Abstract
Aims
Extracellular enzymes mediate the decomposition of organic matter and the release of plant-available nutrients. Current theory predicts that enzyme production by soil microbes is regulated by the stoichiometric demands of microbial biomass and the complexity of environmental resources, but most experiments ignore the potential effect of alleviated carbon limitation in the rhizosphere. Our objective was to investigate linkages between enzyme activities, soil nutrient availability and plant roots in a tropical Oxisol.
Methods
We conducted a greenhouse experiment using soils from the Luquillo Experimental Forest and seedlings of Tabebuia heterophylla. Planted and unplanted pots were fertilized with different combinations of phosphorus and either mineral nitrogen (ammonia chloride) or a nitrogen-rich organic compound (casein). We measured changes in plant and soil nutrients and five extracellular enzyme activities.
Results
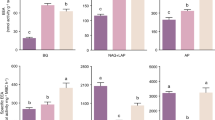
Phosphatase activity declined by 28% in the P and 40% in the complex nitrogen treatment, while N-acetyl glucosaminidase increased 162% in the complex nitrogen treatment. Beta-glucosidase, beta-xylosidase, cellobiohydrolase and N-acetyl glucosaminidase all increased significantly over time in the simple nitrogen treatment (P < 0.05).
Conclusions
Enzymatic responses support microbial resource allocation theory, that is, the concept that soil microbes regulate enzyme production based on scarcity of resources. However, we did not observe any additional effect of roots on extracellular enzyme activities. Enzymatic C:N, C:P and N:P ratios further support the notion that shifts in microbial stoichiometric demand drive responses to nutrients.





Similar content being viewed by others
References
Allison SD, Martiny JBH (2008) Resistance, resilience, and redundancy in microbial communities. Proc Natl Acad Sci U S A 105:11512–11519. doi:10.1073/pnas.0801925105
Allison SD, Vitousek PM (2005) Responses of extracellular enzymes to simple and complex nutrient inputs. Soil Biol Biochem 37:937–944. doi:10.1016/j.soilbio.2004.09.014
Allison SD, Hanson CA, Treseder KK (2007) Nitrogen fertilization reduces diversity and alters community structure of active fungi in boreal ecosystems. Soil Biol Biochem 39:1878–1887. doi:10.1016/j.soilbio.2007.02.001
Allison SD, Weintraub MN, Gartner TB, Waldrop MP (2011) Evolutionary-economic principles as regulators of soil enzyme production and ecosystem function. In: Shukla GC, Varma A (eds) Soil Enzymology. Springer, Berlin Heidelberg
Anderson JM, Swift MJ (1983) Decomposition in tropical forests. In: Sutton SL, Whitmore TC, Chadwick AC (eds) Tropical rain forest: ecology and management. Blackwell Science, Oxford
Averill C, Finzi A (2011) Plant regulation of microbial enzyme production in situ. Soil Biol Biochem 43:2457–2460. doi:10.1016/j.soilbio.2011.09.002
Brookshire ENJ, Gerber S, Menge DNL, Hedin LO (2012) Large losses of inorganic nitrogen from tropical rainforests suggest a lack of nitrogen limitation. Ecol Lett 15:9–16. doi:10.1111/j.1461-0248.2011.01701.x
Brown ALP, Garland JL, Day FP (2009) Physiological profiling of soil microbial communities in a Florida scrub-oak ecosystem: spatial distribution and nutrient limitations. Microb Ecol 57:14–24. doi:10.1007/s00248-008-9461-0
Carreiro MM, Sinsabaugh RL, Repert DA, Parkhurst DF (2000) Microbial enzyme shifts explain litter decay responses to simulated nitrogen deposition. Ecology 81:2359–2365. doi:10.2307/177459
Clarholm M (1993) Microbial biomass-P, labile-P and acid-phosphatase activity in the humus layer of a spruce forest, after repeated additions of fertilizers. Biol Fertil Soils 16:287–292. doi:10.1007/bf00369306
Cleveland CC, Liptzin D (2007) C : N : P stoichiometry in soil: is there a "Redfield ratio" for the microbial biomass? Biogeochemistry 85:235–252. doi:10.1007/s10533-007-9132-0
Cleveland CC, Townsend AR (2006) Nutrient additions to a tropical rain forest drive substantial soil carbon dioxide losses to the atmosphere. Proc Natl Acad Sci U S A 103:10316–10321. doi:10.1073/pnas.0600989103
Compton JE, Watrud LS, Porteous LA, DeGrood S (2004) Response of soil microbial biomass and community composition to chronic nitrogen additions at Harvard forest. Forest Ecol Manage 196:143–158. doi:10.1016/j.foreco.2004.03.017
