant competition. Ecology 92:1303–1313
Wilson GWT, Hartnett DC (1998) Interspecific variation in plant responses to mycorrhizal colonization in tallgrass prairie. Am J Bot 85:1732–1738
Wu QS (2011) Mycorrhizal efficacy of trifoliate orange seedlings on alleviating temperature stress. Plant Soil Environ 57:459–464
Wu QS, Zou YN, Xia RX, Wang MY (2007) Five Glomus species affect water relations of Citrus tangerine during drought stress. Bot Stud 48:147–154
Yang H, Zang Y, Yuan Y, Tang J, Chen X (2012) Selectivity by host plants affects the distribution of arbuscular mycorrhizal fungi: evidence from ITS rDNA sequence metadata. BMC Evol Biol 12:50
Ye CX (2007) Botany. Higher Education Press, Beijing, p 89
Zhang GL, Zhang ST, Xiao LT, Wu XJ, Xiao YH, Chen LY (2013) Effects of high temperature stress on physiological characteristics of anther and pollen traits of rice at flowering stage. Acta Agron Sin 39:177–183
Acknowledgments
This study was funded by the Natural Science Foundation of China (No. 31400373), Natural Science Foundation of Jiangsu Province (No. BK20140689) and China Postdoctoral Science Foundation (No. 2014 M561659).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Responsible Editor: Erik J. Joner.
Rights and permissions
About this article
Cite this article
Yang, H., Zhang, Q., Dai, Y. et al. Effects of arbuscular mycorrhizal fungi on plant growth depend on root system: a meta-analysis. Plant Soil 389, 361–374 (2015). https://doi.org/10.1007/s11104-014-2370-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-014-2370-8