Abstract
Background and aims
Root systems show considerable plasticity in their morphology and physiology in response to variability within their environment. Root elongation below a water-table was expected to slow due to hypoxia, whilst roots above the waterlogged zone were expected to compensate by increasing elongation rates.
Methods
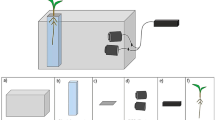
Tomato plants (Solanum lycopersicum L.) were grown in peat in root chambers (300 × 215 × 6 mm) with a transparent front. Root chambers were maintained in flatbed scanners tilted at 30° to vertical and scanned every 3 h before, during and after waterlogging the lower layer for 24 h or 5 days. Root elongation rates were calculated from the displacement of randomly selected root tips between successive scans. Oxygen content was determined in the waterlogged layer and plant and root parameters were determined at cessation of the experiment.
Results
Root elongation rates decreased rapidly when waterlogged. Growth rates of the waterlogged roots decreased, while growth rates of roots above the waterlogged zone increased. In 24 h waterlogged roots new lateral root growth occurred in the lower layer of the root chamber when water was drained while after 5 day waterlogging new root growth had to be initiated from roots above the waterlogged zone.
Conclusions
Plants increased growth rates in roots above the waterlogged zone probably as compensation for the suboptimal conditions in the waterlogged zone which eventually led to roots dying.





Similar content being viewed by others
References
Bengough AG, Gordon DC, Al-Menaie H, Ellis RP, Allan D, Keith R, Thomas WTB, Forster BP (2004) Gel observation chamber for rapid screening of root traits in cereal seedlings. Plant Soil 262:63–70
Bengough AG, McKenzie BM, Hallett PD, Valentine TA (2011) Root elongation, water stress, and mechanical impedance: a review of limiting stresses and beneficial root tip traits. J Exp Bot 62:59–68
Dat JF, Capelli N, Folzer H, Bourgeade P, Badot PM (2004) Sensing and signalling during plant flooding. Plant Physiol Biochem 42:273–282
Dresbøll DB, Thorup-Kristensen K (2012) Spatial variation in root system activity of tomato (Solanum lycopersicum L.) in response to short and long-term waterlogging as determined by 15N uptake. Plant Soil 357:161–172
Drew MC (1997) Oxygen deficiency and root metabolism: injury and acclimation under hypoxia and anoxia. Ann Rev Plant Physiol Plant Mol Biol 48:223–250
Eavis BW (1972) Soil physical conditions affecting seedling root growth 3. Comparisons between root growth in poorly aerated soil and at different oxygen partial pressures. Plant Soil 37:151–158
Fischer WR, Flessa H, Schaller G (1989) pH values and redox potentials in microsites of the rhizosphere. Z Pflanzenernähr Bodenkd 152:191–195
Gersani M, Sachs T (1992) Development correlations between roots in heterogeneous environments. Plant Cell Environ 15:463–469
Gonzalez JA, Gallardo M, Hilal M, Rosa M, Prado FE (2009) Physiological responses of quinoa (Chenopodium quinoa Willd.) to drought and waterlogging stresses: dry matter partitioning. Bot Stud 50:32–42
Gregory PJ, Bengough AG, Grinev D, Schmidt S, Thomas WTB, Wojciechowski T, Young IM (2009) Root phenomics of crops: opportunities and challenges. Funct Plant Biol 36:922–929
Hammac WA, Pan WL, Bolton RP, Koenig RT (2011) High resolution imaging to assess oilseed species’ root hair responses to soil water stress. Plant Soil 339:125–135
Hargreaves CE, Gregory PJ, Bengough AG (2009) Measuring root traits in barley (Hordeum vulgare ssp. vulgare and ssp. spontaneum) seedlings using gel chamber, soil sacs and X-ray microtomography. Plant Soil 316:285–297
Hodge A (2009) Root decisions. Plant Cell Environ 32:628–640
Hodge A, Robinson D, Griffiths BS, Fitter AH (1999) Why plants bother: root proliferation results in increased nitrogen capture from an organic patch when two grasses compete. Plant Cell Environ 22(7):811–820
Kläring HP, Zude M (2009) Sensing of tomato plant response to hypoxia in the root environment. Sci Hortic 122:17–25
Laan P, Clement JMAM, Blom CWPM (1991) Growth and development of rumex roots as affected by hypoxic and anoxic conditions. Plant Soil 136:145–151
Licausi F (2011) Regulation of the molecular response to oxygen limitations in plants. New Phytol 190:550–555
McKenzie BM, Bengough AG, Hallet PD, Thomas WTB, Forster B, McNicol JW (2009) Deep rooting and drought screening of cereal crops: a novel field-based method and its application. Field Crop Res 112:165–171
Morard P, Silvestre J (1996) Plant injury due to oxygen deficiency in the root environment of soilless culture: a review. Plant Soil 184:243–254
Morard P, Lacoste L, Silvestre J (2000) Effect of oxygen deficiency on uptake of water and mineral nutrients by tomato plants in soilless culture. J Plant Nutr 23:1063–1078
Pociecha E, Koscielniak J, Filek W (2008) Effects of root flooding and stage of development on the growth and photosynthesis of field bean (Vicia faba L. minor). Acta Physiol Plant 30:529–535
Veen BW (1988) Influence of oxygen deficiency on growth and function of plant roots. Plant Soil 111:259–266
Visser EJW, Voesenek LACJ (2004) Acclimation to soil flooding—sensing and signal-transduction. Plant Soil 274:197–214
Volder A, Anderson LJ, Smart DR, Bloom AJ, Lakso AN, Eissenstat DM (2009) Estimating nitrogen uptake of individual roots in container- and field-grown plants using a 15N-depletion approach. Funct Plant Biol 36:621–628
Wojciechowski T, Gooding MJ, Ramsay L, Gregory PJ (2009) The effects of dwarfing genes on seedling root growth of wheat. J Exp Bot 60:2565–2573
Yazdanbakhsh N, Fisahn J (2011) Stable diurnal growth rhythms modulate root elongation of Arabidopsis thaliana. Plant Roots 5:17–23
Zhu J, Ingram PA, Benfey PN, Elich T (2011) From lab to field, new approaches to phenotyping root system architecture. Curr Opin Plant Biol 14:310–317
Acknowledgments
We thank Helle Kjærsgaard Sørensen, Ruth Nielsen, Kaj Ole Didriksen and Connie Damgaard for skilful technical assistance. Funding was provided from The Danish Council for Independent Research | Technology and Production Sciences (Ref. no. 09-061045). The James Hutton Institute receives financial support from the Scottish Government.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Alain Pierret.
Rights and permissions
About this article
Cite this article
Dresbøll, D.B., Thorup-Kristensen, K., McKenzie, B.M. et al. Timelapse scanning reveals spatial variation in tomato (Solanum lycopersicum L.) root elongation rates during partial waterlogging. Plant Soil 369, 467–477 (2013). https://doi.org/10.1007/s11104-013-1592-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-013-1592-5