Cusack DF, Silver WL, Torn MS, Burton SD, Firestone MK (2011) Changes in microbial community characteristics and soil organic matter with nitrogen additions in two tropical forests. Ecology 92:621–632. doi:10.1890/10-0459.1
Drake JE, Darby BA, Giasson MA, Kramer MA, Phillips RP, Finzi AC (2013) Stoichiometry constrains microbial response to root exudation-insights from a model and a field experiment in a temperate forest. Biogeosciences 10:821–838. doi:10.5194/bg-10-821-2013
Fierer N, Jackson RB (2006) The diversity and biogeography of soil bacterial communities. Proc Natl Acad Sci U S A 103:626–631. doi:10.1073/pnas.0507535103
Fierer N, Breitbart M, Nulton J, Salamon P, Lozupone C, Jones R, Robeson M, Edwards RA, Felts B, Rayhawk S, Knight R, Rohwer F, Jackson RB (2007) Metagenomic and small-subunit rRNA analyses reveal the genetic diversity of bacteria, archaea, fungi, and viruses in soil. Appl Environ Microbiol 73:7059–7066. doi:10.1128/aem.00358-07
Fog K (1988) The effect of added nitrogen on the rate of decomposition of organic matter. Biol Rev Camb Philos Soc 63:433–462. doi:10.1111/j.1469-185X.1988.tb00725.x
Frey SD, Knorr M, Parrent JL, Simpson RT (2004) Chronic nitrogen enrichment affects the structure and function of the soil microbial community in temperate hardwood and pine forests. Forest Ecol Manage 196:159–171. doi:10.1016/j.foreco.2004.03.018
Galloway JN, Dentener FJ, Capone DG, Boyer EW, Howarth RW, Seitzinger SP, Asner GP, Cleveland CC, Green PA, Holland EA, Karl DM, Michaels AF, Porter JH, Townsend AR, Vorosmarty CJ (2004) Nitrogen cycles: past, present, and future. Biogeochemistry 70:153–226. doi:10.1007/s10533-004-0370-0
German DP, Weintraub MN, Grandy AS, Lauber CL, Rinkes ZL, Allison SD (2011) Optimization of hydrolytic and oxidative enzyme methods for ecosystem studies. Soil Biol Biochem 43:1387–1397. doi:10.1016/j.soilbio.2011.03.017
Giardina CP, Ryan MG, Binkley D, Fownes JH (2003) Primary production and carbon allocation in relation to nutrient supply in a tropical experimental forest. Glob Chang Biol 9:1438–1450. doi:10.1046/j.1365-2486.2003.00558.x
Henry HAL, Juarez JD, Field CB, Vitousek PM (2005) Interactive effects of elevated CO2, N deposition and climate change on extracellular enzyme activity and soil density fractionation in a California annual grassland. Glob Chang Biol 11:1808–1815. doi:10.1111/j.1365-2486.2005.001007.x
Jolles P, Muzzarelli RAA (1999) Preface. In: Jolles P, Muzzarelli RAA (eds) Chitin and Chitinases. Birkhauser Verlag, Basel
Krashevska V, Maraun M, Ruess L, Scheu S (2010) Carbon and nutrient limitation of soil microorganisms and microbial grazers in a tropical montane rain forest. Oikos 119:1020–1028. doi:10.1111/j.1600-0706.2009.18169.x
Kuzyakov Y (2002) Review: factors affecting rhizosphere priming effects. J Plant Nutr Soil Sci Zeitschrift Fur Pflanzenernahrung Und Bodenkunde 165:382–396, doi:10.1002/1522-2624(200208)165:4<382::aid-jpln382>3.0.co;2-#
Nemergut DR, Townsend AR, Sattin SR, Freeman KR, Fierer N, Neff JC, Bowman WD, Schadt CW, Weintraub MN, Schmidt SK (2008) The effects of chronic nitrogen fertilization on alpine tundra soil microbial communities: implications for carbon and nitrogen cycling. Environ Microbiol 10:3093–3105. doi:10.1111/j.1462-2920.2008.01735.x
Olander LP, Vitousek PM (2000) Regulation of soil phosphatase and chitinase activity by N and P availability. Biogeochemistry 49:175–190. doi:10.1023/a:1006316117817
Phillips RP, Erlitz Y, Bier R, Bernhardt ES (2008) New approach for capturing soluble root exudates in forest soils. Funct Ecol 22:990–999. doi:10.1111/j.1365-2435.2008.01495.x
Priest FG (1977) Extracellular enzyme synthesis in the genus Bacillus. Bacteriol Rev 41:711–753
Rygiewicz PT, Andersen CP (1994) Mycorrhizae alter quality and quantity of carbon allocated below ground. Nature 369:58–60. doi:10.1038/369058a0
Saiya-Cork KR, Sinsabaugh RL, Zak DR (2002) The effects of long term nitrogen deposition on extracellular enzyme activity in an Acer saccharum forest soil. Soil Biol Biochem 34:1309–1315. doi:10.1016/s0038-0717(02)00074-3
Scatena FN, Lugo AE (1995) Geomorphology, disturbance, and the soil and vegetation of 2 subtropical wet steeplant watersheds of Puerto-Rico. Geomorphology 13:199–213. doi:10.1016/0169-555x(95)00021-v
Schimel JP, Weintraub MN (2003) The implications of exoenzyme activity on microbial carbon and nitrogen limitation in soil: a theoretical model. Soil Biol Biochem 35:549–563. doi:10.1016/s0038-0717(03)00015-4
Sinsabaugh RL, Moorhead DL (1994) Resource-allocation to extracellular enzyme production- A model for nitrogen and phosphorus control of litter decomposition. Soil Biol Biochem 26:1305–1311. doi:10.1016/0038-0717(94)90211-9
Sinsabaugh RL, Gallo ME, Lauber C, Waldrop MP, Zak DR (2005) Extracellular enzyme activities and soil organic matter dynamics for northern hardwood forests receiving simulated nitrogen deposition. Biogeochemistry 75:201–215. doi:10.1007/s10533-004-7112-1
Sinsabaugh RL, Lauber CL, Weintraub MN, Ahmed B, Allison SD, Crenshaw C, Contosta AR, Cusack D, Frey S, Gallo ME, Gartner TB, Hobbie SE, Holland K, Keeler BL, Powers JS, Stursova M, Takacs-Vesbach C, Waldrop MP, Wallenstein MD, Zak DR, Zeglin LH (2008) Stoichiometry of soil enzyme activity at global scale. Ecol Lett 11:1252–1264. doi:10.1111/j.1461-0248.2008.01245.x
Sinsabaugh RL, Hill BH, Shah JJF (2009) Ecoenzymatic stoichiometry of microbial organic nutrient acquisition in soil and sediment. Nature 462:795–U117. doi:10.1038/nature08632
Soil Survey Staff, Natural Resources Conservation Service, United States Department of Agriculture (2012) Official Soil Series Descriptions. http://soils.usda.gov/technical/classification/osd/index.htm. Accessed 1 Jul 2012
Stals I, Samyn B, Sergeant K, White T, Hoorelbeke K, Coorevits A, Devreese B, Claeyssens M, Piens K (2010) Identification of a gene coding fora deglycosylating enzyme in Hypocrea jecorina. FEMS Microbiol Lett 303:9–17. doi:10.1111/j.1574-6968.2009.01849.x
Templer PH, Silver WL, Pett-Ridge J, DeAngelis KM, Firestone MK (2008) Plant and microbial controls on nitrogen retention and loss in a humid tropical forest. Ecology 89:3030–3040. doi:10.1890/07-1631.1
Treseder KK (2008) Nitrogen additions and microbial biomass: a meta-analysis of ecosystem studies. Ecol Lett 11:1111–1120. doi:10.1111/j.1461-0248.2008.01230.x
Vance ED, Chapin FS (2001) Substrate limitations to microbial activity in taiga forest floors. Soil Biol Biochem 33:173–188. doi:10.1016/s0038-0717(00)00127-9
Vitousek PM, Sanford RL (1986) Nutrient cycling in moist tropical forest. Annu Rev Ecol Evol Syst 17:137–167. doi:10.1146/annurev.ecolsys.17.1.137
Vitousek PM, Porder S, Houlton BZ, Chadwick OA (2010) Terrestrial phosphorus limitation: mechanisms, implications, and nitrogen-phosphorus interactions. Ecol Appl 20:5–15. doi:10.1890/08-0127.1
Waldrop MP, Zak DR, Sinsabaugh RL (2004) Microbial community response to nitrogen deposition in northern forest ecosystems. Soil Biol Biochem 36:1443–1451. doi:10.1016/j.soilbio.2004.04.023
Wallenstein MD, Burns RG (2011) Ecology of Extracellular Enzyme Activities and Organic Matter Degradation in Soil: A Complex Community-Driven Process. In: Dick RP (ed) Methods of Soil Enzymology. Soil Science Society of America, Inc, Madison
Wallenstein MD, Weintraub MN (2008) Emerging tools for measuring and modeling the in situ activity of soil extracellular enzymes. Soil Biol Biochem 40:2098–2106. doi:10.1016/j.soilbio.2008.01.024
Wallenstein MD, McNulty S, Fernandez IJ, Boggs J, Schlesinger WH (2006) Nitrogen fertilization decreases forest soil fungal and bacterial biomass in three long-term experiments. Forest Ecol Manage 222:459–468. doi:10.1016/j.foreco.2005.11.002
Weaver PL (2007) Roble Blanco, White Cedar. US Forest Service Research Publications. http://www.na.fs.fed.us/pubs/silvics_manual/volume_2/tabebuia/heterophylla.htm. Accessed 23 June 2012
Acknowledgments
Funding was provided by an NSF Graduate Student Research Fellowship to M.M. Stone, and an Andrew Mellon foundation grant to A.H. Johnson. This work was completed under the NSF-funded Luquillo Critical Zone Observatory (LCZO; EAR-0722476) research program. We would like to thank J. McAuley for field assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Benjamin L. Turner.
Rights and permissions
About this article
Cite this article
Stone, M.M., Plante, A.F. & Casper, B.B. Plant and nutrient controls on microbial functional characteristics in a tropical Oxisol. Plant Soil 373, 893–905 (2013). https://doi.org/10.1007/s11104-013-1840-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-013-1840-8